Journal:The effect of a test ordering software intervention on the prescription of unnecessary laboratory tests - A randomized controlled trial
| Full article title | The effect of a test ordering software intervention on the prescription of unnecessary laboratory tests - A randomized controlled trial |
|---|---|
| Journal | BMC Medical Informatics and Decision Making |
| Author(s) |
Martins, C.M.; da Costa Teixeira, A.S.; de Azevedo. L.F.; Sá, L.M.; Santos, P.A.; do Couto, M.L.; da Costa Pereira, A.M.; Hespanhol, A.A.; da Costa Santos, C.M. |
| Author affiliation(s) | University of Porto |
| Primary contact | E-mail: carlosmartins20 at gmail dot com |
| Year published | 2017 |
| Volume and issue | 17 (1) |
| Page(s) | 20 |
| DOI | 10.1186/s12911-017-0416-6 |
| ISSN | 1472-6947 |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://bmcmedinformdecismak.biomedcentral.com/articles/10.1186/s12911-017-0416-6 |
| Download | https://bmcmedinformdecismak.biomedcentral.com/track/pdf/10.1186/s12911-017-0416-6 (PDF) |
Abstract
Background: The way electronic health record and laboratory test ordering system software is designed may influence physicians’ prescription. A randomized controlled trial was performed to measure the impact of a diagnostic and laboratory tests ordering system software modification.
Methods: Participants were family physicians working and prescribing diagnostic and laboratory tests.
The intervention group had modified software with basic shortcut menu changes, where some tests were withdrawn or added, and with the implementation of an evidence-based clinical decision support system based on United States Preventive Services Task Force (USPSTF) recommendations. This intervention group was compared with typically used software (control group).
The outcomes were the number of tests prescribed from those: withdrawn from the basic menu; added to the basic menu; marked with green dots (USPSTF’s grade A and B); and marked with red dots (USPSTF’s grade D).
Results: Comparing the monthly average number of tests prescribed before and after the software modification, from those tests that were withdrawn from the basic menu, the control group prescribed 33.8 tests per 100 consultations before and 30.8 after (p = 0075); the intervention group prescribed 31.3 before and 13.9 after (p < 0001). Comparing the tests prescribed between both groups during the intervention, from those tests that were withdrawn from the basic menu, the intervention group prescribed a monthly average of 14.0 vs. 29.3 tests per 100 consultations in the control group (p < 0.001). From those tests that are USPSTF’s grade A and B, the intervention group prescribed 66.8 vs. 74.1 tests per 100 consultations in the control group (p = 0.070). From those tests categorized as USPSTF grade D, the intervention group prescribed an average of 9.8 vs. 11.8 tests per 100 consultations in the control group (p = 0.003).
Conclusions: Removing unnecessary tests from a quick shortcut menu of the diagnosis and laboratory tests ordering system had a significant impact and reduced unnecessary prescription of tests.
The fact that it was not possible to perform the randomization at the family physicians’ level, but only on the computer servers is a limitation of our study. Future research should assess the impact of different test ordering systems during longer periods.
Trial registration: ISRCTN45427977, May 1st 2014 (retrospectively registered).
Keywords: Preventive health services, primary health care, evidence-based practice, decision support systems, clinical decision making, computer-assisted
Background
Informatics has undoubtedly changed the way societies live, socialize, learn, work, and deal with healthcare. We now live in a period of increasing concern about the excessive presence of medicine in our lives.[1][2][3] When inefficient software is combined with a non-evidence-based medical practice, there is the risk of patient harm, significant impact to quality of life, and damage to the healthcare system due to unnecessary costs.
