Journal:Emerging and established trends to support secure health information exchange
| Full article title | Emerging and established trends to support secure health information exchange |
|---|---|
| Journal | Frontiers in Digital Health |
| Author(s) | Spanakis, Emmanouil G.; Sfakianakis, Stelios; Bonomi, Silvia; Ciccotelli, Claudio; Magalini, Sabina; Sakkalis, Vangelis |
| Author affiliation(s) | Foundation for Research and Technology, Sapienza Università di Roma, Fondazione Policlinico Universitario Agostino Gemelli |
| Primary contact | Email: spanakis at ics dot forth dot gr |
| Editors | Pattichis, Constantinos S. |
| Year published | 2021 |
| Volume and issue | 3 |
| Article # | 636082 |
| DOI | 10.3389/fdgth.2021.636082 |
| ISSN | 2673-253X |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://www.frontiersin.org/articles/10.3389/fdgth.2021.636082/full |
| Download | https://www.frontiersin.org/articles/10.3389/fdgth.2021.636082/pdf (PDF) |
|
|
This article should be considered a work in progress and incomplete. Consider this article incomplete until this notice is removed. |
Abstract
This work aims to provide information, guidelines, established practices and standards, and an extensive evaluation on new and promising technologies for the implementation of a secure information sharing platform for health-related data. We focus strictly on the technical aspects and specifically on the sharing of health information, studying innovative techniques for secure information sharing within the healthcare domain, and we describe our solution and evaluate the use of blockchain methodologically for integrating within our implementation. To do so, we analyze health information sharing within the concept of the PANACEA project, which facilitates the design, implementation, and deployment of a relevant platform. The research presented in this paper provides evidence and argumentation toward advanced and novel implementation strategies for a state-of-the-art information sharing environment; a description of high-level requirements for the national and cross-border transfer of data between different healthcare organizations; technologies to support the secure interconnectivity and trust between information technology (IT) systems participating in a data-sharing “community”; standards, guidelines, and interoperability specifications for implementing a common understanding and integration in the sharing of clinical information; and the use of cloud computing and prospectively more advanced technologies such as blockchain. The technologies described and the possible implementation approaches are presented in the design of an innovative secure information sharing platform in the healthcare domain.
Introduction
Information technology (IT) has long been identified as a cornerstone for efficient, cost-saving, timely, and reliable healthcare delivery.[1][2] The availability of healthcare information and patient records in digital form facilitates the persistence and posterity of valuable information and greatly supports the decision-making process, and even the extraction of new knowledge at both the individual and population levels. In our previous work, we have emphasized the current state of the art about cybersecurity in the healthcare domain, with emphasis on current threats and methodologies.[3] Paraphrasing the famous words of John Donne, “no IT system is an island, entire of itself.” Today, in a highly connected world where geographic boundaries have been largely eliminated and people can freely move between cities, states, countries, or continents, the requirement for two different information management systems to exchange a person's clinical data or medical history becomes vital and persistent. Sharing health information (e.g., via health information exchange [HIE]) through electronic means greatly improves the cost, quality, and patient experience of healthcare delivery.
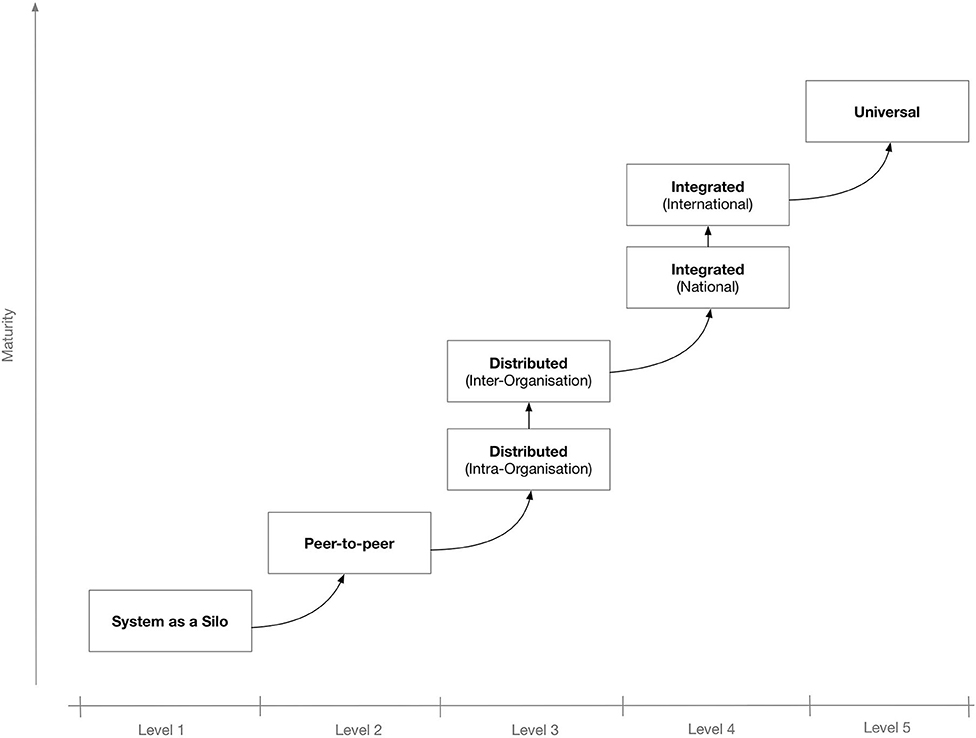
To better secure the IT system's potential for interconnectivity and cooperation with other systems, the use of interoperable technologies and standards is needed. Depending on the extent and scope of the envisaged shared information spaces, there may be different levels of interoperability. Figure 1 shows a proposed “maturity” model for interoperability in eHealth.[4] The model consists of five levels that, incrementally, describe a more mature version of an interoperable infrastructure, starting from Level 1 for non-connected eHealth applications; Level 2 where a single eHealth application is directly linked to another application for simple data exchange[5]; Level 3 for distributed systems that agree on protocols used, data formats, message exchange patterns, etc.[6]; Level 4, where eHealth applications from different suppliers that serve a common goal are linked but the applications do not need to have common objectives[7]; and finally, at the “universal” Level 5, where diverse eHealth applications connect to an open, interoperable infrastructure possibly spanning multiple countries.[8][9]
