Journal:Development and validation of a fast gas chromatography–mass spectrometry method for the determination of cannabinoids in Cannabis sativa L
| Full article title |
Development and validation of a fast gas chromatography–mass spectrometry method for the determination of cannabinoids in Cannabis sativa L |
|---|---|
| Journal | Journal of Food and Drug Analysis |
| Author(s) | Cardenia, Vladimiro; Toschi, Tullia G.; Scappini, Simona; Rubino, Rosamaria C.; Rodriguez-Estrada, Maria T. |
| Author affiliation(s) | University of Bologna, Enecta B.V. |
| Primary contact | Email: tullia dot gallinatoschi at unibo dot it |
| Year published | 2018 |
| Volume and issue | 26(4) |
| Page(s) | 1283–92 |
| DOI | 10.1016/j.jfda.2018.06.001 |
| ISSN | 1021-9498 |
| Distribution license | Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International |
| Website | https://www.sciencedirect.com/science/article/pii/S1021949818301066 |
| Download | https://www.sciencedirect.com/science/article/pii/S1021949818301066/pdfft (PDF) |
Abstract
A routine method for determining cannabinoids in Cannabis sativa L. inflorescence, based on fast gas chromatography coupled to mass spectrometry (fast GC-MS), was developed and validated. To avoid the decarboxylation of the carboxyl group of cannabinoids, different derivatization approaches—i.e., silylation and esterification (diazomethane-mediated) reagents and solvents (pyridine or ethyl acetate)—were tested. The methylation significantly increased the signal-to-noise ratio of all carboxylic cannabinoids, except for cannabigerolic acid (CBGA). Since diazomethane is not commercially available, is considered a hazardous reactive, and requires one-day synthesis by specialized chemical staff, the process of silylation was used along the entire validation of a routine method. The method gave a fast (total analysis time < 7.0 min) and satisfactory resolution (R > 1.1), with a good repeatability (intraday < 8.38%; interday < 11.10%) and sensitivity (LOD < 11.20 ng/mL). The suitability of the fast GC-MS method for detection of cannabinoids in hemp inflorescences was tested; a good repeatability (intraday < 9.80%; interday < 8.63%), sensitivity (LOD < 58.89 ng/mg), and robustness (< 9.52%) was also obtained. In the analyzed samples, the main cannabinoid was cannabidiolic acid (CBDA; 5.19 ± 0.58 g/100 g), followed by cannabidiol (CBD; 1.56 ± 0.03 g/100 g) and CBGA (0.83 g/100 g). Δ9-tetrahydrocannabivarin (THCV) was present at trace levels. Therefore, the developed fast GC-MS method could be a valid, routine alternative for a fast, robust, and highly sensitive determination of the main cannabinoids present in hemp inflorescences.
Keywords: cannabinoids, cannabis, decarboxylation, fast gas chromatography, methylation
Graphical abstract
Introduction
Recently, the interest on Cannabis sativa L. has drastically increased. However, attention has primarily been given to addressing its psychoactive[1] and non-psychoactive compounds, such as Δ9-tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD). In the past, the genus Cannabis was allocated into three main species: a drug-type (C. indica) with high levels of Δ9-THC, a fiber-type (C. sativa L.) with low levels of Δ9-THC, and an intermediate type C. ruderalis Janish.[2] More recently, the different Cannabis species have been divided into two broad types: C. sativa or “hemp” when referring to industrial use (fiber-type), and therapeutic “marijuana” (drug-type) when referring to varieties with a high level of Δ9-THC (>0.6%; w/w). To date, the main use for hemp is largely related to food; in fact, hemp seeds are generally used for producing oil and flour, and, depending on the country's local regulations, they may also be employed on the basis of their pharmacological properties.[3] However, hemp contains more than 500 different cannabinoids, of which about 10 have been classified according to their chemical structure, such as Δ9-tetrahydrocannabivarin (THCV), cannabidiol (CBD), cannabigerol (CBG), Δ8-tetrahydrocannabinol (Δ8-THC), Δ9-tetrahydrocannabinol (Δ9-THC), cannabichromene (CBC), cannabinol (CBN), cannabidiolic acid (CBDA), Δ9-tetrahydrocannabinolic acid (THCA), and cannabigerolic acid (CBGA).[4]
Hemp cannabinoids exhibit diverse biological effects. THCV displays various pharmacological profiles according to the type of molecular target (in vitro antagonistic/inverse agonistic effects and an in vivo agonism effect in an antinociception model).[5] The application of CBD for intractable pediatric epilepsy has also been recently studied.[6] On the other hand, CBC, which is particularly present in freshly harvested C. sativa, normalizes in vivo intestinal motility when intestinal inflammation occurs.[7] It should be pointed out that C. sativa does not produce Δ9-THC, CBD, CBG, and CBC, but their respective carboxylic acid forms (precursors) Δ9-THCA, CBDA, CBGA, and CBCA can undergo decarboxylation by heating or drying and thus exhibit their corresponding biological effects.[3] The galenic preparations of cannabis (such as medicinal oils), which are important for their possibility of being employed as a whole set of cannabinoids, are characterized by a high variability[8] and require a robust, simple quality control method for their titration.
Considering these and other biological effects of cannabinoids, their analysis in cannabis is of great interest and importance. There are several analytical methods for determining cannabinoids, most of which use gas chromatography coupled to mass spectrometry (GC-MS) or a flame ionization detector (GC-FID), or high-performance liquid chromatography coupled to mass spectrometry (LC-MS) or an ultraviolet detector (LC-UV).[2][3][4][9][10][11]
When GC-MS is used, the electron impact ionization (EI) generates mass spectra, which can be compared with those present in compound libraries for their identification. However, with LC-MS and electrospray (ESI) and atmospheric pressure chemical ionization (APCI), only molecular ions are generated, without other useful fragments for compound characterization; as such, expensive equipment able to perform tandem mass spectrometry (MS-MS) experiments is required.[12][13] As reported in literature, LC-MS sensitivity is lower than that of GC-MS.[4] However, there is a lot of criticism around the use of GC for cannabinoid analysis, since the high temperature of both injector and detector lead to decarboxylation of cannabinoid acids if not previously derivatized (e.g., using a process such as silylation).[14][15] Different silylation procedures have been reported for this scope; Purschke et al.[16] utilized N-methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA), while other researchers have used the combination of N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA) or MSTFA, with either trimethylchlorosilane (TMCS) or ethyl acetate.[17] However, no data are reported about esterification of cannabinoid carboxylic acids by diazomethane and the use of fast GC-MS for cannabinoid determination. Fast GC-MS has been demonstrated to provide the advantages of mass spectrometry boosted by the utilization of fast chromatography. In fact, the use of fast GC-MS drastically reduces the time of analysis without impairing sensitivity, resolution, and other analytical parameters (such as repeatability and reproducibility). Fast GC-MS has been successfully utilized for the determination of cholesterol oxidation products in 3.5 min[18], phytosterols and phytostanols in milk dairy products in less than 10 min[19], and heroin and cocaine in 3 min.[20]
To the best of our knowledge, no previous works have been published on the determination of cannabinoids in hemp inflorescences by fast GC-MS. The aim of this work was to develop and validate a fast GC-MS method for determining the main cannabinoids (CBD, CBDA, CBG, CBGA, Δ9-THC, Δ8-THC, THCA, THCV, CBC, and CBN) in hemp inflorescences, as related to different derivatization reagents (through silylation and esterification).
