Journal:Anatomic pathology quality assurance: Developing an LIS-based tracking and documentation module for intradepartmental consultations
| Full article title | Anatomic pathology quality assurance: Developing an LIS-based tracking and documentation module for intradepartmental consultations |
|---|---|
| Journal | Journal of Pathology Informatics |
| Author(s) | Dundar, Bilge; Chen, Stephanie J.; Blau, John L.; Anand Rajan, K.D.; Samuelson, Megan I.; Greek-Lippe, Michelle L.; Lake, Kim S.; Robinson, Robert A. |
| Author affiliation(s) | University of Iowa Hospitals and Clinics |
| Primary contact | bilge-dundar at uiowa dot edu |
| Year published | 2022 |
| Volume and issue | 13 |
| Article # | 100109 |
| DOI | 10.1016/j.jpi.2022.100109 |
| ISSN | 2153-3539 |
| Distribution license | Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International |
| Website | https://www.sciencedirect.com/science/article/pii/S2153353922007039 |
| Download | https://www.sciencedirect.com/science/article/pii/S2153353922007039/pdfft (PDF) |
|
|
This article should be considered a work in progress and incomplete. Consider this article incomplete until this notice is removed. |
Abstract
Background: An electronic intradepartmental consultation system for anatomic pathology (AP) was conceived and developed in the laboratory information system (LIS) of University of Iowa Hospitals and Clinics in 2019. Previously, all surgical pathology intradepartmental consultative activities were initiated and documented with paper forms, which were circulated with the pertinent microscopic slides and were eventually filed. In this study, we discuss the implementation and utilization of an electronic intradepartmental AP consultation system.
Methods: Workflows and procedures were developed to organize intradepartmental surgical pathology consultations from the beginning to the end point of the consultative activities entirely using a paperless system that resided in the LIS.
Results: The electronic consult system allowed electronic documentation of all steps of intradepartmental consultative activities. The system provided a tracking ability for consulted cases and improved access to consult discussion for all departmental personnel, staff, and trainees. A consultation work queue was created for each pathologist, and a summary of individual consultative workload was made possible. Documentation of anatomic pathology quality assurance for intradepartmental consultative activity was easily assessed.
Conclusions: The electronic intradepartmental consult system has allowed our department to electronically track intradepartmental consult cases, store the consultative opinion text with the case, record the pathologists involved, and document the consultation for internal quality assurance review, as well as for accrediting organizations. Summarization of pathologist workload related to consultative activity was quantifiable, and optimization of the consultative process was maximized for education in an academic setting.
Keywords: electronic intradepartmental consultation, anatomic pathology, surgical pathology, laboratory information system
Introduction
Although relatively recent, the benefit of electronic consultation (e-consultation) has been noted across the spectrum in medicine. In the clinical setting, the availability of such consultation has decreased patient waiting times for specialist consultation and has had a particularly positive effect in patient populations that are underserved. [1,2] Surgical specialties have also noted increased efficiency in scheduling surgical intervention in their waiting lists when utilizing e-consultation. [3]
In anatomic pathology (AP), it has been shown that intra‐departmental consultations increase the value and diagnostic accuracy of reports and are vital steps to improve patient care. [4, 5, 6] However, while consultation in AP is long established, e-consultation is less well established.
The University of Iowa Hospitals and Clinics sought to improve its tracking and documentation process for AP intradepartmental consultations by using its laboratory information system (LIS), AP Beaker, in 2019. In this study, we describe the development, implementation, and utilization of an electronic intradepartmental AP consultation system and discuss its advantages and disadvantages.