The implementation of electronic health records (EHRs) has both potential benefits and drawbacks.[4] Among the benefits, the prevention of medical errors and the promotion of patient safety has often been mentioned and confirmed in clinical practice.[4][5][6] Despite the positive effects of EHR implementation in clinical practice, a range of barriers faced by physicians has been identified. These barriers may include technical and financial aspects, time, psychological, social, legal, and organizational changes to the process.[7] After having removed the first barriers to EHR implementation, it is now time to implement continuing improvement and development of the available tools and to incorporate the scientific evidence obtained to this point.[4][8][9][10]
To achieve better patient safety standards and improve healthcare system cost-effectiveness, there has been a worldwide effort to implement an integrated EHR system with diagnostic and laboratory test ordering communication systems.[11][12][13] There have also been attempts to incorporate clinical decision support systems to further improve the quality of medicine. Prescribing diagnostic and laboratory tests is a key component of medical consultation. In the primary health care setting, tests are often ordered with preventive intentions and fulfillment of patient expectations.[14][15] There is also great uncertainty and variability among family physicians’ ordering routines.[16][17][18] The effects of test ordering communication systems integrated with clinical decision support systems have been reported in various clinical practice settings. Main et al. have performed a systematic review of this topic and reported that integration of clinical decision support systems resulted in significant benefits to the prescribing process and practitioner performance outcomes in nearly two-thirds of the 24 studies that met the inclusion criteria.[19]
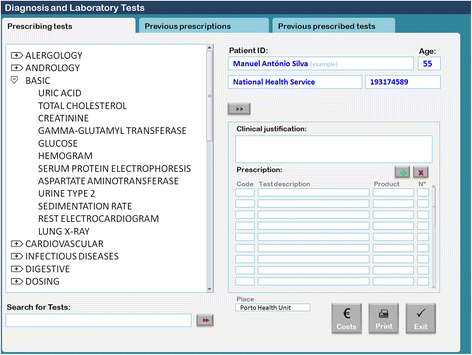
In Portugal, the use of EHR software with a diagnostic and laboratory test order communication system has been mandatory since September 2011. Most of the primary healthcare centers use software called Sistema de Apoio ao Médico (Physician’s Support System [SAM]). In the module used to order diagnostic and laboratory tests, physicians access a searchable test menu by two possible strategies: 1) typing the test name in a search box or 2) browsing by a shortcut menu structure (Fig. 1). Different menus are available for most areas of medicine, including basic, allergology, andrology, cardiovascular, central nervous system, digestive, dosing, endocrinology, gynecology, hematology, infectious diseases, nephrology, obstetrics, oncology, otorhinolaryngology, osteoarticular, preoperative, respiratory, rheumatology, and urology. Under each menu there is a set of specific lab tests. Physicians can choose one or more tests by double-clicking each test or can choose the entire set by double-clicking on the shortcut menu’s title. For example, the basic menu is composed of uric acid, total cholesterol, creatinine, gamma-glutamyl transferase, glucose, hemogram, serum protein electrophoresis, aspartate aminotransferase, urine type 2, sedimentation rate, electrocardiogram, and lung x-ray tests.
|
If a physician would like to choose only hemogram and glucose, they must double-click on each test. However, if they would like the entire basic set of tests, they must double-click on the basic menu title. Our research team suspected that this basic shortcut menu is often selected during routine consultations in which patients ask for routine check-ups without specific reason. As we have shown in a previous study, there is a high prevalence of Portuguese adults (99.2%) that believe they should have routine blood and urine tests annually.[14] This statistic demonstrates the importance of examining the effectiveness and efficiency of this basic sub-menu.
Through a randomized controlled trial, the primary aim of the present study was to compare the effects of modifying the EHR ordering communication system (modified SAM) by changing the basic shortcut menu and adding a clinical decision support system based on the integration of the United States Preventive Services Task Force (USPSTF) recommendations. After the last primary healthcare reform in Portugal, primary healthcare centers have been divided into healthcare center groups.[20] In a healthcare center group, the informatics network is linked through servers that may serve more than one healthcare center. Creating a modified version of the SAM software requires it to be installed at a server level, which determines that all physicians at all healthcare centers served by that server will receive the same version of the software. For this reason, it was not possible to randomize the study at the physician level. Rather, we had to randomize the servers at the healthcare center group level.