Materials and methods
Reagents and solvents
Chloroform, n-hexane, methanol, and ethanol were purchased from Merck (Darmstadt, Germany). N° 1 filters (70 mm diameter) were used (Whatmann, Maidstone, England). N,O-Bis(trimethylsilyl)trifluoroacetamide with trimethylchlorosilane (BSTFA:TMCS, 99:1, v/v), N-methyl-N-(trimethylsilyl)trifluoroacetamide:trimethylchlorosilane (MSTFA:TMCS, 99:1, v/v), and (trimethylsilyl)diazomethane solution were supplied by Sigma Aldrich (Germany). Certified phytocannabinoid mixture 1 (1 mg/mL in acetonitrile; containing CBD, CBDA, CBG, CBGA, Δ9-THC, Δ8-THC, THCA, and CBC at 100 μg/mL of each compound); (−)-Δ9-THC-D3 (THCd3, 0.1 mg/mL in methanol); THCA (1.0 mg/mL in methanol); and Δ9-THC (0.1 mg/mL in methanol) were purchased from LGC Standards S.r.L. (Milano, Italy). Millipore membrane filters (0.45 μm and 0.20 μm) were supplied by Merck (Germany).
Sampling
Three different batches of hemp inflorescences (E.U.-registered Cannabis sativa L. Futura 75 variety; fiber-type), harvested at different growing times (from middle July to the end of August 2017), were supplied by a local company (Green Valley Società Agricola S.R.L., Castelvecchio Subequo, Italy). Each batch was comprised of three independent samples (n = 3), where each sample included 10 inflorescences from 10 different plants (n = 10). Before performing cannabinoids extraction, the collected samples were dried by natural ventilation at 32 ± 1 °C for 60 h. Afterwards, dried hemp's apical and lateral peaks were sifted. The seeds with diameter >1 mm were removed by sieving. Finally, the material was ground with an analytical mill to obtain a homogeneous sample. The powdered samples were stored at −22 °C under nitrogen atmosphere until the analysis.
Extraction
Twenty five milligrams of ground sample were weighed into a glass test tube and 1.5 mg of 5α-cholestane (internal standard 1, IS1) were added. The extraction was performed using 10 mL of a 9:1 (v/v) methanol/chloroform mixture. The sample was stirred for 15 min (350 oscillations/min), sonicated for 10 min, centrifuged (5 min at 1620 g) and the solvent was collected. The extraction was repeated twice, and the surnatants were transferred into a 25 mL flask, which was made up to the flask volume with the same solvent. The extract was then filtered through a millipore filter (0.45 μm). One mL of the filtered extract was transferred to a glass tube that contained 0.5 μg of THCd3 (internal standard 2, IS2), taken to dryness under nitrogen flow and then derivatized.
Derivatization
Two different derivatization reactions were compared: methylation and silylation. For methylation, 1 mL of the filtered extract was added with IS2, dried under nitrogen flow, methylated with 300 μL of diazomethane, vortexed for 30 s and then dried under nitrogen flow. Silylation was then performed at 60 °C for 15 min, using 50 μL of pyridine and 150 μL of n-methyl-n-trimethylsilyltrifluoroacetamide + 1% of chlorotrimethylsilane (MSTFA-TMCS); the silylated sample was then dried at 40 °C and dissolved in 100 μL of n-hexane.
Fast gas chromatography–mass spectrometry (fast GC-MS) analysis
The cannabinoids were determined with a fast GC-MS Shimadzu QP 2010 Plus instrument (Kyoto, Japan) equipped with a Restek RTX 5 column (0.1 μm film thickness, 10 m × 0.1 mm); helium was used as the carrier gas (constant flow; linear velocity of 47.4 cm/s). The oven temperature was programmed from 180 °C (30 s) to 250 °C at 10 °C/min, and then to 350 °C (at 60 °C/min); final temperature was maintained for 5 min. The injector, interface, and ion source temperatures were 300, 330, and 200 °C, respectively, while the filament voltage was 70 eV (electronic impact). One μL of derivatized sample was manually injected (split 1:30).
Validation of the method
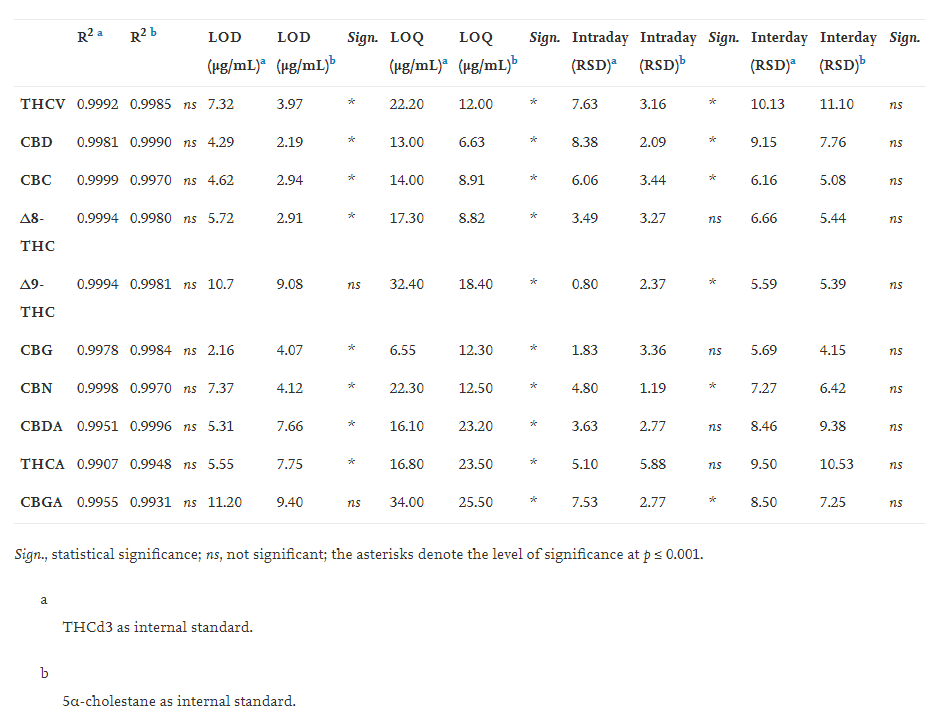
The response linearity was evaluated by means of calibration curves. For each compound, a calibration curve in the concentration range of 0.25 ng/mL–25 μg/mL was built using the internal standard method. Six different concentration levels were tested in triplicates. The cannabinoids were recognized by their mass spectra and were quantified by single ion monitoring (SIM). In particular, one quantifier ion and three qualifier ions were used (Table 1) for both derivatization methods (methylation and silylation).
| |||||||||||||||||||||||||||||||||||||||||||||||||||
The chromatographic peak resolution was determined on a critical pair (CBC and Δ8-THC), according to the following expression:
R = 2(tR2 – TR1) / (w1 + w2)
where tR is the retention time of the chromatographic peak and w is the peak width at its base level. The sensitivity and repeatability precision (intraday and interday) of the method on both standard mixture and matrix were determined. The reproducibility and recoveries on hemp inflorescences were estimated.
The limit of detection and quantification (LOD and LOQ, respectively) were calculated by the signal-to-noise ratio (S/N); LOD was expressed as S/N of 3.3:1, whereas an S/N of 10:1 was used for LOQ. The intraday and interday precision of fast GC–MS, expressed as relative standard deviation (RSD; %) was calculated by manually injecting samples (n = 3) in the same day (intraday precision) for three consecutive days (interday precision, n = 9). The injections were performed by different operators for testing the method precision. The recoveries of cannabinoids were estimated at two spiking levels of phytocannabinoids standard mixture (25 ng/mL [A]; 25.0 μg/mL [B]) in hemp inflorescence, using the following equation:
% recovery = [Cfc - Cc) / Cf] x 100
where Cfc is the cannabinoid amount found in the spiked sample, Cc is the cannabinoid amount present in the unspiked sample, and Cf is the spiked amount of cannabinoid standards. Three independent replicates (n = 3) were run for spiked and non-spiked hemp.
