LII:COVID-19 Testing, Reporting, and Information Management in the Laboratory/Diagnostic testing of COVID-19 and other coronaviruses
2.1 Testing terminology
As you read through this chapter, you may discover terms like "polymerase chain reaction" (PCR) and "lateral flow assay" (LFA). If you're a laboratorian or have a clinical background, you're likely to be familiar with these terms. However, it seems prudent to at least briefly discuss a few of them before delving into coronavirus testing itself.
2.1.1. Introduction
Living organisms store information in their genetic material, using DNA or RNA as the information carrier. That information, or genetic code, essentially provides instructions for organism development, function, growth, and reproduction. In the late twentieth century, researchers were laying the groundwork for molecular diagnostics, the concept of examining an organism's genetic code and its associated biological markers to diagnose and treat disease on a more personalized basis. This requires an assay, an investigative procedure for assessing the presence of, or measuring the amount or functional activity of, a target analyte. In the case of molecular diagnostics, and more broadly molecular biology, the target is biological in nature, and thus biological assays are used. These biological assays are designed to accurately detect the presence of or enable counts of biological molecules, including DNA, RNA, proteins, cells, bacteria, and virus particles (e.g., viral plaque assays).[1]
2.1.2 Polymerase chain reaction (PCR)
Polymerase chain reaction or PCR is a molecular biology method that takes small amounts of DNA sequences and makes copies of (amplifies) them to the point of having enough material to sufficiently study or work with. The base technique can yield results in several hours and has a high level of sensitivity, with its ability to amplify the DNA to counts of millions or billions. PCR has been used in molecular diagnostics for testing prospective parents for being genetic carriers of particular diseases (i.e., expanded carrier screening)[2][3], tissue typing to ensure more effective organ transplants[4], and analyzing mutations in oncogenes to customize cancer treatments.[5] However, the method has also been applied to forensic science[6] and epidemiology.[7]
PCR and its variations have been used to characterize and detect infectious disease organisms such as human immunodeficiency virus (HIV), pathogenic tuberculosis bacteria, and Bordetella pertussis, which causes whooping cough.[8] Additionally, a selection of viruses can have their RNA detected using PCR, though the primers (short single-strand DNA fragments) used in the process must by sympathetic to the virus' genetic structure to ensure that only target virus material is amplified.[9] As it turns out, coronaviruses are RNA viruses, having some of the longest genomes of any RNA virus, and, detrimentally, the highest known frequency of recombination (the exchange of genetic material with another organism); this broadly means high rates of virus mutation, which interferes with maintaining consistent diagnostic detection and therapy.[10][11]
PCR comes in several variant methods. For example, while PCR monitors the amplification portion at the end of the overall process, real-time or "quantitative" PCR (qPCR) allows for the generation rate of the amplified product to be monitored at a particular point during each PCR cycle. Reverse transcription PCR (RT-PCR) is a combinatory process, applying reverse transcription (creating complementary double-stranded DNA [cDNA] from an RNA template) with PCR. If RT-PCR incorporates qPCR, you end up with "real-time RT-PCR" (rRT-PCR), sometimes referred to as "quantitative RT-PCR" (qRT-PCR). In the case of using PCR for detecting coronaviruses, more often than not we see some variation of RT-PCR, with or without real-time amplification monitoring. (It's important to not assume all RT-PCR processes incorporate real-time methods.)