|
Interoperability and data sharing in the healthcare domain is additionally challenging due to the multiplicity of the stakeholders, that is, the entities that operate (or are involved in any way) in this domain and which will be affected by any “disruption” or reform of the system. Some of the most important stakeholders or actors are therefore the following:
- the patients who are actually treated or, in general, are the recipients of the health services;
- the medical professionals (physicians and medical personnel) to provide the medical care;
- the healthcare organizations (HCOs) (healthcare providers) as represented by their board of directors, who actually administer the health delivery from a business perspective;
- the insurance companies that provide health coverage plans;
- the pharmaceutical companies that produce and market medications to be prescribed by physicians for the treatment of patients; and
- the governments and other regulatory parties who control, coordinate, and set the rules, rights, and obligations of any involved party.
All these actors could have an influence in the design of a data sharing system and can also set important—and conflicting in some cases—requirements. For example:
- Patients would like to have their medical record shared, but only after their approval and only with specific authorized personnel in specific circumstances.
- An HCO can be extremely cautious about sharing the data of their patients with another organization because they are concerned by the security and availability of their systems.
- Governments of E.U. member states can impose strict laws about the transfer of their citizens in cross-border healthcare treatment scenarios.
- Medical professionals require fast and effortless access to a patient's medical history in emergency situations, which cannot be the case if time-consuming authorization processes are the norm.
Given these and similar requirements, and even though the objective is to design a technical solution for the sharing of clinical data, it is imperative that all these constraints and requirements are considered and addressed in a satisfying manner.
From a strictly technical point of view, the sharing platform may need to be interoperable with a large number and diverse set of IT systems, each with their own protocols, data formats, etc. Some of the most important systems that manage patient-related data, and could be used as data sources for information sharing, include electronic health records (EHRs), personal health records (PHRs), laboratory information systems (LISs), and picture archiving and communication systems (PACS). EHRs are patient-centered systems that store and manage clinical information, such as a patient's medical history, diagnoses, medications, immunization dates, allergies, radiology images, and lab and test results. They are managed by authorized personnel, usually in the context of a single HCO, although they can span beyond that. PHRs are electronic applications that are used by people managing their own health information in a private and confidential environment. They are simpler systems than EHRs, and in some cases, they can be connected (temporarily or otherwise) to more enterprise-level HCO systems (e.g., EHRs or other hospital information systems). An LIS is used inside hospitals and clinics to record, manage, and store data for clinical laboratories in a patient-centric way (e.g., sending laboratory test orders to lab instruments, tracking those orders, and then recording the results in a searchable database). And PACS are systems used in a clinical setting for the storage and convenient access to medical images from multiple modalities (source machine types). Digital Imaging and Communications in Medicine (DICOM) is the standard format and suite of protocols for the storage and transfer of images from PACS services. Of course, in a health ecosystem there may be additional systems, for example, for the management of insurance claims and for clinidal diagnoses (e.g., clinical decision support systems [CDSSs]).