Methods
Trial design
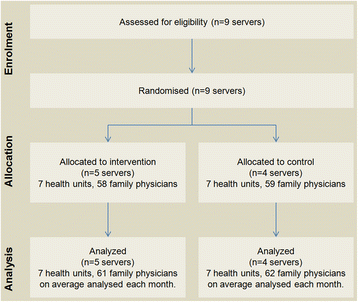
All servers of the Western Oporto grouping of health centers counted for randomization, except that serving the center where study authors worked (to avoid possible bias). The remaining nine servers were randomized into two groups: 1) five servers were randomly allocated to the intervention group and 2) four servers to the control group.
Participants
All family physicians working and prescribing diagnostic and laboratory tests in the Western Oporto group of health centers (except in those where the authors worked) participated in this study.
Data of the diagnostic and laboratory tests prescriptions were centrally collected by informatics staff belonging to the Ministry of Health and sent to the research team without patients’ or physicians’ identifications.
Interventions
The control group continued to use the standard version of the EHR software (SAM) as presented in Fig. 1. The intervention group used a modified version of the software (SAM modified) installed in each server (Fig. 2). Software modifications consisted of two principal changes:
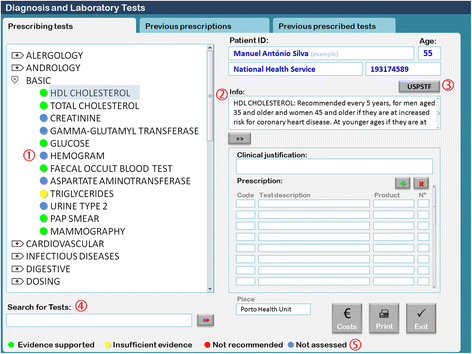
1. Basic shortcut menu changes: The composition of the basic menu set of diagnostic laboratory tests was changed. Some tests were removed, including uric acid, serum protein electrophoresis, sedimentation rate, electrocardiogram, and lung x-ray. Other tests were added, including HDL cholesterol, fecal occult blood test, triglycerides, Pap smear, and mammography. Although some tests were removed from the basic menu, physicians were still able to request them by typing their names in the search for tests box.
2. Addition of an evidence-based decision support: For the tests listed in Table 1, we added traffic light-based colored dots according to the USPSTF recommendations and an additional information box containing the summary of the USPSTF recommendation and a link to the integral recommendation at the USPSTF website (Fig. 2).[21]
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The selection of the specific tests to be removed from or added to the basic shortcut menu was made after discussion and debate with eight family physicians selected by proximity and convenience. The main rational was to remove tests that are not recommended to be used as routine screening tools and to substitute them with others that may be recommended for certain risk groups. However, creating a change that was not too disruptive to the current family physicians’ practices was a concern, and that is why tests such as creatinine, gamma-glutamyl transferase, hemogram, aspartate aminotransferase, and urine type 2 were left in the basic shortcut menu.
EHR software modifications were implemented on May 30–31, 2012 in all of the intervention groups’ servers. Prospective monthly monitoring and data collection occurred until January 31, 2013. To allow a pre-post analysis in both groups, a retrospective monthly data collection of both control and intervention groups was also performed between December 1, 2011 and May 31, 2012.
The collected data included monthly parameters, including numbers of prescribing family physicians, face-to-face consultations made, and number of each prescribed diagnostic and laboratory test.
Independent variables
Group (control and intervention) and time (before and after the modifications) were the independent variables.
Dependent variables
Primary outcomes were chosen to assess the impact of our intervention on the number of diagnostic and laboratory tests prescribed by physicians using four different perspectives: 1) impact on the number of the prescriptions of diagnostic and laboratory tests that were withdrawn from the basic menu; 2) impact on the number of the prescriptions of diagnostic and laboratory tests that were added to the basic menu; 3) impact on the number of the prescriptions of diagnostic and laboratory tests that were marked with green dots (USPSTF recommendation grades A and B); and 4) impact on the number of prescriptions of diagnostic and laboratory tests that were marked with red dots (USPSTF recommendation grade D). Diagnostic and laboratory tests were the dependent variables.