Background
Institutional details and historical context of paper-based intradepartmental consultations
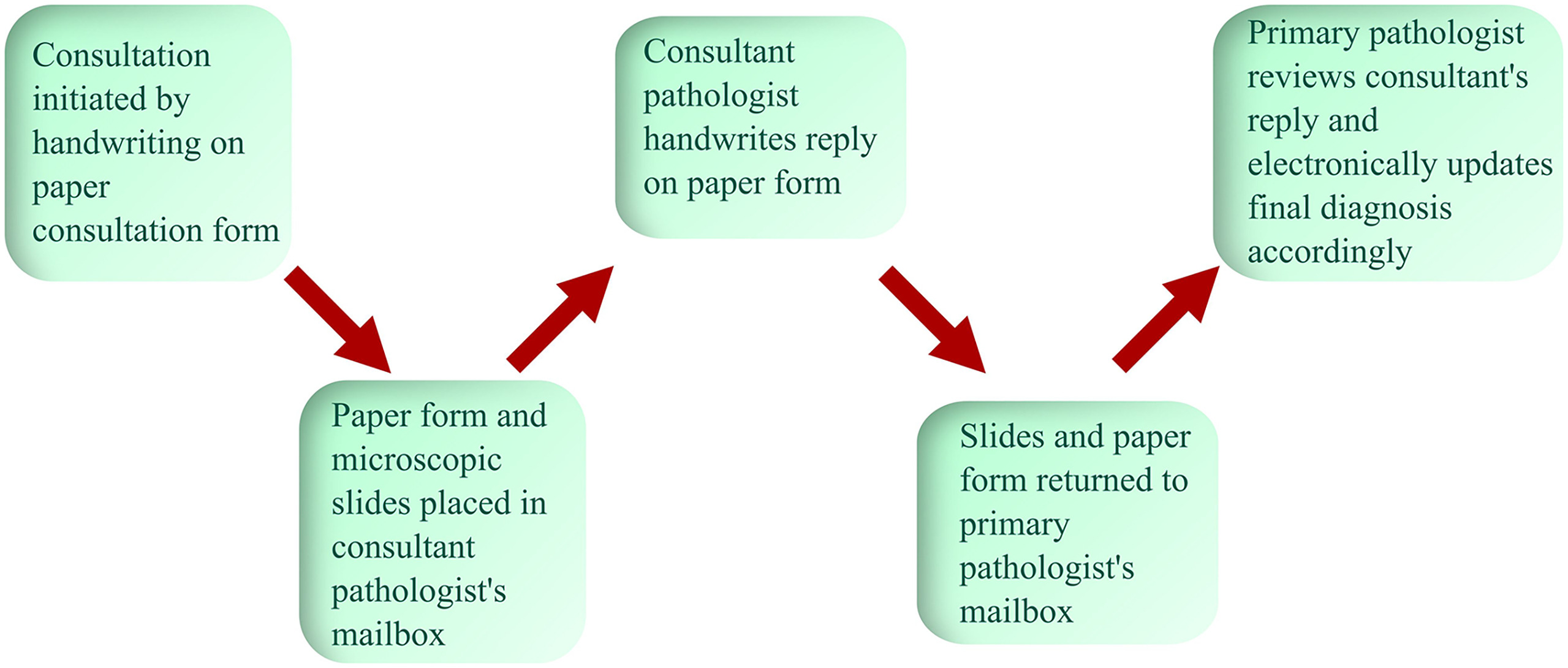
The Department of Pathology at our institution is a mid-sized academic pathology program (presently 36 clinical faculty, 20 residents, nine fellows) with a partially subspecialized service model in anatomic pathology, including renal pathology, hematopathology, dermatopathology, and neuropathology serving as dedicated subspecialty services. Gastrointestinal pathology and cytopathology services are essentially subspecialized. Prior to converting to an electronic form of consultation in AP, a long-standing paper system was in place and utilized despite the implementation of a now legacy electronic LIS (Cerner Corporation's PathNet) several decades ago and of the current AP LIS (Epic Systems Corporation's AP Beaker), which went live in 2015. Neither LIS had modules to implement or store intradepartmental consultation discussions and did not allow intradepartmental consultations to be performed electronically. In the paper-based workflow, an 8.5 x 11-inch form was colored yellow to stand out from other forms and reports (Fig. 1). Consultations were initiated by handwriting on the paper form. These consultation forms were placed with the slides and directed to the consultant faculty, who in turn wrote their consultative thoughts and returned the sheet with the slides to the primary pathologist (Fig. 2). The primary pathologist recorded the consulting pathologist in the final report in a dedicated data field in the LIS.
|
|
References
Notes
This presentation is faithful to the original, with only a few minor changes to presentation, grammar, and spelling. In some cases important information was missing from the references, and that information was added. No other modifications were made in accordance with the "no derivatives" portion of the distribution license.