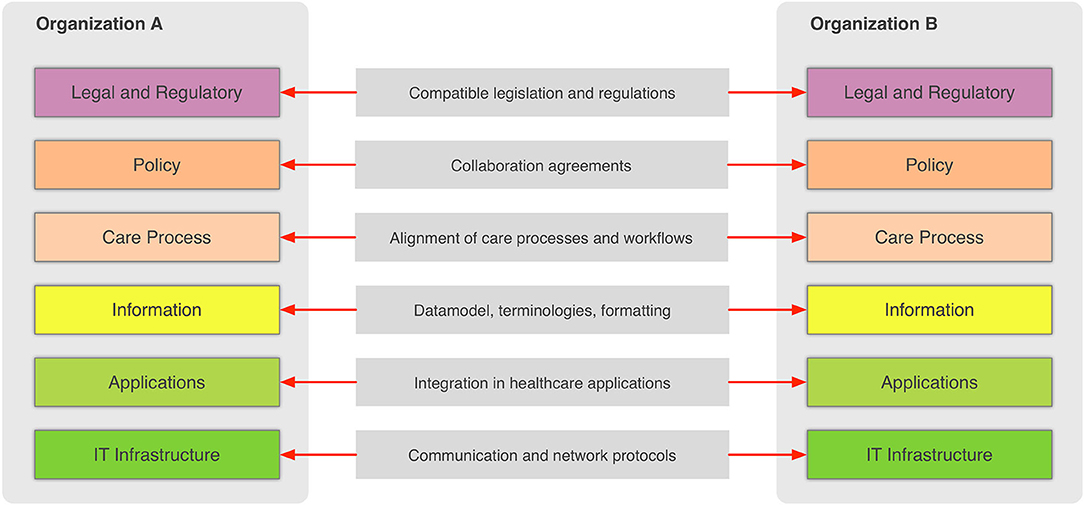
In the general context, information sharing involves more than one party (healthcare providers, organizations, etc.,) that need to cooperate and agree on the way the exchange of information happens, and what the rules and policies are that govern it. Interoperability involves many different aspects, such as legislation and guidelines, contracts, and agreements between exchanging parties, governance and maintenance, shareable workflows, standardized data elements, semantic and syntactic choices, applications, technical infrastructure, and safety and privacy issues. The Refined eHealth European Interoperability Framework (EIF) is a set of recommendations that specify the standards, protocols, procedures, and policies that when deployed can improve the interoperability of eHealth applications within the E.U. and across its member states by providing specific recommendations for all these aspects.[10] Figure 2 depicts how these aspects can be represented in interoperability “levels” that permit two different organizations to communicate.
|
This framework provides a great overview of the needed “glue” so that two or more healthcare environments can collaborate and serve as a common, multilevel, and multi-perspective model on the interoperability requirements. The six different levels of the (refined) EIF are:
- Legal and regulatory: This level represents the legislation and regulatory guidelines that define the boundaries for interoperability not only across borders, but also within a country or region.
- Policy: This level represents the contracts and agreements between the sharing organizations so that trust is established and responsibilities are assigned.
- Care process: This level represents the shared workflows that define how the integrated care is delivered and how these workflows are managed.
- Information: This level defines the data models, the concepts and their values, the terminologies, and controlled vocabularies that cater for the common understanding of the exchanged electronic messages.
- Applications: This level defines how the data are extracted from and imported to the healthcare information systems and how the transport of the data takes place using health-specific technologies and standards.
- IT infrastructure: This level represents the lowest level and corresponds to general-purpose communication and network protocols.
This generic EIF is highly relevant for the use cases of the health information sharing since sharing is greatly facilitated between interoperable systems and organizations. Here, we focus on the sharing of health-related information across HCOs and even across countries and continents. This is an important use case to improve the secure and efficient delivery of health care across Europe.[11][12] The importance of cross-border health care in Europe has been recognized since 2011's Directive 2011/24/EU, which established patients' rights to access safe and high-quality healthcare, including across national borders within the E.U., and their right to be reimbursed for such healthcare.[13] As can be deduced by considering the Refined eHealth EIF (Figure 2), the cross-border sharing of clinical information is a complex scenario due to the fact that data need to be transferred between different countries and therefore, requires overcoming barriers such as establishing a common trust framework, uniquely identifying citizens, and translating between different schemas and terminologies. This paper presents current approaches to address most of these issues in the European context while presenting and evaluating emerging technologies such as blockchain that have been in the limelight recently. Our objective is to take advantage of both well-established and novel technologies that complement each other in order to design an architecture for the secure exchange of clinical information in the European context.