How does PCR work in practice? The simplified version (see this JAMA Patient Page for a useful graphical explanation, using COVID-19 as an example) has a clinician obtaining a biological specimen from the appropriate location or source material. Then, special techniques are used to isolate viral (or in some cases, bacterial) genetic material from the specimen. (If RT-PCR is performed, the next step of reverse transcription of the isolated viral RNA into cDNA is also performed.) Once the viral genetic material is isolated, suitable primers that are sympathetic to the structure of the isolated genetic material are introduced. Those primers bind to the virus' genetic material and begin making copies of it. Fluorescent or other biomarkers that were attached to the copies during the PCR process eventually release from the copies, and an attempt is made to detect the presence of those biomarkers. The presence or absence of these markers drives the determination of a positive or negative detection for the sought-after virus.[12]
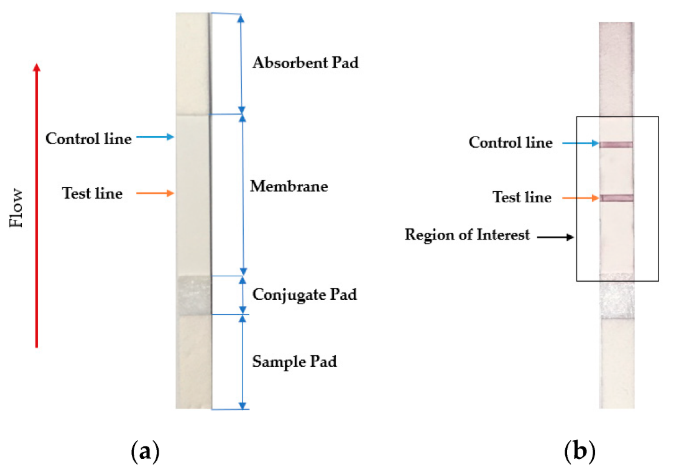
2.1.3 Lateral flow assay (LFA)
The lateral flow assay or LFA is another molecular biology method for detecting the presence of a target analyte in specimen material. In comparison to PCR, LFA has the advantages of being more rapid, low-cost, easy-to-use, and applicable at the point of care, though the average LFA is at best semi-quantitative in its results and has slightly lower sensitivity.[13][14] The method involves a cellulose-based strip with an ordered collection of specific "pads" and reagents that are reactive to a target analyte in a liquid, which is placed on the strip and moves across the various reagents using capillary and electrostatic interactions.[13][15][16] LFA has been used in molecular diagnostics for testing urine, saliva, sweat, serum, plasma, and other biological fluids for the presence of specific antigens and antibodies, as well as signs of gene amplification. The method has also been effectively applied to other industries outside healthcare, including the food and beverage, chemical, and environmental industries.[13]
In the realm of infectious disease, the LFA has played an important role in disease diagnosis and control, particularly in resource-constrained settings where resources are limited and point-of-care testing is critical to filling the gap.[17] Testing for the presence of an infectious agent in body fluids using LFA can be performed in two ways: lateral flow immunoassay (LFI) or lateral flow nucleic acid (LFNA). The immunoassay variant looks for antibodies created as a result of the presence of an infectious agent, whereas the nucleic acid variant is built to detect an amplified nucleic acid sequence specific to a target infectious agent.[18] Speaking specifically to the SARS-CoV-2 coronavirus, the antibody-based immunoassay method of lateral flow is predominantly used, targeting one of either a monoclonal antibody directed at a viral antigen, or a viral antigen that is recognizable by a patient's developed antibodies.[19][20]
Testing using LFI is generally as follows. A specialized adsorbent sample pad at one end of the LFI strip receives the specimen material. That material then migrates to the next conjugate release pad, where the specimen material is exposed to "antibodies that are specific to the target analyte and are conjugated to coloured or fluorescent particles."[13] The material then progresses to a detection zone containing antibodies or antigens that are fixed in the zone and intended to react to a specific analyte. If the analyte is present, a test line produces a visual, qualitative response, and a control line ensures proper liquid flow across the strip. A wicking pad at the other end properly maintains the flow of liquids across the strip.[13]
2.2.1 Severe acute respiratory syndrome (SARS)
Severe acute respiratory syndrome, otherwise known as SARS, arose in South China in late 2002. Caused by the SARS coronavirus (SARS-CoV) and believed to have originated from horseshoe bats[21], SARS eventually was contained in the summer of 2003. The last known infection was in April 2004, due to a laboratory accident.[22] During that time, the following sample collection and test procedures evolved from the related outbreaks (note that this is only a summary; consult the cited literature directly for full details)[23][24][25][26][27]:
- Determine that the patient is indicating clinical and/or epidemiological evidence of SARS (meets case definitions). As Knobler et al. put it: "SARS-CoV testing should be considered if no alternative diagnosis is identified 72 hours after initiation of the clinical evaluation and the patient is thought to be at high risk for SARS-CoV disease (e.g., is part of a cluster of unexplained pneumonia cases)."[25]
- Collect multiple specimen types at different time points of the patient's illness. Respiratory and plasma or serum specimens should be collected early into the first week of illness. Respiratory samples should be from nasopharyngeal aspirates and swabs of the upper respiratory tract or, in some cases, fluids from the lower respiratory tract using bronchoalveolar lavage, tracheal aspiration, or a pleural tap. (Sputum can also be collected.) Whole blood (5 to 10 ml) is collected into either a serum separator tube for blood serum or EDTA tube for blood plasma. Stool samples are also of import early on for virus isolation or detection and are useful in at least the first and second weeks of the illness. Blood serum is useful in weeks two and three for detecting a rising titre. Additionally, the literature also makes reference to methods of collecting specimens postmortem.
- Conduct testing. At the time, the two primary test types used were enzyme immunoassay (EIA; today more commonly known as ELISA[28]) for detection of serum antibody and reverse transcription polymerase chain reaction (RT-PCR) for detection of the virus' RNA. The U.S. Centers for Disease Control and Prevention (CDC) had this to say about these tests in May 2004[24]:
Both the EIA and the RT-PCR tests are sensitive and highly specific for SARS-CoV. The ability to diagnose SARS-CoV infection in a patient is often limited, however, by either the low concentration of virus in most clinical specimens (RT-PCR assays) or the time it takes a person to mount a measurable antibody response to SARS-CoV (serologic assays). The likelihood of detecting infection is increased if multiple specimens (e.g., stool, serum, respiratory tract specimens) are collected at several times during the course of illness.
