LII:Justifying LIMS Acquisition and Deployment within Your Organization/Introduction to LIMS and its acquisition and deployment
1. Introduction to LIMS and its acquisition and deployment
Your laboratory was created to carry out specific tasks that its parent organization needed to meet its business goals. The lab's biggest output is analytical results of tests it conducts, and it's imperative those analytical results are accurate, timely, and unaltered. Today, labs more than ever depend on a laboratory information management system (LIMS) to better meet those requirements. But justifying the benefits over the costs can be a challenge, particularly when upper management or critical stakeholders aren't keen on increasing spend, let alone aren't always well-informed about what LIMS can actually do for the lab.
The point of this guide is to give you the background you need to better make a case to your organization's management and key stakeholders for acquiring and deploying a LIMS. While pursuing this topic, we're going to make a couple of simple assumptions. We'll assume that your organization's senior management may not understand what goes on in your lab and how it carries out its work. We'll also assume they have a lot of things to contend with in their roles, giving them limited time to fully understand your lab's needs and the tools required to help it succeed. You want to make the explanation and justification for LIMS acquisition and deployment as easy and succinct as possible while at the same time demonstrating that this is the right decision.
The justification process for LIMS has a long history, and a lot has changed since the 1980s, when the technology was first introduced. Therefore, we need to examine what a LIMS is; what alternatives people commonly turn to; how the technology has evolved; what the history of LIMS justification is; what organizational, economic, and practical considerations surround LIMS adoption; how to develop a LIMS project proposal; and how to address any concerns management may raise during the justification process. This guide will attempt to address these things and more. First, however, we need to look at what LIMS is, what alternatives exist, and what the acquisition process looks like then and now. We'll also begin to address why a LIMS matters to today's laboratory in this chapter, with further points of justification coming later.
1.1 What is a laboratory information management system (LIMS)?
A LIMS is a specialized, software-based information management solution that offers a set of key features that support a modern laboratory's operations. Introduced in the late 1970s and early 1980s[1], the design and functionality of this software has changed significantly over the years, such that today a majority of a laboratory's processes can benefit from the LIMS' implementation and use. A LIMS has functionality that addresses most of the workflow phases of a laboratory, including[2]:
- the reception and log in of a sample and its associated customer data;
- the assignment, scheduling, and tracking of the sample and the associated analytical workload;
- the processing and quality control (QC) associated with the sample and the utilized equipment and inventory;
- the storage and access management of data associated with the sample analysis; and
- the inspection, approval, and compilation of the sample data for reporting and/or further analysis.
Related is the laboratory information system (LIS), which is tailored to the workflow of a clinical laboratory. That said, the distinction between the LIMS and LIS has blurred over time, with some LIMS having the same clinical information management features of an LIS. In both cases, the software—when developed, implemented, and maintained well—can improve laboratory workflows and workloads while enhancing safety, quality, and compliance. A well-implemented LIMS can reduce the silos of information and data in a laboratory, while at the same time make that information and data more secure, readily accessible, and actionable. Additionally, many modern LIMS are able to be pre-configured out of the box with analytical and QC workflow support tools that can be further optimized to a lab's unique industry-based workflow. When integrated with instruments and other software systems, fewer transcription errors occur and traceability becomes easier, while monitoring of supply chain issues, QC data, instrument use, and more is further enabled, particularly when paired with configurable dashboards and alert mechanisms. This also means that the lab can react more rapidly to issues that compromise compliance with certification to the ISO/IEC 17025 standard, as is necessary in, for example, the food and beverage industry.[3][4][5]
1.2 What are the alternatives to a LIMS?
Introducing new technologies and products often causes people to balk because they represent a change to current operations and an expense, even if the change is beneficial. As such, some may view the acquisition, deployment, use, and maintenance of a LIMS to be too daunting. The organization may even see the value in a LIMS yet ask several questions in regards to its necessity:
- What happens if we don’t make the change to a LIMS?
- Is there an alternative technology that is less costly?