Sample size
Given administrative and technical barriers related to the permission process for the introduction of EHR software modifications, our sample size was obtained by convenience. We decided to run this trial at the Western Oporto grouping of healthcare centers, which includes a total of 15 primary care healthcare centers and an informatics network of 10 servers. One of the servers, which served the healthcare center in which some of the study authors worked, was excluded from the study.
The power calculation was done for the mean number of monthly prescribed tests per 100 consultations comparison between control and intervention groups. A significance level of 0.05, a power of 0.8, and a standard deviation of two tests prescribed per 100 consultations per month was considered. A mean difference of 3.3 tests prescribed per 100 consultations per month between control and intervention groups was used.
Randomization
The remaining nine servers were sequentially numerated and randomly assigned to the intervention and control group by an investigator blinded to the server identification. The allocation sequence was computer generated resulting in five servers (seven healthcare centers, 58 family physicians) allocated to the intervention group, and four servers (seven healthcare centers, 59 family physicians) allocated to the control group.
To guarantee allocation anonymity, family physicians at each healthcare center only received information about this trial implementation after randomization has been performed. Given the nature of this trial, no consent at physician level was obtained. Consent was obtained from the Northern Regional Health Administration and the Executive Council of the Western Oporto Group of Health Centers.
Statistical methods
To examine whether the software modification changed test prescription trends we performed an interrupted time series analysis with an autoregressive integrated moving average (ARIMA) model, using the monthly number of tests prescribed per 100 consultations and the intervention (the software modification) as a dichotomous variable (before and after intervention). In the control group, we also performed an ARIMA model analysis using the monthly number of tests prescribed per 100 consultations and the same dichotomous variable (before and after intervention), although there was no intervention in this group. The before and after intervention comparison of the monthly average number of diagnostic and lab tests prescribed per 100 consultations between control and intervention groups was made using an independent sample t test. The Bonferroni correction was used for adjusting for multiple testing (several diagnostic and laboratory tests).[22] A significance level of 0.05 was used. As size effect measure, Cohen’s d was calculated and a d of ≥0.8 was considered a large effect.
Ethical considerations
This study was approved by the Northern Regional Health Administration Medical Ethics Committee with several considerations: “1) Given that the study will collect only anonymous data there is no need for the obtainment of informed consent by physicians; 2) The study is of great relevance and with an expected practical interest of the results; and 3) The methodology used safeguards the rights of participants.”
In accordance with the Medical Ethics Committee, before the implementation of the study (but after the randomized allocation), a letter was sent to all Western Oporto Group of healthcare centers informing them of and explaining the study aims and methodology.
Results
The number of servers enrolled was constant without loss (Fig. 3). The number of family physicians after the intervention ranged from 58 to 64 in the intervention group and from 55 to 70 in the control group. Recruitment was performed in April 2012, and the trial began on June 1, 2012, with a follow-up period until January 31, 2013 (eight month follow-up). Baseline data are presented in Table 2.