- The literature also makes reference to an immunofluorescence assay (IFA) for detecting antibody, with the CDC calling its results "essentially identical to those for the EIA for SARS antibody."[24] Tangentially, isolation of SARS-CoV in cell culture from a clinical specimen is also referenced, though such activity is reserved for Biosafety Level 3 (BSL-3) laboratories.
- Confirm the results. Laboratory confirmation is based on one of 1. initial local lab detection and subsequent national reference lab confirmation of a validated serology-based test detection; 2. isolation of SARS-CoV in cell culture with subsequent confirmation from a validated test; or 3. initial local lab detection and subsequent national reference lab confirmation of SARS-CoV RNA from a validated RT-PCR test which used either two clinical specimens from different sources or two same-source clinical specimens from two different days.
- Additionally, in the case of serology, one of the following must be true:
- SARS-CoV serum antibodies are detected in a single serum specimen; or,
- a "four-fold or greater increase in SARS-CoV antibody titer between acute- and convalescent-phase serum specimens tested in parallel"[24] is detected; or,
- a "negative SARS-CoV antibody test result on acute-phase serum and positive SARS-CoV antibody test result on convalescent-phase serum tested in parallel"[24] is detected.
- Of note is the World Health Organization's (WHO) January 2004 cautionary message about serological diagnostics in not only SARS-CoV but other types of coronaviruses. At that time, they showed a level of unsurety in regards to how coronaviruses elicited serological cross-reactions and generated antigenic recall. They also preached caution in interpreting serological results in non-epidemic periods and when no viral sequence data are available. Finally, they also mentioned the added difficulties of rate cases when coinfection with a related human coronavirus occurs, "although the use of expressed proteins in Western blots may help to sort this out."[26] More than 15 years later, Loeffelholz and Tang put this concept into clearer terms, indicating that while "serological assays are not routinely used for diagnosis of [human coronavirus] infections due to the lack of commercial reagents," they still have important value "for understanding the epidemiology of emerging [human cornaviruses], including the burden and role of asymptomatic infections," as well as for antibody detection of novel and emerging coronaviruses.[29]
- Arrange for confirmatory testing to be performed by an appropriate test site in the case of a positive RT-PCR test.
- Report to state or local health departments details of patients radiographically confirmed with pneumonia with at least one SARS-CoV risk factor for exposure, clusters of healthcare workers with unexplained pneumonia, and any positive SARS-CoV test results. Additional international reporting of SARS by WHO Member States in regards to probable and laboratory-confirmed cases is also requested.
- Send off for an additional verification by an external member of the WHO's SARS Reference and Verification Laboratory Network before internationally announcing results as a laboratory-confirmed case.
2.2.2 Middle East respiratory syndrome (MERS)
Unlike SARS, Middle East respiratory syndrome, or MERS, continues to appear in the human population. Since its appearance in 2012, several thousand laboratory-confirmed cases of MERS have been reported to the WHO.[30] The virus MERS-CoV is believed to have originated from bats, which at some unknown point spread to Dromedary camels. Approximately 55 percent of MERS-CoV infections have come from direct contact with such camels, though it's not entirely clear how the rest of known cases have been caused[31] (Alshukairi et al. suggest asymptomatic or mildly symptomatic camel workers may serve as a possible transmission source[32]). The following sample collection and test procedures have evolved from working with the MERS-CoV virus (note that this is only a summary; consult the cited literature directly for full details)[33][34][35][36][37]:
- Determine that the patient is indicating clinical and/or epidemiological evidence of MERS (meets case definitions). "Testing for other respiratory pathogens using routinely available laboratory procedures, as recommended in local management guidelines for community-acquired pneumonia, should also be performed but should not delay testing for MERS-CoV."[35]
- Collect at a minimum both lower respiratory and upper respiratory tract samples. Lower respiratory tract specimens are typically the most revealing, as they have been shown to contain the highest viral load (due to the expression of the virus's cellular receptor DPP4 in the lower respiratory system). Bronchoalveolar lavage, tracheal aspiration, or a pleural tap can be used to collect specimens from the lower respiratory tract. (Sputum can also be collected.) Upper respiratory tract specimens (in this case, both a nasopharyngeal and an oropharyngeal swab are recommended) are also valuable in diagnosis, though extra care should be taken to ensure nasopharyngeal swabs gather secretions from the nasopharynx and not just the nostril. Nasopharyngeal aspiration is also an acceptable sample collection method for the upper respiratory tract.