The answer to the first question is straightforward: lab costs increase, operations stagnate, sample backlogs increase, and lab personnel—including management—become increasingly frustrated. This in turn means the lab may fail to achieve its goals. That second question is politically charged, however, particularly in medium to larger organizations. There are two frequently encountered answers, both involving internal software development: let's use a spreadsheet, or let's use an enterprise resource planning (ERP) system, with similar characteristics to a LIMS, as a solution. The latter often occurs if a company has recently invested in an ERP solution with the idea that it will take care of all of the company’s needs (i.e., they may not have checked with its labs to see if actually will).
Before we get into a response to that second question, here's a question for you. What business is your company in? Is it a software development organization, or does it want to become one? This is an important question because someone in the organization inevitably proposes custom software development as an alternative. Suppose you are seriously considering developing a LIMS alternative in-house or through a consulting firm. In that case, that is something you have to think through, taking on all the issues that plague large software development projects, including ongoing maintenance and support once the project is completed.
Software development projects are fraught with issues. Reasons for software project failure include[6][7][8]:
- insufficient organizational leadership;
- insufficient understanding of business and department problems and requirements, even as they evolve during development;
- inadequate planning and project management;
- undefined project roles and responsibilities;
- inaccurate time and cost projections;
- inadequate use of available resources;
- insufficient understanding of changing software development practices;
- poor or mismanaged communication methods;
- poor response to project challenges that inevitably arise;
- lack of focus on project success;
- lack of focus on soft skills such as training and team building; and
- scope creep.
The bottom line is this: software projects develop issues, and while they may eventually succeed, they may take a year or more to work through during development. Meanwhile, your lab is suffering under whatever issues caused you to look for a LIMS in the first place. When you're done with developing the solution, will you have something better than commercial products or just the best you can settle for? Commercial products will be continually improved with new features and capabilities added; that's their business. Do you have the resources and commitment to make it yours?
We find that software development approaches have their own issues to address, but what about spreadsheets and ERP systems? Spreadsheet-based systems, while seemingly inexpensive, are costly in development time and use. Additionally, spreadsheets are single-user-at-a-time systems. Everyone else has to wait their turn if someone is working with the system. They are fraught with errors creeping in, especially through changes that may not be documented. They are also difficult to validate and keep under modification control. Those factors are red flags to regulatory inspectors, particularly regarding development documentation.[9] As for ERPs, those systems may address some of the requirements of your lab, but they will have significant problems with instrument connections, a significant source of productivity gains in the laboratory. Do these and similar alternatives represent the most cost-effective way to manage risk and utilize your resources towards the goal of improving laboratory productivity and compliance? The purpose-built LIMS may make more sense given the challenges posed by forcibly shoehorning these alternatives into a role they weren't necessarily designed for.
1.3 LIMS acquisition then
The laboratory environment of the 1980s, viewed from a behavioral standpoint, would be easily recognizable today: growing test workloads, everyone’s sample was a top priority, and everyone was very busy as all work was manually done. From a technology perspective, things were a lot different. For the most part, there were no computers, and instrument data systems (IDSs) were entering the marketplace. Still, IDSs hadn't become the almost automatic combined instrument-computer combination they are today. Networks were in their early stage of development. Robotics was also in an earlier part of its evolution; Zymark Corporation of Hokinton, Massachusetts (which later became Caliper Life Sciences), was an early developer of lab robotics, which was just beginning to impact the laboratory market. Additionally, there was no severe shortage of educated laboratory personnel to fill open lab positions. However, few were knowledgeable in laboratory informatics.
The cost of lab operations was becoming a serious concern for lab management. Although people were available, there was a limit to how many you could hire, given the limitations of laboratory space and equipment utilization. As a result, economics played a significant role in the justification for equipment and instrumentation, in general, and LIMS, a new technology, in particular. One of the significant differences between then and today is that installing a LIMS meant installing a local computer system; cloud-based systems didn't exist.