The method robustness (RSD; %) was assessed by determining cannabinoids in hemp inflorescence (from their extraction to the fast GC-MS analysis) in triplicates by two different operators.
Statistical analysis
Statistical analysis of data was performed by SPSS 21.0 (IBM-SPSS Inc., Chicago, Illinois, USA). Analysis of variance (ANOVA) was carried out to investigate the effect of derivatization reagents, solvents, and analytical conditions. Tukey's honest significance test and T-test were carried out at a 99% confidence level to separate means of parameters. P-values under the significance level of 0.001 were considered statistically significant.
Results and discussion
Derivatization study
As reported in literature[4], acid cannabinoids are converted into free cannabinoids (such as CBD, CBG, and Δ9-THC) after exposure to high temperature. In fact, decarboxylation is an important reaction for efficient production of the major active components in cannabis; however, it represents an important factor to be considered when the determination of cannabinoids is to be performed by GC. The high temperature of the injector and detector could lead to significant component loss, and thus it is important to prevent their decarboxylation when both acid and free cannabinoids determination is to be carried out by GC. To date, the main strategy has been to silanize acid cannabinoids.[13][16] However, to our knowledge, no data about the methylation of cannabinoid carboxylic acids has been reported. Therefore, on the basis of published GC methods[16][21][22][23], the silylation reaction of cannabinoids was compared to methylation with diazomethane. In order to define the best conditions of silylation, different variables were evaluated. According to previous studies, both MSTFA and BSTFA with 1% of TMCS (MSTFA-TMCS and BSTFA-TMCS, respectively) were compared. In addition, since the presence of pyridine or ethyl acetate could increase the silylation yield[24][25][26], the effect of the solvent was also tested.
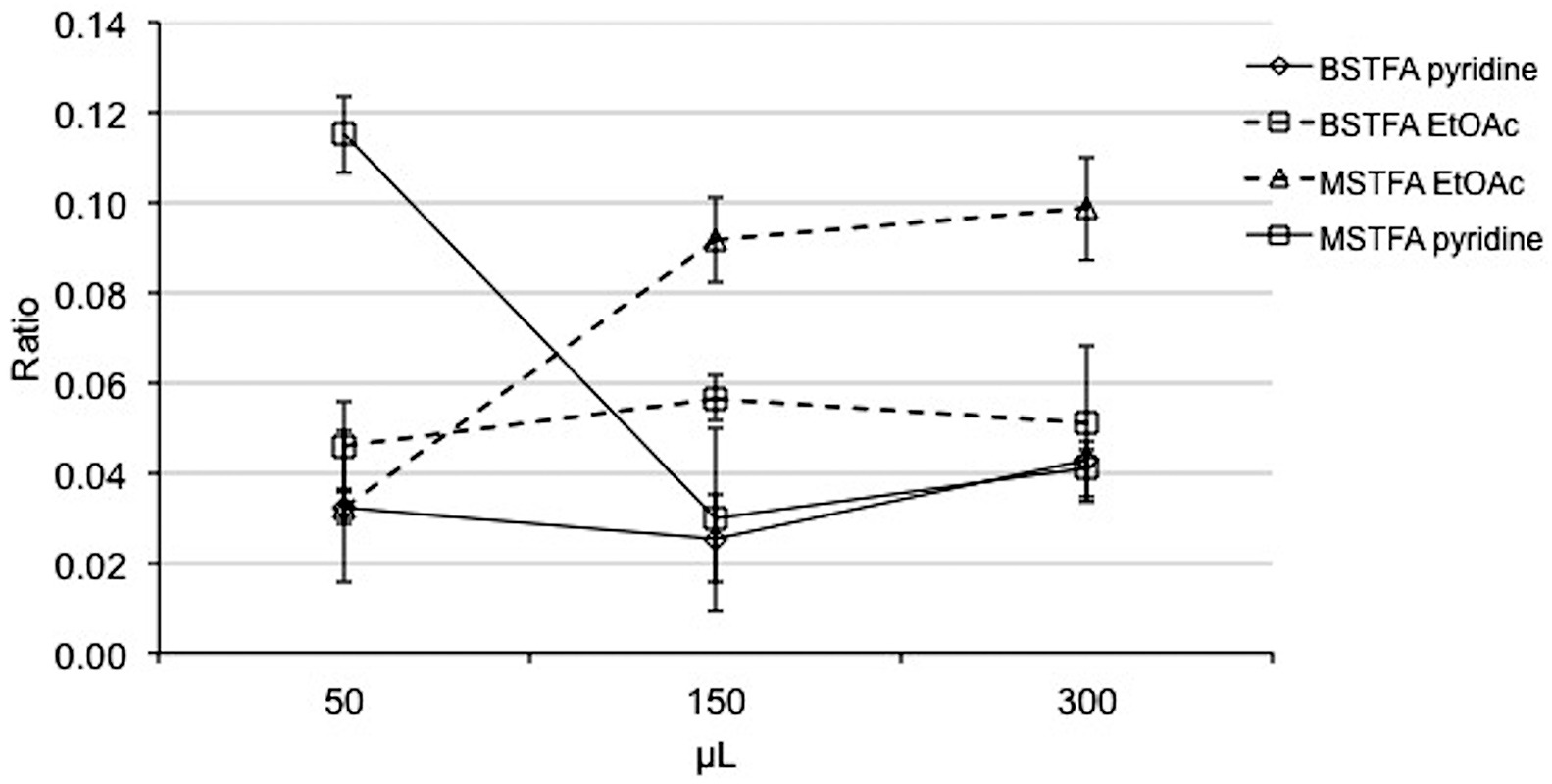
A fixed amount of Δ9-THCA standard (0.5 μg) and THCd3 standard (1 μg; internal standard, IS) reacted with 150 μL of MSTFA-TMCS or BSTFA-TMCS, in presence of an increasing amount (50, 150, or 300 μL) of pyridine or ethyl acetate at 60 °C for 30 min. The solvent was then evaporated and the sample was dissolved in 100 μL of n-hexane and injected (1 μL) into the fast GC-MS. The reaction efficiency was estimated by means of the AreaTHCA/AreaIS ratio. As reported in Fig. 1, the highest response was obtained with the lowest amount of pyridine, while the ethyl acetate led to highest reaction yields when 150 and 300 μL of solvent were used. However, the use of ethyl acetate gave a lower response than that achieved with 50 μL of pyridine. Moreover, when the reaction time was reduced to 15 min, the highest reaction yield was reached (data not shown).