- Regarding serum specimens, slight differences in guidance appear between WHO guidance and CDC guidance. The WHO appears to differentiate between symptomatic and asymptomatic patient testing, whereas the U.S. CDC seems to only indirectly differentiate the two. The WHO suggests if testing symptomatic patients, stick with lower and upper respiratory tract specimens, which will be tested using nucleic acid amplification (molecular) testing (NAAT). Serological testing of serum specimens should be used for symptomatic patients "only if NAAT is not available."[35] If this is the case, the WHO recommends paired samples, one collected within the first week of illness and the second about three to four weeks later. For asymptomatic patients in high-contact outbreak scenarios, the WHO recommends all three sample types (with respiratory samples taken preferably within 14 days of last documented contact).
- The current CDC guidance differentiates between molecular testing for active infections and serology for previous infections. The CDC adds that "MERS-CoV serology tests are for surveillance or investigational purposes and not for diagnostic purposes."[33] Whether or not to collect a serum specimen in MERS diagnostics may depend on the assay used, however. For example, the CDC, in its Version 2.1 guidance, indicates that testing using the CDC MERS rRT-PCR assay requires collection of serum in addition to upper and lower respiratory tract specimens. For that specific assay, the CDC differentiates between patients who've had symptom onset less than 14 days prior and those who've had it 14 days or later: if prior, serology is for the rRT-PCR test, and if later, serology is for antibody testing. In either case, 200 µL of serum is required.
- Conduct testing. NAAT methods like real-time reverse-transcription polymerase chain reaction (rRT-PCR) assays have been the most common tool for diagnosing MERS-CoV infection due to their high sensitivity. According to late 2018 research by Kelly-Cirino et al., at least 11 commercial single assay and five commercial multiplex assay kits are available (see Table S1, a PDF file, from their highly relevant paper), perhaps more as of April 2020. Serological antibody detection is performed using ELISA, indirect immunofluorescence (IIF), and microneutralization.
- Confirm the results. Laboratory confirmation of MERS-CoV infection is the same for both the WHO and the CDC: one of either a validated NAAT test providing a positive result for at least two different genomic targets, or a validated NAAT test providing a positive result for a specific genomic target along with sequencing confirmation of a separate genomic target. Persons under investigation who receive one negative NAAT result on a recommended specimen are considered to be negative for active MERS-CoV infection. The laboratory should consider testing additional specimens after the first negative. The CDC considers known MERS patients to be negative for active MERS-CoV infection after two consecutive negative NAAT tests on all specimens. The WHO adds: "A patient with a positive NAAT result for a single specific target without further testing but with a history of potential exposure and consistent clinical signs is considered a probable case."[35] The WHO also has additional guidance on using serology for confirming MERS-CoV infection for purposes of reporting under the International Health Regulations.
- Report using national reporting requirements. More broadly, state or local health departments should receive details about received specimens to be tested for MERS-CoV, even before testing begins. Regardless of result, the final positive or negative laboratory confirmation should also be reported to national authorities. If the infection becomes widespread, updates for each new confirmed case or suspected positive should also be made.
2.2.3 The common cold
Approximately 10 to 15 percent of cases of what we call the "common cold" are associated with an endemic coronavirus, of which are two distinct groups: HCoV-229E and HCoV-OC43.[38] Disease symptoms associated with these coronaviruses—typically in the form of respiratory infection and the symptoms that come with it—by themselves are typically mild[29], and laboratory testing isn't necessarily indicated for those immunocompetent individuals capable of self-limiting.[39] However, symptom overlap with pharyngitis and bronchitis, as well as the complication of pharyngitis and sinusitis also potentially having bacterial origin, can complicate clinical diagnosis. Additionally, as more antivirals that target a specific virus are created, and as concerns of unnecessarily using antibiotics to treat viral diseases grows[40][41], laboratory methods of respiratory virus diagnosis—particularly for those who are immunocompromised—have value.[38][39]
RT-PCR, a molecular method, has been used for well over a decade for detecting coronaviruses.[38][42] However, as molecular methods of analysis have expanded over the years, more rapid solutions for testing have been developed. For example, the GenMark ePlex rapid multiplex molecular diagnostics instrument and the ePlex Respiratory Pathogen Panel were evaluated in a multicenter trial by Babady et al. in 2017.[39] The panel is capable of testing for the presence of 15 viral types—including the -229E, -OC43, and two other coronaviruses—and two bacterial types in nasopharyngeal swab specimens, with results in typically less than two hours.[39] The cost associated with these sorts of tests, compared to their benefits, likely limits ubiquitous use at the first sign of a cold[39], but as molecular diagnostic technologies become more compact and easy-to-use, testing for infection by endemic human coronaviruses may become slightly more commonplace. However, as the authors point out, with no treatment for these endemic coronaviruses, any additional utility beyond diagnosing an illness as viral rather than bacterial would primarily be found in epidemiological studies of the associated genotyping data.[39]
2.3 Organizational and agency guidance on COVID-19 testing
NOTE: Information shown here may rapidly become outdated given how quickly response to pandemic testing can change. A full attempt to keep the content relevant will be made.