As the productivity issue became more acute, the interest in automation and the efficiency of lab operations grew. In clinical chemistry work, the response was particularly strong with the development of an industry-wide total laboratory automation (TLA) initiative that focused on driving down the cost of laboratory testing through automation and the development of communications standards to make it easier to connect instruments and computers together into fully functioning networks, which would relieve much of the manual clerical effort.
Much of the information we have on the effectiveness of laboratory automation and LIMS installations comes from the life science and clinical industries. The results of TLA could be seen in 1992, when Markin and Hald noted[10]:
The tangible [financial] benefits which were measured prior to LIS justification totaled $153,471.84 per year based upon fiscal year 1985-86 dollars. The objective re-evaluation of these activities four years after implementation and operation resulted in a measured $115,326.93 per year savings ... Implementation of the laboratory information system at our institution resulted in a 75% capture of the estimated/projected cost avoidance or cost savings.
In a more modern example from 2018, Yu et al., who wishing to improve workflow efficiency in their clinical laboratory, implemented TLA "that connected our pre-analytic processing system with various testing (hematology, coagulation, and chemistry)."[11] They reported their results as such[11]:
The implementation of our TLA system resulted in 86% fewer discrete processing steps in specimen handling, even when starting from a partially automated laboratory. Instrument consolidation reduced the testing footprint by 45% and reduced the number of testing personnel by 2.5 full-time employees (FTEs). An 82% reduction in hands-on time associated with add-on processes was achieved. Combining STAT and outreach work on the testing system did not impact turnaround time.
Outside of TLA, lacking other driving factors, the primary motivation for implementing automation and LIMS or LIS in the lab early on was productivity and, as a result, finances. A 1986 article by Joseph H. Golden (then at Laboratory Management Systems, Inc.), titled “Economic Considerations of Laboratory Information Management Systems,” addressed that point directly. Considerations and concerns he had included (quoting directly)[12]:
- the "impact on lab operations, reducing administrative work [and] speeding up work";
- "how the laboratory contributes to the corporate bottom line";
- the "need to consider not just what LIMS will do but what it’s worth";
- in regards to R&D, that "productivity improvements amount to at least 10% to 20% of the total staff resources of a laboratory complex”;
- in regards to QA/QC, that "time-value of information" and "ability to accept/reject raw material and avoid off-spec products" is considered, "plus 10-20% saving on clerical costs";
- that commercial testing saw "improved profitability";
- the consideration of "common lab management problems," such as "increasing data volume from increased use of smart instruments and from increased testing and record retention requirements imposed by the Environmental Protection Agency (EPA), Food and Drug Administration (FDA), Occupational Safety and Health Administration (OSHA), and other regulatory agencies; constantly rising operating and material costs; and ever-tightening constraints on staff and material expenditures, which in turn are "manifested by increasingly burdensome paperwork, inefficient utilization of resources, and exasperating searches for misplaced samples and data"; and
- the costs of "equipment, facilities, cost of money, and investment tax credits."
The justification for LIMS acquisition in the 1980s was based on comparing two states: the status quo vs. the introduction of laboratory informatics in the form of an on-premises LIMS. Among the more significant management concerns were whether the costs could be contained and the project kept on track. Large software projects were notorious for cost overruns and scheduling delays during that time. However, demand for improved processes in labs was also increasing, particularly in regulated settings like the pharmaceutical industry. Failure to keep accurate records, and be able to find them, could result in plant closures, and that alone could justify the acquisition for a LIMS.
Table 1 compares concessions and considerations made in the 1980s of maintaining existing lab operations vs. acquiring and implementing an on-premises LIMS.
| ||||||
1.4 LIMS acquisition now
Today, laboratories of all kinds face a variety of factors that drive them to become better at managing their operations. The tools for this improvement are available, and regulatory agencies expect labs to use the best technologies to meet regulatory requirements. Although it has been technically possible to meet regulatory requirements without computing technology—using paper-based systems—electronic systems make the work much easier and less costly, while improving the lab’s ability to meet inspectors' requests promptly, as well as the lab's workflows themselves. In the pre-electronic era, paper-based approaches to conducting lab operations were the only option, and these approaches tended to be slow, inefficient, and expensive. This changed in the late 1970s and into the 1980s, as computerized systems held promise in speeding up lab processes and making them more efficient, though they were still expensive to implement. Today, laboratory informatics options are less expensive, while also living up to the promise of improving lab operations. Laboratories produce data and information as their primary product; the ability to put those products to work and gain value from them is paramount. Continuing to use a paper-based system places limitations on that ability while incurring additional hidden and apparent costs to the lab. A LIMS and other informatics solutions make more sense than ever, particularly given stronger economic and competitive pressures.