Materials and methods
References
- ↑ U.S. Congress, Office of Technology Assessment (September 1995). "Bringing Health Care Online: The Role of Information Technologies" (PDF). U.S. Government Printing Office. https://ota.fas.org/reports/9507.pdf.
- ↑ Kolodner, R.M.; Cohn, S.P.; Freidman, C.P. (2008). "Health Information Technology: Strategic Initiatives, Real Progress". Health Affairs 27 (Supp. 1): w391–5. doi:10.1377/hlthaff.27.5.w391.
- ↑ Spanakis, E.G.; Bonomi, S.; Sfakianakis, S. (2020). "Cyber-attacks and threats for healthcare - A multi-layer thread analysis". Proceedings of the 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society: 5705–08. doi:10.1109/EMBC44109.2020.9176698. PMID 33019270.
- ↑ 4.0 4.1 Van Velsen, L.; Hermens, H.; d'Hollosy, O.-N. (2016). "A maturity model for interoperability in eHealth". Proceedings of the IEEE 18th International Conference on e-Health Networking, Applications and Services: 1–6. doi:10.1109/HealthCom.2016.7749533.
- ↑ Chronaki, C.E.; Chiarugi, F.; Mavrogiannaki, E. et al. (2003). "An eHealth platform for instant interaction among health professionals". Proceedings of the Computers in Cardiology 2003: 101–4. doi:10.1109/CIC.2003.1291100.
- ↑ Spanakis, M; Lelis, P.; Chiarugi, F. et al. (2005). "R&D Challenges in Developing an Ambient Intelligence eHealth Platform". IFMBE Proceedings 11 (1): 1727–983. http://139.91.210.27/CBML/PROCEEDINGS/2005_EMBEC/Embec%202005/Abstracts/Abstract791.html.
- ↑ Tsiknakis, M.; Spanakis, M. (2010). "Adoption of innovative eHealth services in prehospital emergency management: A case study". Proceedings of the 10th IEEE International Conference on Information Technology and Applications in Biomedicine: 1–5. doi:10.1109/ITAB.2010.5687752.
- ↑ Spanakis, E.G.; Psaraki, M.; Sakkalis, V. (2018). "Congestive Heart Failure Risk Assessment Monitoring through Internet of things and mobile Personal Health Systems". Proceedings of the 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society: 2925-28. doi:10.1109/EMBC.2018.8513024. PMID 30441013.
- ↑ Spanakis, M.; Sfakianakas, S.; Sakkalis, V. et al. (2019). "PharmActa: Empowering Patients to Avoid Clinical Significant Drug⁻Herb Interactions". Medicines 6 (1): 26. doi:10.3390/medicines6010026. PMC PMC6473432. PMID 30781500. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6473432.
- ↑ "eHealth Network: Refined eHealth European Interoperability Framework" (PDF). European Union. 23 November 2015. pp. 24. https://ec.europa.eu/health/sites/default/files/ehealth/docs/ev_20151123_co03_en.pdf.
- ↑ Katehakis, D.G.; Pangalos, G.; Prentza, A. (2017). "Security Improvements for Better and Safer Cross-Border ePrescription and Patient Summary Services". International Journal of Reliable and Quality E-Healthcare 6 (1): 2. doi:10.4018/IJRQEH.2017010102.
- ↑ Tinholt, D.; Carrara, W.; Tol, T. et al. (18 June 2013). "Final Report - Study on Analysis of the Needs for Cross-Border Services and Assessment of the Organisational, Legal, Technical and Semantic Barriers (SMART 2011/0074)". European Commission. doi:10.2759/10003. https://ec.europa.eu/digital-single-market/en/news/final-report-study-analysis-needs-cross-border-services-and-assessment-organisational-legal.
- ↑ Peeters, M. (2012). "Free movement of patients: Directive 2011/24 on the application of patients' rights in cross-border healthcare". European Journal of Health Law 19 (1): 29–60. doi:10.1163/157180912x615158. PMID 22428388.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation, though grammar and word usage was substantially updated for improved readability. In some cases important information was missing from the references, and that information was added.