|
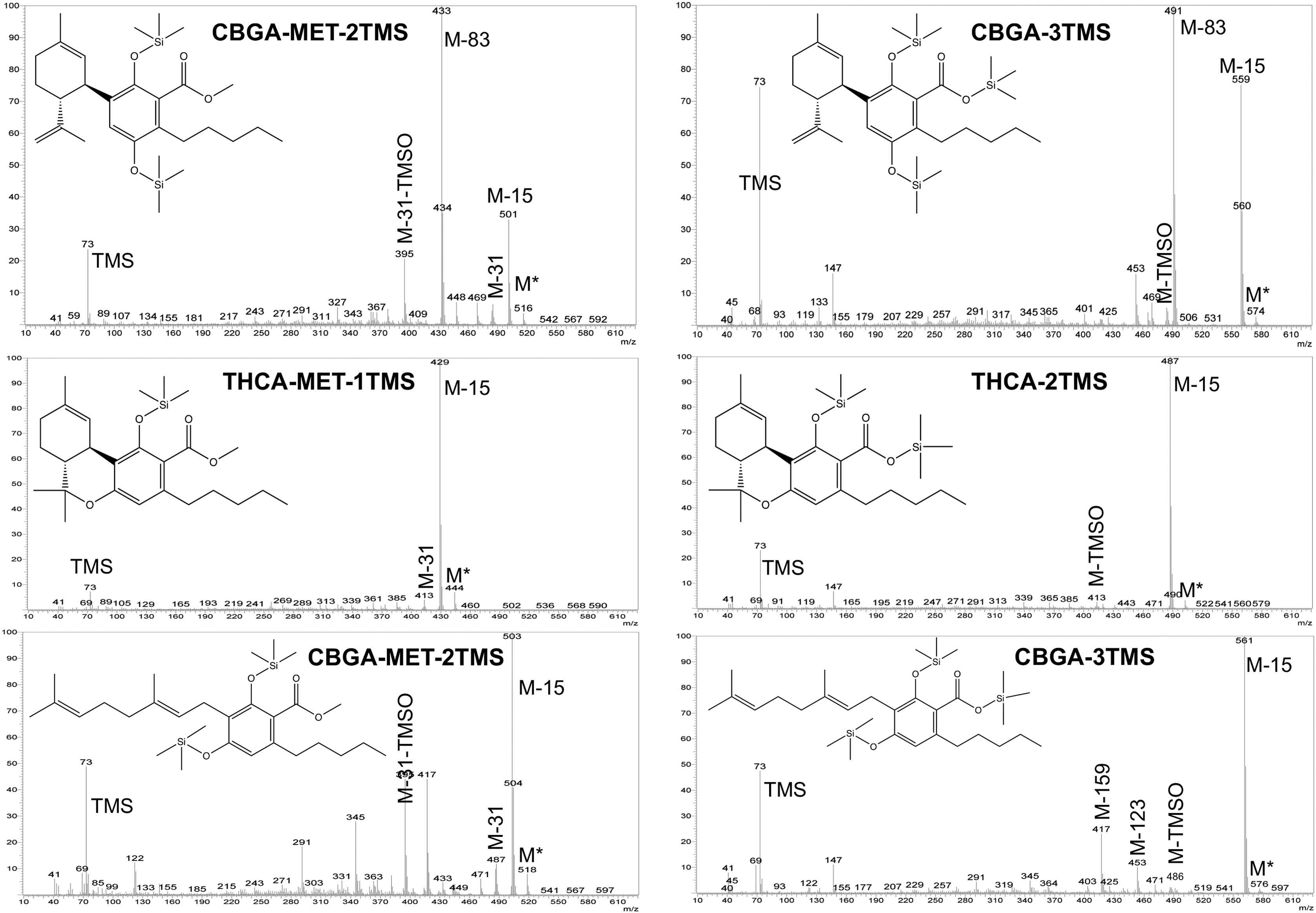
On the other hand, the carboxylic group of the cannabinoid acids could be also esterified by using diazomethane. Fig. 2 compares the mass spectra of CBDA, THCA, and CBGA, respectively, obtained by silylation with MSTFA-TMCS at 60 °C for 15 min, as well as by methylation-silylation (MET-TMS). As expected, diverse mass spectra were obtained according to the different derivatization process used. To the best of our knowledge, this is the first study that reports the mass spectra of methylated carboxylic cannabinoids. In general, for both methylated-silylated (MET-TMS) and silylated (TMS) cannabinoid acids, the molecular ion (M*) showed a low intensity. The M−15 fragment, which corresponds to a methyl group loss, was generally present in both MET-TMS and TMS cannabinoid acids, representing the base peak (100% of relative abundance) for THCA and CBGA (Fig. 2). In CBDA, instead, the M−83 fragment was its base peak, which corresponds to the loss of the side-chain (Fig. 2). In addition, the 73 m/z fragment (TMS group) was more noticeable in TMS cannabinoid acids than in MET-TMS cannabinoids, while M* tended to be higher in MET-TMS cannabinoids. In particular, the MET-TMS acid cannabinoids mass spectra were characterized by the presence of M−31 m/z (—OCH3) and M-31-TMSO. Furthermore, the TMS cannabinoid acid mass spectra were depicted by the presence of the M-TMSO fragment. In addition, as shown in Fig. 2, the M−15 fragment in CBDA-MET-2TMS was less intense than in CBDA-3TMS. Regarding CBGA, similar fragmentation was obtained with the two derivatization procedures, but with different relative intensity of the m/z fragments. As evidenced by these results, the derivatization reagent could thus affect the signal-to-noise ratio, impacting the method sensitivity.
|
The response ratio (Areacannabinoid/AreaIS) of all 10 cannabinoids obtained by methylation-silylation vs. silylation with MSTFA-TMCS was evaluated. Diazomethane generally increased the signal response, while esterification (MET-TMS) led to a significant response increase (p < 0.001) in CBD, Δ9-THC, CBG, CBN, CBDA, and THCA. This effect could be ascribed to the different reaction yield, as related to the steric hindrance of the derivatizing reagent functional group; probably the reaction of the carboxyl group with diazomethane is less affected by steric hindrance, thus leading to a complete esterification. When just silylation was considered (TMS), the contemporary presence of different TMSO groups could be disturbed by steric hindrance, and the reaction could generate diverse interferences, as reported by Van Look.[27] However, since diazomethane is not commercially available and requires one-day synthesis by specialized chemical technician, trimethylsilyldiazomethane, a similar derivatizing reagent, was also tested. However, when trimethylsilyldiazomethane was used, the signal-to-noise ratio dramatically decreased and the number of unidentified peaks (to be ascribed to reagent interferences) increased, which confirms the better suitability of diazomethane as derivatizing reagent.
Optimization of the separation by the fast GC-MS method
On the basis of the previously mentioned results and considering that diazomethane is not commercially available and is unsuitable for routine methods, the silylation with MSTFA-TMCS at 60 °C for 15 min was used for the entire method validation.
Literature reports that CBD, CBDA, CBGA, and CBC are the main cannabinoids present in hemp inflorescences[4]; however, it is of utmost importance to carry out an accurate determination of the other minor cannabinoids, such as Δ9-THC, Δ9-THCA, CBN, THCV, CBG, and Δ8-THC, which are generally present at levels that are about 100 times lower than CBD and CBDA. To overcome that problem, a double internal standard method was used, where 5α-cholestane was utilized to quantify high-concentration cannabinoids (i.e., CBD and CBDA in hemp) and THCd3 for the rest of the cannabinoids.