|
| ||||||||||||||||||||
For both groups, Table 3 compares the monthly average number of diagnostic and laboratory tests prescribed for each 100 consultations before and after EHR software modification, under a time series analysis perspective (ARIMA model). Within the five tests that were withdrawn from the basic menu, we observe a statistically significant reduction in the prescription of four tests (uric acid, serum protein electrophoresis, sedimentation rate, and lung x-ray tests) in the intervention group after software modification. Within the five tests that were added to the basic menu, we observed a significant increase in prescription of the fecal occult blood test in the intervention group after software modification. Within the tests that were marked with green dots (USPSTF recommendation grades A and B) there were no significant increases in either the control or intervention groups. Within the tests that were marked with red dots (USPSTF recommendation grade D), there was a significant reduction only in the prescription of cancer antigen 19-9, but the amount of this test that was prescribed was so low that this variation may be justified by other factors and not a result of software modification.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4 presents a direct comparison between the control and intervention groups before and after software modifications (excluding the first month following modification that was considered as a washout period). Regarding the set of tests that were withdrawn from the basic menu, we observe that there were no significant differences between either group before software modification, but that there was a significantly lower prescription rate in the intervention group for the following tests after software modification: 1) uric acid; 2) serum protein electrophoresis; and 3) sedimentation rate. When we consider all tests belonging to the set withdrawn from the basic menu, we have verified that after software modification the intervention group prescribed less than half of those tests when compared with the control group (14.01 tests per 100 consultations versus 29.29 tests per 100 consultations, p < 0.001). Regarding the five tests that were added to the basic menu, we observed no significant differences after software modification between the two groups. Considering the tests that were marked with green dots (USPSTF recommendation grades A and B) we observed no significantly higher prescription rates in the intervention group after the software modification compared to the control group. For the tests that were marked with red dots (USPSTF recommendation grade D), we observed significant lower prescription rates in five tests (cancer antigen 19-9, exercise electrocardiography, carotid artery ultrasound, hepatitis B surface antigen, and hepatitis C antibodies) in the intervention group after software modification.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
Our results show that removing unnecessary tests from a quick shortcut menu of diagnostic and laboratory tests available on the EHR test ordering system might have significantly impacted ordering habits and reduced unnecessary test prescriptions. This result emphasized the importance of careful attention and scientific rigor when building shortcut menus in ordering systems. This impact was significant, and it could be observed from two perspectives of analysis: 1) when we compared the results for the intervention group before and after the modification of the software and 2) when we compared the intervention group with the control group.
Furthermore, our results demonstrated that the introduction of an evidence-based decision support tool had no significant impact on the test prescription profiles. However, it should be noted that as a passive instrument, the decision for support addition did not interfere with the normal flow of the ordering system. The doctors had only visual contact with the colored recommendations and optional access to the additional information from the USPSTF recommendations. The absence of impact of this support decision tool may also be related to a high level of agreement between the way Portuguese family doctors perform preventive health services and the USPSTF recommendations as shown in a previous national cross-sectional study.[22]
Limitations of our study include the sampling method and sample size. The moderate size of the sample might have limited our conclusions regarding the less frequently prescribed tests, but this is unlikely to have affected the main conclusion of our study.
The fact that it was not possible to perform randomization at the family physician level, but only at that of the computer servers, also constitutes a limitation and a possible biasing factor, since the number of servers in the Western Oporto Grouping of healthcare centers was small. In the Western Oporto Grouping of healthcare centers we had nine servers. Since our intervention was a software modification, we had to randomize at the level of the electronic server. One hypothesis would be used to consider each server as a cluster, but the problem is that some servers serve only one health unit, not being a real cluster, while others serve two or three healthcare units. The ideal would be to consider each healthcare unit as a cluster of family physicians. For that, however, we had to have access to the number of tests prescribed by each family physician, which we did not. The Ministry of Health only allowed us access to the tests prescribed by each healthcare unit. This is the reason why it is not possible to consider this study as a cluster randomized controlled trial. Without the number of tests prescribed by each family physician, we were not able to calculate the intracluster correlation coefficient.
Another limitation of our study is that the time available for its completion was relatively short. An extended study period could have allowed us to draw more extensive conclusions (for example, regarding the tests that are prescribed less frequently). A prolonged study period would also have diluted possible seasonal effects that might exist in seeking medical consultations and also minimized the impact of factors inherent in the economic situation of Portugal during the study period. In the context of the economic crisis, there was an increase in user fees paid by patients for medical consultations and laboratory tests in addition to Ministry of Health publications of clinical guidelines for prescriptions of some diagnostic and laboratory tests. These facts may have contributed to the gradual decrease in test prescriptions that we have observed during the study period both in the control and intervention groups. The extension of the study might also have diluted this effect and could have allowed a clearer reading of the impact of software modifications. However, the socio-economic status of intervention and control medical centers was identical. All healthcare centers involved in the study are located in the urban area of Oporto in a geographic area of approximately 22Km2.[23] All study physicians were exposed to the same Ministry of Health guidelines and general education materials.