Early in the COVID-19 pandemic, laboratory guidance for testing for SARS-CoV-2 was relatively quick to evolve. The timely development and organized use of accurate assays and meaningful screening protocols, however, has been inconsistent but improving worldwide, with some countries more urgently and agilely responding than others.[43][44][45] With any novel virus, clinicians and public health experts are dealing with unknown factors. However, public health organizations and agencies have had a base to work from when creating laboratory testing guidance for a novel coronavirus, with more than 40 years of experience with coronavirus biology, pathogenesis, and diagnosis.[46] And while there are fundamental differences between SARS-CoV-2 and its predecessor SARS-CoV, they still share approximately 70 to 80 percent of their genetic code.[47][48] In fact, the WHO had draft guidance for laboratory testing out as early as January 10, 2020, before gene sequencing was even completed.[49] This guidance and similar draft guidance from national public health organizations and agencies have received steady revisions since as understanding of the virus has grown.
Similar to its predecessors SARS-CoV and MERS-CoV, RT-PCR has largely been the predominant diagnostic method used in guidance for detecting SARS-CoV-2's RNA in specimens and thus laboratory confirmation of COVID-19 cases. Other diagnostic methods such as isothermal amplification (e.g., LAMP) and antigen testing have also emerged as the pandemic has progressed. Serology has its place in testing as well, though with similar lessons from SARS and MERS that it's best used to test for past infection (typically after 14 days of suspected contact with a carrier, or mild symptoms) and thus potential short-term immunity due to the presence of antibodies in blood. It also has other uses; in its September 2020 guidance, the WHO said: "If negative NAAT results are obtained from a patient in whom SARS-CoV-2 infection is strongly suspected, a paired serum specimen could be collected."[50] On April 3, the U.S. Food and Drug Administration (FDA) approved the country's first COVID-19 serology test, created by Cellex, though Mayo Clinic was also on the verge of rolling out its own in-house serology test as well[51] As of September 2021, the U.S. FDA has granted emergency use authorizations (EUA) for 88 serology/antibody tests.[52] (Note: Johns Hopkins also appears to be maintaining a page tracking approved serology tests around the world.)
Primary testing guidance
The following sample collection and test procedures have evolved from the COVID-19 pandemic (note that this is only a summary; consult the cited literature directly for full details)[50][53][54][55][56]:
- Determine that the patient is indicating clinical and/or epidemiological evidence of COVID-19 (meets case definitions). Early on in the pandemic, case definitions and testing criteria were initially strict due to lack of test kits[57][58][59], but test kit availability has ramped up since, allowing for testing a wider group of symptomatic patients, as well as asymptomatic patients. However, clinicians are still encouraged to consider other causes for respiratory illness.[53]
- Collect at a minimum an upper respiratory tract (URT) specimen. Some guidance like that found in the U.K. also suggests collecting lower respiratory tract (LRT) specimens, whenever possible.[55] Broadly, it appears lower respiratory tract specimens such as sputum and bronchoalveolar lavage fluid are a more reliable specimen type for RT-PCR applications, as they have been shown to contain the highest viral load, in comparison to upper respiratory tract specimens.[60][61] In most cases, a URT will be sufficient; however, an LRT is especially useful when a patient tests negative with a URT but is still high suspicious of having COVID-19. Yet, as Wang et al. point out, "testing of specimens from multiple sites may improve the sensitivity and reduce false-negative test results,"[60] which is largely reflected in WHO, CDC, Public Health England (PHE), and Public Health Laboratory Network (PHLN; Australia) testing guidance.