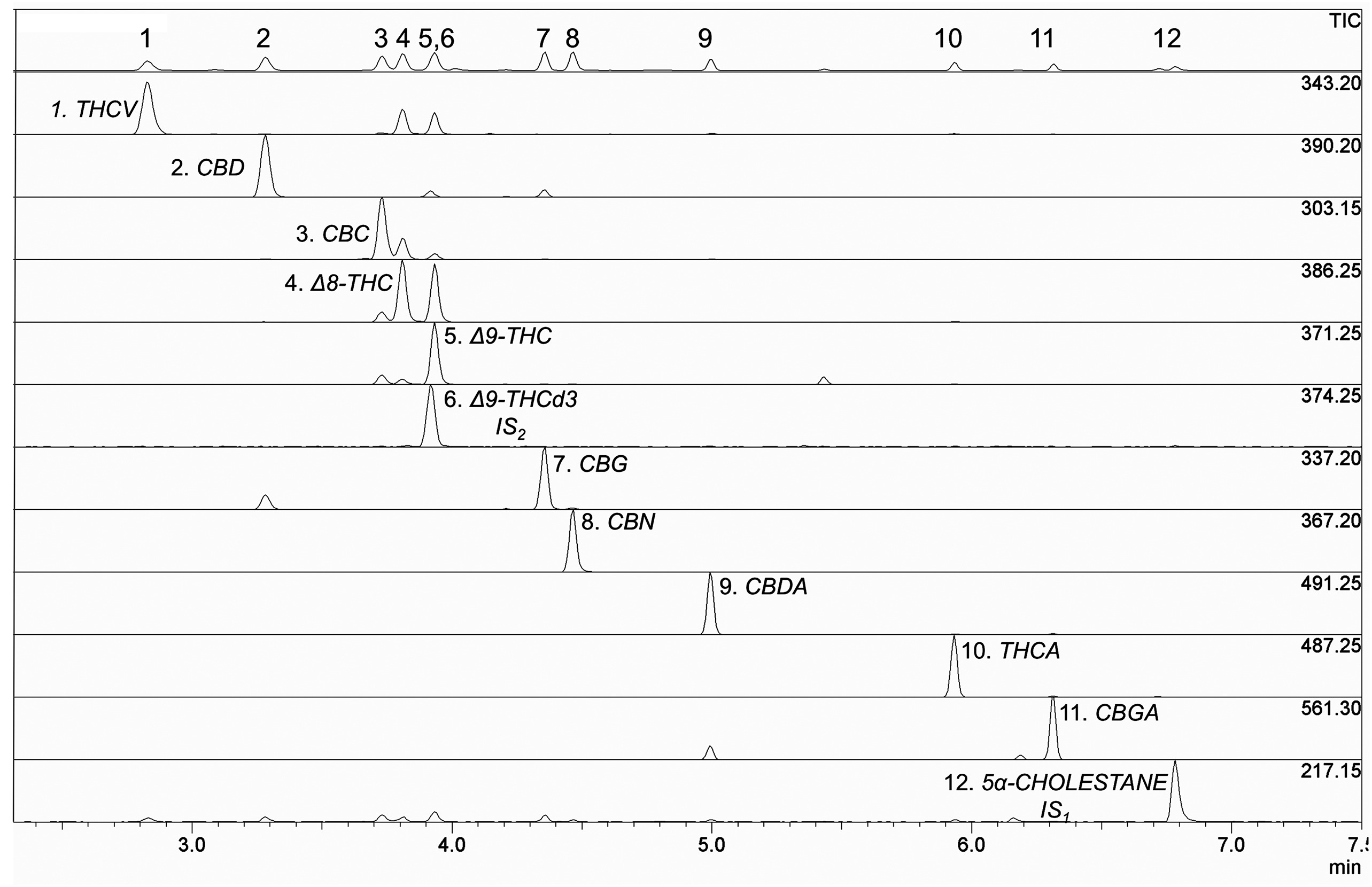
Using the analytical conditions reported in the "Materials and methods" section, all compounds were well separated in less than seven minutes, within a time frame of three minutes (Fig. 3). Each cannabinoid was recognized by using its characteristic mass fragmentation pattern (mass spectra) produced by EI. One of the main advantages of fast separation is that all cannabinoids are fully resolved on the baseline (except for the full overlap of THC with THCd3). Compare this to the use of HPLC analysis, where it is difficult to get a baseline resolution for three chromatographic pairs: Δ8-THC–Δ9-THC, CBDA–CBGA, and CBD–CBG.[3]
|
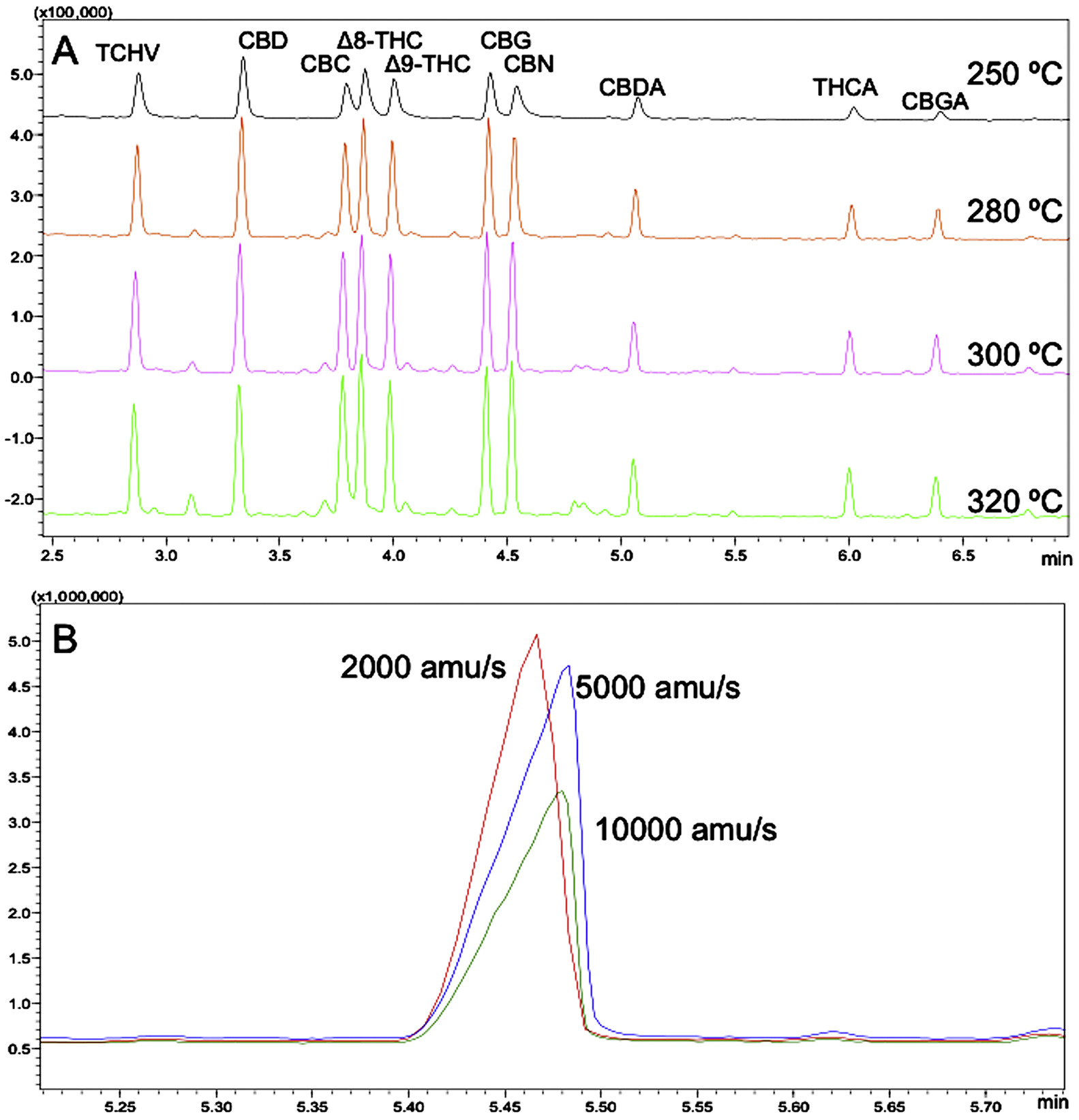
Different chromatographic and mass spectrometer conditions were tested, in order to obtain the final, optimized conditions. Several oven programs with different initial temperatures (180–250 °C), temperature increasing rates (3–25 °C/min), and linear velocities of carrier gas (0.45–1.50 mL/min), were examined; due to the thermo-sensitivity of cannabinoids, the effect of injector temperature was studied as well. As reported in Fig. 4A, when the injector temperatures was set up at 250 °C, the peak resolution decreased (R = 0.7), while higher temperatures improved peak resolution (R > 1.1) and signal intensity. However, the presence of unidentified peaks (i.e., matrix and/or reagent interferences) at higher temperature was observed. It should be noticed that the extraction method of cannabinoids was not selective, since high-boiling lipid molecules (such as triacylglycerols and sterols) were also present in the same extracted fraction. Thus, the risk of condensation phenomena of these high-boiling molecules in the injector is higher as the injector temperature decreases; for that reason, the injector temperature was set at 300 °C.