Regulatory pressures have also increased as the regulatory landscape has changed. During the mid-1980s, when Mr. Golden's analysis was presented, regulatory requirements were beginning to gain new ground. The EPA and FDA had records retention requirements, and companies were starting to understand the FDA's guidelines, first published in 1978. There were still a lot of open issues; the words were there, but the intent wasn't clear. What did "validation" mean? What was the "first written record" of a result? The latter point made sense on paper, but what did it mean when applied to computers acquiring data and writing results into memory?
In the next few years:
- regulatory requirements were produced by OHSA;
- standards like ISO 9000 were developed by the International Organization for Standardization (ISO);
- the EPA’s good laboratory practices (GLP) came into existence; and
- the International Society of Pharmaceutical Engineers (ISPE) released the Good Automated Manufacturing Practices (GAMP).
Companies and their laboratories increasingly had to organize and document their work according to external standards, guidelines, and regulations instead of relying on internally defined business practices. These covered more than experimental records; they extended to personnel and their qualifications, equipment maintenance and calibrations, reagent maintenance and testing, and test method documentation and validation. It created a large amount of clerical and administrative work. In addition, the results of that work had to be produced on-demand. Today, regulatory compliance remains one of the primary reasons for adopting a LIMS.[13]
Additionally, the requirements established by regulatory agencies provided the initial thrust for concerns over data governance, including data management, integrity, and utility. When everything was on paper, it was a matter of keeping paper documents well organized. There wasn't much else you could do. Then things changed, bringing with them new responsibilities and opportunities.
The integrity, security, and integration of instrument data is also a concern. Most instruments today either contain computing capabilities or are attached to computer systems with data storage, some temporary (e.g., pH meters and scales) and some long-term (e.g., IDSs). The more instruments we have, the more storage devices that hold laboratory results. In some cases, those results only exist on one storage device as long as a power surge doesn’t disrupt that system. Lab personnel probably know where the data they produce is, but that knowledge walks out the door every night. As such, laboratories need a centralized system to track all the data, information, results, calibrations, schedules, documents, etc., that labs produce. A small lab might be able to get by with a paper-based approach until they discover it no longer works, often when data previously believed to be secure becomes lost. Today, that centralized system is a LIMS.
Table 2 looks at a variety of these technological, regulatory, data governance, and data security issues, comparing what the state of thought was in the 1980s vs. today.
| ||||||||||||||||||||||||
1.5 Why a LIMS matters
Though external pressures to improve operations may have increased, the goals of laboratory work today are much the same as they were in the past: produce high-quality results, with the best productivity possible while keeping initial and ongoing costs to a reasonable level. The goals differ only slightly among the different types of labs:
- Testing: Whether supporting research programs or conducting contract-based analytical testing for clients, work efficiently and effectively to produce high-quality and accurate analytical test results while maintaining profitability.
- Research: Develop new products, solve production problems, and solve real-world problems (e.g., curing disease, developing alternative fuels, improving food production, etc.), while ensuring at the same time that analyses and studies are conducted consistently, accurately, and ethically.
- QA/QC: Support production and manufacturing operations through accurate and timely testing, while promoting higher levels of quality, consumer safety, and consumer satisfaction.
