Difference between revisions of "Template:Article of the week"
Shawndouglas (talk | contribs) (Updated article of the week text) |
Shawndouglas (talk | contribs) (Updated article of the week text) |
||
| Line 1: | Line 1: | ||
<div style="float: left; margin: 0.5em 0.9em 0.4em 0em;">[[File: | <div style="float: left; margin: 0.5em 0.9em 0.4em 0em;">[[File:Fig2 DiNardo Toxins2020 12-4.png|240px]]</div> | ||
'''"[[Journal: | '''"[[Journal:Enzyme immunoassay for measuring aflatoxin B1 in legal cannabis|Enzyme immunoassay for measuring aflatoxin B1 in legal cannabis]]"''' | ||
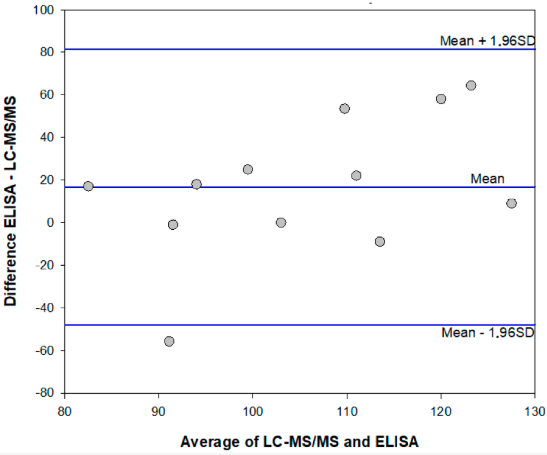
The [[ | The diffusion of the legalization of [[wikipedia:Cannabis|cannabis]] for recreational, medicinal, and nutraceutical uses requires the development of adequate analytical methods to assure the safety and security of such products. In particular, [[wikipedia:Aflatoxin|aflatoxins]] are considered to pose a major risk for the health of cannabis consumers. Among analytical methods that allow for adequate monitoring of food safety, [[immunoassay]]s play a major role thanks to their cost-effectiveness, high-throughput capacity, simplicity, and limited requirement for equipment and skilled operators. Therefore, a rapid and sensitive [[enzyme immunoassay]] has been adapted to measure the most hazardous [[wikipedia:Aflatoxin B1|aflatoxin B<sub>1</sub>]] in cannabis products. The assay was acceptably accurate (recovery rate: 78–136%), reproducible (intra- and inter-assay means coefficients of variation 11.8% and 13.8%, respectively), and sensitive (limit of detection and range of quantification: 0.35 ng mL<sup>−1</sup> and 0.4–2 ng mL<sup>−1</sup> ... ('''[[Journal:Enzyme immunoassay for measuring aflatoxin B1 in legal cannabis|Full article...]]''')<br /> | ||
<br /> | <br /> | ||
''Recently featured'': | ''Recently featured'': | ||
: ▪ [[Journal:The regulatory landscape of precision oncology laboratory medicine in the United States: Perspective on the past five years and considerations for future regulation|The regulatory landscape of precision oncology laboratory medicine in the United States: Perspective on the past five years and considerations for future regulation]] | |||
: ▪ [[Journal:Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes|Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes]] | : ▪ [[Journal:Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes|Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes]] | ||
: ▪ [[Journal:Health care and cybersecurity: Bibliometric analysis of the literature|Health care and cybersecurity: Bibliometric analysis of the literature]] | : ▪ [[Journal:Health care and cybersecurity: Bibliometric analysis of the literature|Health care and cybersecurity: Bibliometric analysis of the literature]] | ||
Revision as of 17:55, 19 July 2021
"Enzyme immunoassay for measuring aflatoxin B1 in legal cannabis"
The diffusion of the legalization of cannabis for recreational, medicinal, and nutraceutical uses requires the development of adequate analytical methods to assure the safety and security of such products. In particular, aflatoxins are considered to pose a major risk for the health of cannabis consumers. Among analytical methods that allow for adequate monitoring of food safety, immunoassays play a major role thanks to their cost-effectiveness, high-throughput capacity, simplicity, and limited requirement for equipment and skilled operators. Therefore, a rapid and sensitive enzyme immunoassay has been adapted to measure the most hazardous aflatoxin B1 in cannabis products. The assay was acceptably accurate (recovery rate: 78–136%), reproducible (intra- and inter-assay means coefficients of variation 11.8% and 13.8%, respectively), and sensitive (limit of detection and range of quantification: 0.35 ng mL−1 and 0.4–2 ng mL−1 ... (Full article...)
Recently featured:
- ▪ The regulatory landscape of precision oncology laboratory medicine in the United States: Perspective on the past five years and considerations for future regulation
- ▪ Institutional ELN-LIMS deployment: Highly customizable ELN-LIMS platform as a cornerstone of digital transformation for life sciences research institutes
- ▪ Health care and cybersecurity: Bibliometric analysis of the literature