|
The acquisition frequency of the detector is a critical variable to be considered when a mass spectrometry analytical method is to be developed. As expected, the signal response was affected by the acquisition frequency; it is clear that, as the frequency increased from 2,000 to 10,000 amu/s, the peak symmetry and signal significantly changed, thus impacting the instrumental sensitivity (Fig. 4B). Considering the results and the peak resolution, a frequency of 2,000 amu/s was selected.
Method validation
The linearity of the fast GC-MS method was evaluated by analyzing the standard solutions of phytocannabinoids using two different internal standards: 5α-cholestane and THCd3. Three independent replicates (n = 3) of each concentration level of the calibration curve were analyzed. For each individual cannabinoid, a calibration curve was generated as related to the different internal standard used. As shown in Table 2, these curves displayed a linear behavior within the concentration ranges tested, having determination coefficients (R2) that varied from 0.9907 to 0.9999 when THCd3 was used and from 0.9931 to 0.9996 when 5α-cholestane was tested. However, no significant differences (p > 0.001) were found on the linearity as related to different internal standards. The sensitivity was determined as reported by Cardenia et al..[18] In general, the sensitivity was significantly affected by the internal standards used; indeed, when 5α-cholestane was tested, the LOD (2.19–9.40 μg/mL) was lower than those found using THCd3 (2.16–11.20 μg/mL), except for Δ9-THC and CBGA. As expected, the LOQ was also significantly affected by the different internal standards. The intraday and interday precision of the fast GC-MS method was calculated by manually injecting (with different operators) the phytocannabinoids standard solutions (n = 3) in the same day (intraday precision) for three consecutive days (interday precision, n = 9) (Table 2). Again, the use of 5α-cholestane as an internal standard significantly reduced the intraday and interday precision of the method. However, the results agree with literature data.[10]
|
The developed method reduced the analysis time, without losing sensitivity. The results obtained agree with available conventional GC methods[17], while the fast GC-MS method demonstrated greater sensitivity than HPLC methods[28], even though the use of an MS-MS experiment could drastically improve the method sensitivity.[12][29]
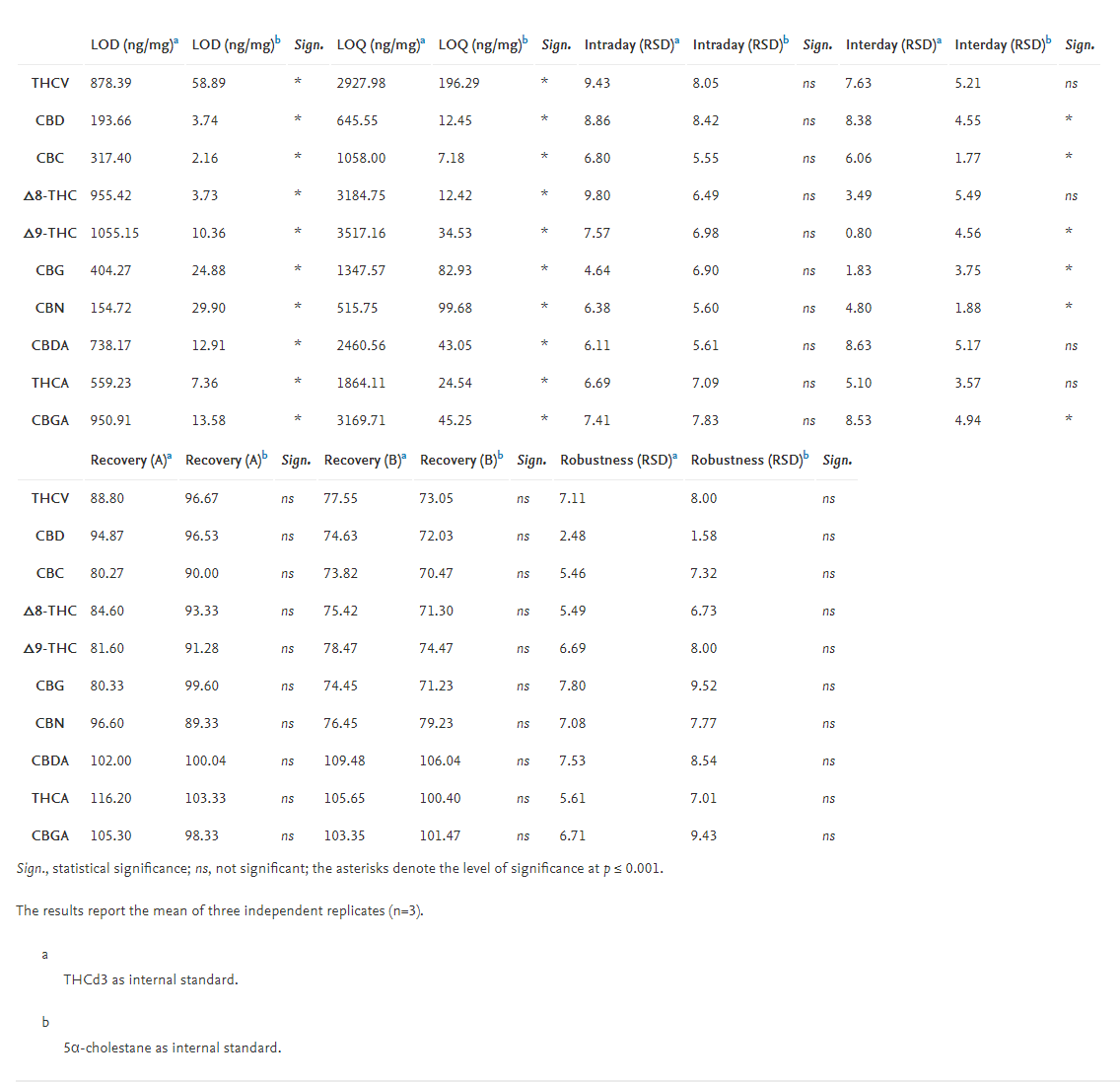
One sample of hemp inflorescence was used to assess the sensitivity, intraday and interday precision, recoveries, and robustness of the method. As reported in Table 3, higher sensitivity was obtained using 5α-cholestane as an internal standard, since both LOD (2.16 [CBC] – 58.86 [THCV] ng/mg inflorescence) and LOQ (7.18 [CBC] – 196.29 [THCV] ng/mg inflorescence) were relatively lower than those obtained using THCd3. However, the intraday repeatability was not affected by the different internal standards, whereas with 5α-cholestane the interday precision was lower for all cannabinoids, except for THCV, Δ8-THC, CBDA, and THCA. Again, the sensitivity of the developed method was higher than that reported for HPLC analysis of cannabinoids in inflorescences.[2] Nevertheless, 5α-cholestane is a saturated C27 tetracyclic triterpene, having thus a chemical structure slightly different from those of cannabinoids. In fact, 5α-cholestane does not have hydroxyl or carboxyl groups to be derivatized, which could explain the different results obtained when compared with THCd3 as an internal standard choice. Other chemicals have also been employed as internal standards for cannabinoid quantification, including prazepam, ibuprofen, diazepam, and di-n-octyl phatalate[9][17][27][29]; however, the use of deuterated cannabinoids represents a more powerful and expensive alternative.