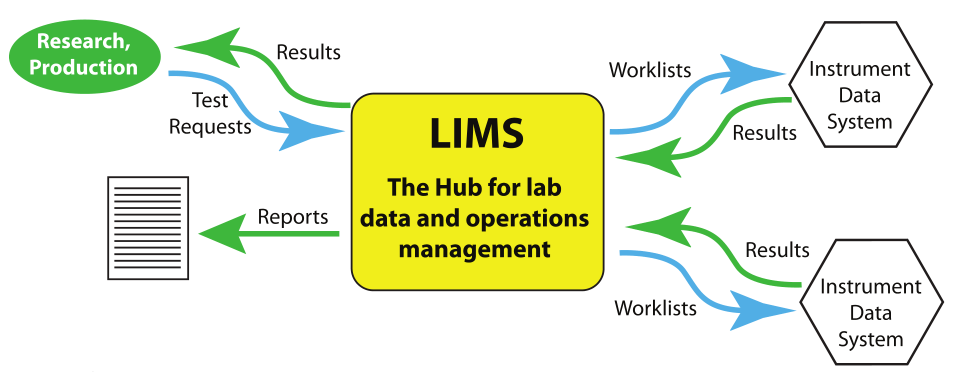
The goals are typically driven by management and other key stakeholders within the organization. Those same people hopefully recognize that meeting those goals requires more than simply managing the lab's personnel. The lab's success also hinges upon the management of equipment, infrastructure (including power), data, information, and the systems used to produce and manage the products of the lab work: analytical data and information. The previous sections examined the transition from paper-based to electronic lab, and from that examination we begin to see that LIMS and other laboratory informatics solutions are increasingly an important part of the equation for organizational success. This growing informatics architecture is a hierarchical structure with work requests and the needs of regulatory and corporate compliance flowing into it, and produced worklists being distributed to stations when the analysis is completed. The results then flow back into the LIMS, the central hub for this activity. Lab results can be interrogated, analyzed, and distributed as needed (Figure 1).
|
Your organization must be able to support the lab's goals, meet the demands of regulators, and meet your organization's responsibilities for data governance, all while maintaining an economically sound operation. Can that realistically be done without a LIMS supporting your operations? For example, would you run your accounting department without a software system? From that viewpoint, your accounting system's benefits to that department's work are similar in principle to those provided to your lab by a LIMS.
This section only touches upon why a LIMS matters to a laboratory and meeting its operational goals. Introducing a LIMS is an opportunity to solve operational management issues, improve productivity, make it easier to meet regulatory requirements and guidelines, turn the lab's data and information into a valuable asset, and meet modern demands for data governance. The next chapter goes into greater detail about the organizational, economic, and practical justifications behind acquiring and deploying a LIMS in your lab, further clarifying why the LIMS should matter to you and your organization, and why the benefits of LIMS far outweigh the costs.
References
- ↑ Gibbon, G.A. (1996). "A brief history of LIMS". Laboratory Automation and Information Management 32 (1): 1–5. doi:10.1016/1381-141X(95)00024-K.
- ↑ Skobelev, D.O.; Zaytseva, T.M.; Kozlov, A.D. et al. (2011). "Laboratory information management systems in the work of the analytic laboratory". Measurement Techniques 53 (10): 1182–1189. doi:10.1007/s11018-011-9638-7.
- ↑ Smith, K. (2 July 2019). "Integrated Informatics: Optimizing Food Quality and Safety by Building Regulatory Compliance into the Supply Chain". Food Safety Tech. https://foodsafetytech.com/feature_article/integrated-informatics-optimizing-food-quality-and-safety-by-building-regulatory-compliance-into-the-supply-chain/. Retrieved 19 July 2023.
- ↑ Apte, A. (20 October 2020). "Is Your Food Testing Lab Prepping for an ISO/IEC 17025 Audit?". Food Safety Tech. https://foodsafetytech.com/column/is-your-food-testing-lab-prepping-for-an-iso-iec-17025-audit/. Retrieved 19 July 2023.
- ↑ McDermott, P. (31 July 2018). "How Digital Solutions Support Supply Chain Transparency and Traceability". Food Safety Tech. https://foodsafetytech.com/column/how-digital-solutions-support-supply-chain-transparency-and-traceability/. Retrieved 19 July 2023.