These limitations were mainly due to technical issues related to the fact that our research could not disrupt the normal functioning of resources and services of the Ministry of Health. However, we consider it unlikely that any of these limitations limited the main conclusion of this study: the considerable impact of unnecessary test removal in the shortcut menu of the ordering system.
Considering the potential generalizability of our results to other groups of healthcare centers of the Portuguese National Health Service, we believe that there is a high probability that the same effect will be found with similar software intervention. We believe that this is the case for two reasons: 1) the size of the obtained effect was evident and 2) the main features of the ordering system of other groups of healthcare centers are similar.
For this study, we used the recommendations of the USPSTF as our standard guidelines because it bases its recommendations on freely accessible, evidence-based systematic reviews with recognized methodological quality that covers a considerable range of topics. Portugal is in a phase of transition regarding clinical guidelines and recommendations. The Ministry of Health has recently published guidelines covering some topics related with test prescriptions. However, many of these guidelines are still in the public discussion phase and are not based on systematic reviews but rather on experts' opinion. For these reasons we chose the USPSTF recommendations. Exceptions to this are the recommendations of the Portuguese Ministry of Health for three tests: 1) breast cancer screening by mammography every two years for women 50–69 years old; 2) colorectal cancer screening by a fecal occult blood test every one to two years for adults 50–74 years old; and 3) cervical cancer screening with a cervicovaginal cytology for women between 25 and 60 years every three years after two annual normal tests.[24] These recommendations are in line with those from USPSTF but could have influenced our results.
When designing this trial, the researchers also considered the possibility of causing some negative effects as a result of the intervention. There could be two main type of negative effects: 1) on physicians because they could not find the tests in the basic shortcut menu as they previously had, and it would consume more consultation time to type the name in the search box to order one of the removed tests and 2) on patients because they could be deprived of some necessary tests. After debating this issue with some family physicians, these potential negative effects were considered negligible, mainly because there was no interference with physicians' freedom to prescribe any medical test, and the use of the search box as an alternative to the shortcut menu was not considered a major obstacle.
The reduction of unnecessary test prescriptions that we verified was in line with other studies in which changes in laboratory request forms also resulted in the reduction of unnecessary tests requests.[25][26][27] However, there are some practical implications that may result from this study. For example, the Portuguese Health Authorities could use these results to improve test ordering software and to reduce the prescription of unnecessary tests at a national level. This study also reinforces the importance of adequately testing medical related software tools, and we hope that in the future this will be the rule for other medical software tools.
Conclusions
Our results might have a significant impact on improving the design of shortcut menus of diagnostic and laboratory test ordering systems either in the Portuguese National Health Service or in other countries’ health systems. These improvements can help reduce the prescription of unnecessary tests, leading to the reduction of negative patient effects and to the reduction of unnecessary costs. These study results demonstrate the importance of testing and evaluating various aspects of medical informatics programs to improve efficiency and contribute to improved clinical practice and clinical outcomes.
Abbreviations
ARIMA: Autoregressive integrated moving average
EHR: Electronic health records
SAM: Sistema de Apoio ao Médico (Physician’s Support System)
USPSTF: United States Preventive Services Task Force
Declarations
Acknowledgements
Not applicable.
Funding
We have received financial support of 15,000 Eur from the Astrazeneca Portugal Foundation’s 2010 program for support of research. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The co-author Andreia Teixeira thanks the financial support by FCT (SFRH/BPD/86383/2012).
Availability of data and materials
The data are not available because the ethics committee only provided authorization for the research team to perform the raw data analysis.