- Slight differences in upper respiratory tract specimen collection procedures can be found between the WHO/CDC and PHE/PHLN. Both the WHO and CDC offer nasopharyngeal and oropharyngeal swabs as options. The WHO doesn't appear to give a preference, whereas the CDC has a preference for nasopharyngeal swabs but maintains oropharyngeal as still remaining "an acceptable specimen type."[54] In comparison, the latest PHE and PHLN guidance prefer the approach of collecting from both pharynx locations—even with the same swab—"to optimize the chances of virus detection."[56] Nasopharyngeal aspiration is also an acceptable sample collection method for the upper respiratory tract according to all mentioned entities except the PHLN, which appears to have removed mention of nasopharyngeal aspirate from its guidance sometime in 2021.[56]
- Regarding serum specimens, statements differ slightly. The WHO notes serology to be useful for retrospective case definition, using paired specimens from the acute and convalescent phases of the disease. The CDC doesn't make reference to serum or serology in their clinical specimen guidance. The PHE used to suggest hospital patients have "a sample for acute serology" taken, but that appears to have been removed from 2021 guidance.[55] The PHLN initially provided similar advice as the WHO, but in late April they expanded their guidance to discuss the value of serology.[56] They have also added collection recommendations for serology, in separate guidance, indicating that "serological testing before two weeks from the onset of symptoms may result in false negative results."[62]
- Finally, and more recently, potential evidence of saliva having diagnostic value for detecting SARS-CoV-2 has arisen. Xu et al. noted in published April 2020 research that the "diagnostic value of saliva specimens for ... nucleic acid examination remains limited but promising."[63] Another paper published in September 2020 provided similar thoughts, though was generally more optimistic than the paper published by Xu et al., suggesting saliva from the opening of the mouth (in contrast to Xu et al. and their finding of better results from saliva in the throat) may be viable specimen.[64] In fact, an April 2020 EUA by the FDA had been made for the first saliva-based COVID-19 test, produced by Vault Health, Inc.[65]
- As these and similar studies have been peer reviewed and methods validated, saliva has increasingly looked like a viable sample type. The CDC updated their guidance in October 2020 regarding saliva as a testing substrate. The CDC now notes: "Collect 1-5 ml of saliva in a sterile, leak-proof screw cap container. No preservative is required."[54] This is presumably in conjunction with tests approved for the use of saliva. Australia's PHLN updated their guidance in 2021 to include a full section on saliva testing and how to approach it, though cautioning it "does not advise routine use of saliva for diagnostic testing except in specific situations."[56]
- Conduct testing. NAAT methods like qRT-PCR have been the primary tools for diagnosing SARS-CoV-2 infection due to their high sensitivity. The PHLN provides the most background about PCR in their guidance, noting that "RT-PCR or TMA are the methods of choice to detect SARS-CoV-2 during the acute illness."[56] Viral cultures are little mentioned, though the PHLN underscores the idea that viral cultures for routine diagnoses are "of limited utility" and, if attempted, should only be performed in Biosafety Level 3 (BSL-3) laboratories.[56] As of August 2020, only the PHLN has made any specific recommendations for how serological testing should be conducted for testing past cases of COVID-19.[56] The current set of approved serology tests from around the world appear to use lateral flow immunoassay, ELISA, or neutralization methods.[59] Also note that at least in the U.S., the FDA in October 2020 discontinued review and approval of laboratory developed tests (LDTs), in favor of tests that would be more likely to increase access to testing or overall test capacity.[66]
- Confirm the results. The WHO notes that optimally a positive result should come from a NAAT method "with at least two independent targets on the SARS-CoV-2 genome." However, they recognize that "in areas with widespread transmission of SARS-CoV-2, a simple algorithm might be adopted with one single discriminatory target," though monitoring of potential mutations is recommended.[50] Most guidance notes that if testing produces one or more negative results, that doesn't necessarily rule out SARS-CoV-2 infection. If suspicion of infection remains high, particularly if only upper respiratory tract specimens were collected, additional specimens from the lower respiratory tract should be collected and analyzed. They also emphasize that both external and internal controls should be applied to NAAT runs to limit the chance of incorrect results.
- Report using state and, if applicable, national reporting requirements. (See the next chapter for more on reporting.) Regardless of result, the final positive or negative laboratory confirmation should also be reported to state and national authorities. In the U.S., for example, this means reporting to the local or state health department using the CDC's COVID-19 Worksheet. In Canada, reports are sent to the Public Health Agency of Canada (PHAC) via their Coronavirus Diseases (COVID-19) Case Report Form.