- ↑ "5 reasons why software projects fail and how to make them succeed". DAC.digital Blog. DAC.digital. 19 April 2022. https://dac.digital/why-software-projects-fail-and-how-to-make-them-succeed/. Retrieved 19 July 2023.
- ↑ Singh, S. (15 June 2022). "23 Reasons Why Software Projects Fail (with Solutions)". Net Solutions Insights. Net Solutions. https://www.netsolutions.com/insights/23-reasons-why-software-projects-fail-with-solutions/. Retrieved 19 July 2023.
- ↑ Forbes Technology Council (31 March 2020). "14 Common Reasons Software Projects Fail (And How To Avoid Them)". Forbes. https://www.forbes.com/sites/forbestechcouncil/2020/03/31/14-common-reasons-software-projects-fail-and-how-to-avoid-them/amp/. Retrieved 19 July 2023.
- ↑ Liscouski, J. (April 2023). "Improving Lab Systems: From Paper to Spreadsheets to LIMS". LabLynx, Inc. https://www.lablynx.com/library/articles/improving-lab-systems-from-paper-to-spreadsheets-to-lims/. Retrieved 19 July 2023.
- ↑ Markin, Rodney S.; Hald, David L. (1 December 1992). "Cost justification of a laboratory information system: An analysis of projected tangible and intangible benefits" (in en). Journal of Medical Systems 16 (6): 281–295. doi:10.1007/BF00996362. ISSN 0148-5598. http://link.springer.com/10.1007/BF00996362.
- ↑ 11.0 11.1 Yu, Hoi-Ying Elsie; Lanzoni, Harold; Steffen, Tracy; Derr, Warren; Cannon, Kim; Contreras, Jeanene; Olson, Jordan Erik (1 January 2019). "Improving Laboratory Processes with Total Laboratory Automation" (in en). Laboratory Medicine 50 (1): 96–102. doi:10.1093/labmed/lmy031. ISSN 0007-5027. https://academic.oup.com/labmed/article/50/1/96/5047095.
- ↑ Golden, J.H. (1986). "Chapter 2: Economic Considerations of Laboratory Information Management Systems". In Provder, Theodore (in en). Computer Applications in the Polymer Laboratory. ACS Symposium Series. 313. Washington, DC: American Chemical Society. pp. 6–16. doi:10.1021/bk-1986-0313.ch002. ISBN 978-0-8412-0977-0. https://pubs.acs.org/doi/book/10.1021/bk-1986-0313.
- ↑ "Astrix 2020 LIMS Market Research Survey Report" (PDF). Astrix Technology, LLC. March 2021. https://astrixinc.com/wp-content/uploads/2021/03/Astrix-2020-LIMS-Market-Research-Report.pdf. Retrieved 19 July 2023.
- ↑ Halstead, Diane C; Sautter, Robert L (5 January 2023). "A Literature Review on How We Can Address Medical Laboratory Scientist Staffing Shortages" (in en). Laboratory Medicine 54 (1): e31–e36. doi:10.1093/labmed/lmac090. ISSN 0007-5027. https://academic.oup.com/labmed/article/54/1/e31/6712256.
- ↑ McDowall, R.D. (2017). "Understanding Data Governance, Part I". Spectroscopy 32 (2): 32–38. https://www.spectroscopyonline.com/view/understanding-data-governance. Retrieved 19 July 2023.
- ↑ McDowall, R.D. (2017). "Understanding Data Governance, Part II". Spectroscopy 32 (4): 12–18. https://www.spectroscopyonline.com/view/impact-data-governance-laboratory. Retrieved 19 July 2023.
Citation information for this chapter
Chapter: 1. Introduction to LIMS and its acquisition and deployment
Title: Justifying LIMS Acquisition and Deployment within Your Organization
Edition: First Edition
Author for citation: Joe Liscouski, Shawn E. Douglas
License for content: Creative Commons Attribution-ShareAlike 4.0 International
Publication date: July 2023