Authors' contributions
CMSM had the original idea of the study, designed the trial and made the quality control during the data collection, drafted and revised the paper. LFRA contributted to the trial design and revised the paper. ASCT and CMNCS planned, supervised and has performed the statistical data analysis, contributed to the paper draft and revised the paper. LMBS, PAAPS, MLGDC, AMRCP and AAOPH revised the paper. All the authors read and approved the final version of the manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
This study was approved by the Northern Regional Health Administration Medical Ethics Committee that considered: a) Given that the study will collect only anonymous data there is no need for the obtainment of informed consent by physicians; b) The study is of great relevance and with an expected practical interest of the results; and c) the methodology used safeguards the rights of participants.
In accordance with the Medical Ethics Committee, before the implementation of the study (but after the randomized allocation), a letter was sent to all Western Oporto Group of health centers informing and explaining the aims and methodology of the study.
References
- ↑ Glasziou, P.; Moynihan, R.; Richards, T. et al. (2013). "Too much medicine; too little care". BMJ 347: f4247. doi:10.1136/bmj.f4247. PMID 23820022.
- ↑ Moynihan, R.; Doust, J.; Henry, D. (2012). "Preventing overdiagnosis: How to stop harming the healthy". BMJ 344: e3502. doi:10.1136/bmj.e3502. PMID 22645185.
- ↑ Getz, L.; Sigurdsson, J.A.; Hetlevik, I. (2003). "Is opportunistic disease prevention in the consultation ethically justifiable?". BMJ 327 (7413): 498–500. doi:10.1136/bmj.327.7413.498. PMC PMC188390. PMID 12946974. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC188390.
- ↑ 4.0 4.1 4.2 Raposo, V.L. (2015). "Electronic health records: Is it a risk worth taking in healthcare delivery?". GMS Health Technology Assessment 11: Doc02. doi:10.3205/hta000123. PMC PMC4677576. PMID 26693253. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4677576.
- ↑ Ben-Assuli, O. (2015). "Electronic health records, adoption, quality of care, legal and privacy issues and their implementation in emergency departments". Health Policy 119 (3): 287–97. doi:10.1016/j.healthpol.2014.11.014. PMID 25483873.
- ↑ Ben-Assuli, O.; Leshno, M. (2016). "Assessing electronic health record systems in emergency departments: Using a decision analytic Bayesian model". Health Informatics Journal 22 (3): 712–29. doi:10.1177/1460458215584203. PMID 26033468.
- ↑ Boonstra, A.; Broekhuis, M. (2010). "Barriers to the acceptance of electronic medical records by physicians from systematic review to taxonomy and interventions". BMC Health Services Research 10: 231. doi:10.1186/1472-6963-10-231. PMC PMC2924334. PMID 20691097. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2924334.
- ↑ Roukema, J.; Los, R.K.; Bleeker, S.E. (2006). "Paper versus computer: Feasibility of an electronic medical record in general pediatrics". Pediatrics 117 (1): 15–21. doi:10.1542/peds.2004-2741. PMID 16396855.
- ↑ Bowman, S. (2013). "Impact of electronic health record systems on information integrity: Quality and safety implications". Perspectives in Health Information Management 10: 1c. PMC PMC3797550. PMID 24159271. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3797550.
- ↑ Nguyen, L.; Bellucci, E.; Nguyen, L.T. (2014). "Electronic health records implementation: An evaluation of information system impact and contingency factors". International Journal of Medical Informatics 83 (11): 779–96. doi:10.1016/j.ijmedinf.2014.06.011. PMID 25085286.
- ↑ Moen, A.; Hackl, W.O.; Hofdijk, J. et al. (2013). "eHealth in Europe - Status and challenges". Yearbook of Medical Informatics 8: 59–63. PMID 23974549.
- ↑ Olsson, S.; Lymberis, A.; Whitehouse, D. (2004). "European Commission activities in eHealth". International Journal of Circumpolar Health 63 (4): 310-6. PMID 15709306.
- ↑ Iakovidis, I.; Purcarea, O. (2008). "eHealth in Europe: From vision to reality". Studies in Health Technology and Informatics 134: 163–8. PMID 18376043.