|
The recoveries of cannabinoids in spiked industrial hemp inflorescence at two spiking levels (0.25 ng [A]; 25.00 μg [B]), were evaluated. As reported in Table 3, the recoveries at both spiked levels were not significantly affected by the different internal standards, thus confirming their suitability for quantification of cannabinoids in hemp. When 0.25 ng were spiked into hemp inflorescences, the recoveries were 80.3–116.2% (with THCd3) and 89.3–103.3% (with 5α-cholestane); spiking with 25.00 μg led to 73.8–109.5% and 70.5–106.0% recoveries when THCd3 and 5α-cholestane were used, respectively. These results agree with those reported in literature; Escrivá obtained recoveries that ranged from 97.2% to 109.6% by using HPLC-DAD[17], which were marginally higher than those reported by Brighenti et al.[2] (74–91%). Citti et al.[9] found recoveries that varied from 89.2% to 99.6% in hemp seed oil when using HPLC-MS (quadrupole time of flight [QToF]) and MS-MS.
The determination of cannabinoids in hemp inflorescence (considering the whole procedure, from their extraction to fast GC-MS analysis) performed in triplicates by two different operators, was used to assess the method robustness. No significant differences were found as related to different internal standards; the robustness was lower than 7.80% and 9.52% when THCd3 and 5α-cholestane were used as internal standards, respectively (Table 3). Therefore, the results confirm that the developed method can be used for routine determination of cannabinoids with a reduced analysis time and a less expensive internal standard (5α-cholestane) with respect to a deuterated one (THCd3).
Quantitative determination of cannabinoids in hemp inflorescences
The amount of cannabinoids can significantly change depending on pedo-climatic conditions, cultivation location, genetic variability, and harvesting time.[2] In order to evaluate a method's suitability for determining cannabinoids in hemp samples, three different batches of hemp inflorescences were supplied by a local company. Each batch was composed by three independent samples (n = 3), where each sample contained 10 inflorescences of 10 different plants.
The use of double internal standards allowed to reduce data dispersion, thus increasing the accuracy for a wide concentration range, from more concentrated cannabinoids (such as CBDA and CBD) to less concentrated (such as CBN) or traces (such as THCV). In agreement with literature, CBDA (5.2 ± 0.58 g/100 g) was the most abundant compound, followed by CBD (1.56 ± 0.03 g/100 g), CBGA (0.83 ± 0.10 g/100 g), and CBC (0.54 ± 0.01 g/100 g). Considering that CBGA is the precursor of other cannabinoids, as well as the low presence of CBG, it can be hypothesized that all samples were stored in a conservative way, thus preventing them from degrading.[30] On the other hand, the similar amounts detected for Δ9-THC, Δ8-THC, and CBN confirm the protective storage and treatment of hemp during the whole extraction and analytical procedure.
Conclusions
In conclusion, the fast GC-MS method herein proposed allows the quali-quantitative determination of the most common cannabinoids in short time (lower than 7.0 min), with a good resolution (R > 1.1). The method was set up and validated in-house using a commercial standard mixture testing Cannabis sativa L. This fast GC-MS method proved to be sensitive, with a high repeatability and robustness in both cannabinoid standard mixtures and hemp inflorescence samples. The analytical performance, together with the consequent significant reduction in the analysis time and consumables, demonstrates that fast GC-MS is a good alternative to other analytical and often expensive methods, and it evinces the great potential of such an analytical technique, which could be also applied for the routine analysis of cannabinoids in hemp inflorescences. These results contribute to the research on Cannabis sativa L. derivatives, which currently show a growing trend as a raw material for drugs, food, fiber, or building components. However, regardless of the final use of cannabis, a reliable and accurate identification and quantification of cannabinoids for the correct cannabis classification (fiber or drug type) still remains the crucial point from the legal standpoint.
Acknowledgements
The authors thank Enecta B.V. (Amsterdam, NL) for having supported the research and also for supplying samples through the collaboration with Green Valley Società Agricola A.R.L. (Castelvecchio Subequo, Italy).
Conflicts of interest
The authors declare that there are no conflicts of interest.
References
- ↑ Feng, L.Y.; Battulga, A.; Han, E. et al. (2017). "New psychoactive substances of natural origin: A brief review". Journal of Food and Drug Analysis 25 (3): 461–71. doi:10.1016/j.jfda.2017.04.001. PMID 28911631.
- ↑ 2.0 2.1 2.2 2.3 2.4 Brighenti, V.; Pellati, F.; Steinbach, M. et al. (2017). "Development of a new extraction technique and HPLC method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L. (hemp)". Journal of Pharmaceutical and Biomedical Analysis 143: 228–36. doi:10.1016/j.jpba.2017.05.049. PMID 28609672.
- ↑ 3.0 3.1 3.2 3.3 Citti, C.; Braghiroli, D.; Vandelli, M.A. et al. (2018). "Pharmaceutical and biomedical analysis of cannabinoids: A critical review". Journal of Pharmaceutical and Biomedical Analysis 147: 565-579. doi:10.1016/j.jpba.2017.06.003. PMID 28641906.
- ↑ 4.0 4.1 4.2 4.3 4.4 Leghissa, A.; Hildenbrand, Z.L.; Schug, K.A. et al. (2018). "A review of methods for the chemical characterization of cannabis natural products". Journal of Separation Science 41 (1): 398-415. doi:10.1002/jssc.201701003. PMID 28986974.
- ↑ McPartland, J.M.; Duncan, M.; Do Marzo, V. et al. (2015). "Are cannabidiol and Δ(9) -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review". British Journal of Pharmacology 172 (3): 737-53. doi:10.1111/bph.12944. PMC PMC4301686. PMID 25257544. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4301686.
- ↑ Tzadok, M.; Uliel-Siboni, S.; Linder, I. et al. (2016). "CBD-enriched medical cannabis for intractable pediatric epilepsy: The current Israeli experience". Seizure 35: 41–4. doi:10.1016/j.seizure.2016.01.004. PMID 26800377.
- ↑ Izzo, A.A.; Capasso, R.; Aviello, G. et al. (2012). "Inhibitory effect of cannabichromene, a major non-psychotropic cannabinoid extracted from Cannabis sativa, on inflammation-induced hypermotility in mice". British Journal of Pharmacology 166 (4): 1444–60. doi:10.1111/j.1476-5381.2012.01879.x. PMC PMC3417459. PMID 22300105. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3417459.
- ↑ Carcieri, C.; Tomasello, C.; Simiele, M. et al. (2018). "Cannabinoids concentration variability in cannabis olive oil galenic preparations". Journal of Pharmacy and Pharmacology 70 (1): 143–49. doi:10.1111/jphp.12845. PMID 29057480.
- ↑ 9.0 9.1 9.2 Citti, C.; Pacchetti, B.; Vandelli, M.A. et al. (2018). "Analysis of cannabinoids in commercial hemp seed oil and decarboxylation kinetics studies of cannabidiolic acid (CBDA)". Journal of Pharmaceutical and Biomedical Analysis 149: 532-540. doi:10.1016/j.jpba.2017.11.044. PMID 29182999.