Additional considerations for point-of-care testing and self-collection
One of the long-term goals of healthcare professionals, governments, and test kit manufacturers has been to develop and increase access to point-of-care (POC) testing solutions for COVID-19. This has come with many challenges, but it is largely being realized. The CDC continues to update its Guidance for SARS-CoV-2 Point-of-Care and Rapid Testing[67], and, after the first "collect and test" at-home kit—the Lucira COVID-19 All-In-One Test Kit—was approved by the FDA in November 2020[68], many more were eventually approved for emergency use.[69] However, while these easy-use or quick-result tests are promising and welcome, a few additional testing considerations come with them. For example, the CDC has indicated that though FDA-authorized POC tests are approved for symptomatic individuals, "CLIA will temporarily allow CLIA-certified laboratories and other testing sites to use SARS-CoV-2 point-of-care and rapid antigen tests on asymptomatic people for the duration of the COVID-19 public health emergency."[67] Other recommendations include not reusing test devices or components, changing gloves at strategic points, and limiting opening of test materials until ready to begin testing.[67]
In some cases, such as drive-through testing sites, patient self-collection of a sample may be required. In these cases, it's vital that the patient is given clear and concise instructions on how to collect the sample before they begin the process. The American Association for Clinical Chemistry (AACC) notes that false-negative results are more likely with poor instruction and recommends healthcare providers provide patients with written or video instructions, along with any verbal instructions. Less invasive nasal swabs are typically being used in this case. During self-collection, the patient inserts the entire swab tip in one nasal cavity, makes four to five sweeping circles along the nasal wall for about 10 to 15 seconds, and then repeats the process in the opposite nostril.[70]
In the case of at-home test kits, instructions for sample collection and system use should be followed carefully. For example, the Lucira All-In-One Test Kit can be used in a CLIA-certified lab or "for prescription home use," meaning a healthcare provide must approve a prescription in order for the patient can use the test at home. Additionally, the healthcare provider will still be held responsible for reporting the test results they receive from the patient using the kit at home.[71]
Mitigating risk associated with false negatives
Before moving on, words of caution should be issued in regard to any COVID-19 testing conducted: false-negative results can be problematic.[72][73] One of the primary reasons they are problematic is that it may leave an otherwise asymptomatic individual to continue to unknowingly spread the virus further. Those individuals may relax physical distancing measures and become lax with their mask wearing, affecting others outside the clinical setting. Inside a clinical setting, a patient with a false negative "may be sent to the frontlines of care and inadvertently transmit the virus to patients and colleagues, further straining the already precarious ability of the health care system to respond to the pandemic."[73]
In a perspective piece published in Mayo Clinic Proceedings, West et al. of the Mayo Clinic offer four critical recommendations for society as we attempt to mitigate the risk associated with false negatives when performing clinical testing for COVID-19. Those recommendations are[73]:
- 1. Continue protective and preventative measures inside and outside the testing facility. This includes efforts such as physical distancing, regular hand-washing, regular disinfection of surfaces, and adequate personal protective equipment (PPE) for clinical staff (as well as the encouragement of proper mask wearing by others).
- 2. Develop and improve PCR and serological assays to be more sensitive and specific. The development and improvement process must include methodologically rigorous studies designed to limit the risk of biased results, as well as clearly reported test performance characteristics.
- 3. Assess patients carefully for their potential risk level for being infected. Confidence in negative test results may need to be lowered for health care workers and individuals in other high-risk groups. In general, given the uncertainty around viral load, asymptomatic transmission, and other disease characteristics, caution should be used with negative results in general.
- 4. Establish risk-based protocols for managing negative COVID-19 results. Truly low-risk individuals may not be a major concern when results come back negative. However, individuals in higher-risk categories may require more judicious protocols, e.g., delaying a return to a workplace (for self-isolation) despite receiving a negative and having no symptoms. (This may require a more sensitive follow-up test or at least a second negative in a repeat test, particularly among clinical workers.)
2.3.1 Regulatory considerations: HIPAA and CLIA
HIPAA
The Health Insurance Portability and Accountability Act (HIPAA) is a set of U.S. federal regulatory requirements that attempts to modernize the flow of healthcare information, stipulate how personally identifiable information (PII) (often referred to as "protected health information" or "PHI") maintained by healthcare providers and insurers should be protected from fraud and theft, and address limitations on healthcare insurance coverage in the U.S. HIPAA spans five sections or "titles," mandating health care information access, portability, privacy, and security, as well as stipulations on medical savings accounts, group health insurance requirements, and other tax- and legal-status-related issues.
Normally, HIPAA regulations would put strict requirements on how and when PII can be managed, used, shared, and stored. However, the COVID-19 pandemic has seen a relaxation of some of those requirements by the U.S. Department of Health and Human Services' (HHS) Office for Civil Rights (OCR). The HHS is currently maintaining a list of announcements, notifications, guidance documents, bulletins, and other resources as they relate to HIPAA and the public health emergency. Important notes pulled from that information reveal[74]:
- Family, friends, and others identified by the patient as being involved in their care may receive PHI from a covered health care provider, particularly when they deem that sharing in the patient's best interest. Additionally, "[a] covered entity also may share information about a patient as necessary to identify, locate, and notify family members, guardians, or anyone else responsible for the patient’s care, of the patient’s location, general condition, or death."
- "Covered health care providers will not be subject to penalties for violations of the HIPAA Privacy, Security, and Breach Notification Rules that occur in the good faith provision of telehealth during the COVID-19 nationwide public health emergency. This Notification does not affect the application of the HIPAA Rules to other areas of health care outside of telehealth during the emergency." This exercising of enforcement discretion will last until OCR provides a notice otherwise. Telehealth should optimally be performed in a private setting, whenever possible, using non-public facing communication technologies. Providers should opt to use the most secure services possible, "but will not be penalized for using less secure products in their effort to provide the most timely and accessible care possible to patients during the public health emergency."