- ↑ 14.0 14.1 Martins, C.; Azevedo, L.F.; Ribeiro, O. (2013). "A population-based nationwide cross-sectional study on preventive health services utilization in Portugal - What services (and frequencies) are deemed necessary by patients?". PLOS One 8 (11): e81256. doi:10.1371/journal.pone.0081256. PMC PMC3836775. PMID 24278405. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3836775.
- ↑ van Bokhoven, M.A.; Pleunis-van Empel, M.C.; Koch, H. et al. (2006). "Why do patients want to have their blood tested? A qualitative study of patient expectations in general practice". BMC Family Practice 7: 75. doi:10.1186/1471-2296-7-75. PMC PMC1769380. PMID 17166263. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1769380.
- ↑ van der Weijden, T.; van Bokhoven, M.A.; Dinant, G.J. et al. (2002). "Understanding laboratory testing in diagnostic uncertainty: A qualitative study in general practice". British Journal of General Practice 52 (485): 974–80. PMC PMC1314466. PMID 12528582. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1314466.
- ↑ van Bokhoven, M.A.; Koch, H.; Dinant, G.J. et al. (2008). "Exploring the black box of change in improving test-ordering routines". Family Practice 25 (3): 139-45. doi:10.1093/fampra/cmn022. PMID 18535302.
- ↑ Verstappen, W.H.; ter Riet, G.; Dubois, W.I. et al. (2004). "Variation in test ordering behaviour of GPs: Professional or context-related factors?". Family Practice 21 (4): 387-95. doi:10.1093/fampra/cmh408. PMID 15249527.
- ↑ Main, C.; Moxham, T.; Wyatt, J.C. et al. (2010). "Computerised decision support systems in order communication for diagnostic, screening or monitoring test ordering: Systematic reviews of the effects and cost-effectiveness of systems". Health Technology Assessment 14 (48): 1–227. doi:10.3310/hta14480. PMID 21034668.
- ↑ da Costa Pereira, A.; Giest, S.; Dumortier, J.; Artmann, J. (October 2010). "eHealth Strategies - Country Brief: Portugal" (PDF). European Commission. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.476.4177&rep=rep1&type=pdf.
- ↑ U.S. Preventive Services Task Force (2014). "The Guide to Clinical Preventive Services 2014". https://www.overdrive.com/media/2062549/clinical-preventive-services-2014. Retrieved 08 October 2016.
- ↑ 22.0 22.1 Martins, C.; Azevedo, L.F.; Santos, C. (2014). "Preventive health services implemented by family physicians in Portugal - A cross-sectional study based on two clinical scenarios". BMJ Open 4 (5): e005162. doi:110.1136/bmjopen-2014-005162. PMC PMC4039810. PMID 24861550. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4039810.
- ↑ Administração Regional de Saúde do Norte, IP (2010), "Action plan 2010-2012 of the Western Oporto grouping of health centers", {{{website{{{}}}}}}
- ↑ da Saúde, M. (2007), "National Plan for Prevention and Control of Oncological Diseases 2007/2010", {{{website{{{}}}}}}
- ↑ Zaat, J.O.; van Eijk, J.T.; Bonte, H.A. (1992). "Laboratory test form design influences test ordering by general practitioners in The Netherlands". Medical Care 30 (3): 189–98. PMID 1538607.
- ↑ Barth, J.H.; Balen, A.H.; Jennings, A. (2001). "Appropriate design of biochemistry request cards can promote the use of protocols and reduce unnecessary investigations". Annals of Clinical Biochemistry 38 (Pt 6): 714–6. doi:10.1258/0004563011900957. PMID 11732657.
- ↑ Bailey, J.; Jennings, A.; Parapia, L. (2005). "Change of pathology request forms can reduce unwanted requests and tests". Journal of Clinical Pathology 58 (8): 853–5. doi:10.1136/jcp.2004.023101. PMC PMC1770878. PMID 16049288. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1770878.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. In some cases important information was missing from the references, and that information was added. Grammar edits were made to make the document more readable. The numerical data in the last quarter of Table 4 changed to two decimal places for no clear reason; however, it was retained as-is.