- ↑ 10.0 10.1 Tsai, Y.-Y.; Tai, S.-J.; Huang, M.-C. et al. (1999). "Solid-phase extraction and gas chromatography-mass spectrometry analysis of 11-nor-9-carboxy-δ9-tetrahydrocannabinol in urine for monitoring marijuana abuse". Journal of Food and Drug Analysis 7: 177–84.
- ↑ Lin, S.Y.; Lee, H.H.; Lee, J.F. et al. (2018). "Urine specimen validity test for drug abuse testing in workplace and court settings". Journal of Food and Drug Analysis 26 (1): 380–84. doi:10.1016/j.jfda.2017.01.001. PMID 29389577.
- ↑ 12.0 12.1 Angeli, I.; Casati, S.; Ravelli, A. et al. (2018). "A novel single-step GC-MS/MS method for cannabinoids and 11-OH-THC metabolite analysis in hair". Journal of Pharmaceutical and Biomedical Analysis 155: 1–6. doi:10.1016/j.jpba.2018.03.031. PMID 29602054.
- ↑ 13.0 13.1 Pacifici, R.; Marchei, E.; Salvatore, F. et al. (2017). "Evaluation of cannabinoids concentration and stability in standardized preparations of cannabis tea and cannabis oil by ultra-high performance liquid chromatography tandem mass spectrometry". Clinical Chemistry and Laboratory Medicine 55 (10): 1555–1563. doi:10.1515/cclm-2016-1060. PMID 28207408.
- ↑ Mudge, E.M.; Murch, S.J.; Brown, P.N. (2017). "Leaner and greener analysis of cannabinoids". Analytical and Bioanalytical Chemistry 409 (12): 3153–63. doi:10.1007/s00216-017-0256-3. PMC PMC5395585. PMID 28233028. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5395585.
- ↑ Casiraghi, A.; Roda, G.; Casagni, E. et al. (2018). "Extraction Method and Analysis of Cannabinoids in Cannabis Olive Oil Preparations". Planta Medica 84 (4): 242–49. doi:10.1055/s-0043-123074. PMID 29202510.
- ↑ 16.0 16.1 16.2 Purschke, K.; Heinl, S.; Lerch, O. et al. (2016). "Development and validation of an automated liquid-liquid extraction GC/MS method for the determination of THC, 11-OH-THC, and free THC-carboxylic acid (THC-COOH) from blood serum". Analytical and Bioanalytical Chemistry 408 (16): 4379-88. doi:10.1007/s00216-016-9537-5. PMC PMC4875941. PMID 27116418. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4875941.
- ↑ 17.0 17.1 17.2 17.3 Escrivá, Ú.; Andrés-Costa, M.J.; Andreu, V. et al. (2017). "Analysis of cannabinoids by liquid chromatography-mass spectrometry in milk, liver and hemp seed to ensure food safety". Food Chemistry 228: 177–85. doi:10.1016/j.foodchem.2017.01.128. PMID 28317711.
- ↑ 18.0 18.1 Cardenia, V.; Rodriguez-Estrada, M.T.; Baldacci, E. et al. (2012). "Analysis of cholesterol oxidation products by Fast gas chromatography/mass spectrometry". Journal of Separation Science 35 (3): 424-30. doi:10.1002/jssc.201100660. PMID 22258809.
- ↑ Inchingolo, R.; Cardenia, V.; Rodriguez-Estrada, M.T. (2014). "Analysis of phytosterols and phytostanols in enriched dairy products by Fast gas chromatography with mass spectrometry". Journal of Separation Science 37 (20): 2911-9. doi:10.1002/jssc.201400322. PMID 25069661.
- ↑ Amirav, A. (2017). "Fast Heroin and Cocaine Analysis by GC–MS with Cold EI: The Important Role of Flow Programming". Chromatographia 80 (2): 295–300. doi:10.1007/s10337-017-3249-7.
- ↑ Emídio, E.S.; Prata Vde, M.; Dórea, H.S. (2010). "Validation of an analytical method for analysis of cannabinoids in hair by headspace solid-phase microextraction and gas chromatography-ion trap tandem mass spectrometry". Analytica Chimica Acta 670 (1–2): 63–71. doi:10.1016/j.aca.2010.04.023. PMID 20685418.
- ↑ Gasse, A.; Pfeiffer, H.; Köhler, H. et al. (2016). "Development and validation of a solid-phase extraction method using anion exchange sorbent for the analysis of cannabinoids in plasma and serum by gas chromatography-mass spectrometry". International Journal of Legal Medicine 130 (4): 967–74. doi:10.1007/s00414-016-1368-6. PMID 27072011.
- ↑ Aldlgan, A.A.; Torrance, H.J. (2016). "Bioanalytical methods for the determination of synthetic cannabinoids and metabolites in biological specimens". TrAC Trends in Analytical Chemistry 80: 444–57. doi:10.1016/j.trac.2016.03.025.
- ↑ Lachenmeier, D.W.; Kroener, L.; Musshoff, F. et al. (2004). "Determination of cannabinoids in hemp food products by use of headspace solid-phase microextraction and gas chromatography-mass spectrometry". Analytical and Bioanalytical Chemistry 378 (1): 183–9. doi:10.1007/s00216-003-2268-4. PMID 14598006.
- ↑ Nadal, X.; Del Río, C.; Casano, S. et al. (2017). "Tetrahydrocannabinolic acid is a potent PPARγ agonist with neuroprotective activity". British Journal of Pharmacology 174 (23): 4263-4276. doi:10.1111/bph.14019. PMC PMC5731255. PMID 28853159. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5731255.
- ↑ Citti, C.; Battisti, U.M.; Braghiroli, D. et al. (2018). "A Metabolomic Approach Applied to a Liquid Chromatography Coupled to High-Resolution Tandem Mass Spectrometry Method (HPLC-ESI-HRMS/MS): Towards the Comprehensive Evaluation of the Chemical Composition of Cannabis Medicinal Extracts". Phytochemical Analysis 29 (2): 144–55. doi:10.1002/pca.2722. PMID 28915313.
- ↑ 27.0 27.1 van Look, G. (1988). Silylating agents: Derivatization reagents, protecting-group reagents, organosilicon compounds, analytical applications, synthetic applications. Fluka Chemie AG. ISBN 9783905617023.
- ↑ Patel, B.; Wene, D.; Fan, Z.T. (2017). "Qualitative and quantitative measurement of cannabinoids in cannabis using modified HPLC/DAD method". Journal of Pharmaceutical and Biomedical Analysis 146: 15–23. doi:10.1016/j.jpba.2017.07.021. PMID 28841427.
- ↑ 29.0 29.1 Jamwal, R.; Topletz, A.R.; Ramratnam, B. et al. (2017). "Ultra-high performance liquid chromatography tandem mass-spectrometry for simple and simultaneous quantification of cannabinoids". Journal of Chromatography B 1048: 10–18. doi:10.1016/j.jchromb.2017.02.007. PMC PMC5697720. PMID 28192758. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5697720.
- ↑ Hanuš, L.O.; Meyer, S.M.; Muñoz, E. et al. (2016). "Phytocannabinoids: A unified critical inventory". Natural Product Reports 33 (12): 1357-1392. doi:10.1039/c6np00074f. PMID 27722705.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. Some grammar and punctuation was cleaned up to improve readability. In some cases important information was missing from the references, and that information was added. Everything else remains true to the original article, per the "NoDerivatives" portion of the distribution license.