- The OCR extended its enforcement discretion to include business associates of covered health care providers, allowing them to share PHI data "without risk of a HIPAA penalty." The OCR adds that "[s]ome HIPAA business associates have been unable to timely participate in these efforts [to ensure the health and safety of the public] because their BAAs do not expressly permit them to make such uses and disclosures of PHI." Like the enforcement discretion of telehealth provision by covered health care providers, the business associate must still make a good-faith effort in the "use or disclosure of the covered entity’s PHI" for public health and health oversight activities. Similarly, this exercising of enforcement discretion will last until OCR provides a notice otherwise.
- The OCR also extended its enforcement discretion to include covered health care providers and business associates operating community-based COVID-19 testing sites (CBTS)—"which includes mobile, drive-through, or walk-up sites that only provide COVID-19 specimen collection or testing services to the public"—during the public health emergency. This enforcement discretion, however, "does not apply to covered health care providers or their business associates when such entities are performing non-CBTS related activities, including the handling of PHI outside of the operation of a CBTS."
- Additionally, the OCR extended its enforcement discretion to "covered health care providers or their business associates in connection with the good faith use of online or web-based scheduling applications ... for the scheduling of individual appointments for COVID-19 vaccinations during the COVID-19 nationwide public health emergency."
- "[T]he HIPAA Privacy Rule permits a covered entity to disclose the protected health information (PHI) of an individual who has been infected with, or exposed to, COVID-19, with law enforcement, paramedics, other first responders, and public health authorities1 without the individual’s HIPAA authorization, in certain circumstances." OCR provides numerous examples, including: "when the disclosure is needed to provide treatment," "when such notification is required by law," when a public health authority must be notified "to prevent or control spread of disease," "when first responders may be at risk of infection," when preventing or lessening "a serious and imminent threat to the health and safety of a person or the public," and when responding to a law enforcement inquiring concerning a lawfully detained inmate or other individual.
- "The HIPAA Privacy Rule permits HIPAA covered entities (or their business associates on the covered entities’ behalf) to use or disclose PHI for treatment, payment, and health care operations, among other purposes, without an individual’s authorization." ICR provides more details in its guidance, noting however that reasonable effort must still be made to protect PHI, and that this does not apply to activities that constitute marketing.
- Similarly, the HIPAA Privacy Rule "permits covered entities or their business associates to disclose PHI to [a health information exchange (HIE)] for the HIE to report PHI to a [public health authority (PHA)] conducting public health activities" in a variety of circumstances. For more on those circumstances, see the December 2020 guidance].
- The public health emergency does not change restrictions on disclosing PHI to the media. This includes the prohibition of media crews in, for example, emergency departments where COVID-19 patients are being treated, as PHI is found everywhere in that setting. Only when every patient who is or will be in a potentially filmed area has signed a HIPAA authorization form can this be done.
CLIA
The Clinical Laboratory Improvement Amendments (CLIA) are a set of U.S. federal regulatory requirements applied to all non-research-based clinical laboratory testing performed on humans. These requirements are intended to further ensure a higher standard of quality in clinical laboratory testing, focusing in on improving the accuracy, reliability, and timeliness of tests. (The implications of these requirements on U.S. clinical labs and their ability to test for COVID-19 are discussed later, in the next section.)
CLIA uses seven different criteria to gauge and assign one of three complexity levels to laboratory devices and assays: high, moderate, and waived.[75][76] Additionally, CLIA mandates clinical laboratories handling specimens originating from the U.S. and its territories to apply for a CLIA certificate that is appropriate for the type of testing it performs. Labs using complex devices and assays would have to apply for a high complexity certificate, and so on. Waived tests are recognized as simple to perform with a low risk of erroneous results and include among others urinalysis for pregnancy and drugs of abuse, blood glucose and cholesterol tests, and fertility analysis.
Anything but waived testing requires meeting "the CLIA quality system standards, such as those for proficiency testing, quality control and assessment, and personnel requirements. The standards for moderate and high complexity testing differ only in the personnel requirements."[75] As the Centers for Medicare and Medicaid Services (CMS) points out in a frequently asked questions (FAQ) document—and as can be verified on the FDA's EUA page[52]—a huge majority of COVID-19 tests are only authorized for moderate or high complexity testing, and thus labs certified to do that sort of testing.[77] As of September 2021, only 14 of 260 FDA EUAed molecular diagnostic tests are approved to be performed in a CLIA-waived laboratory setting.[52]
CMS' FAQ, as well as their March 2020 guidance document, provides additional insight in regards CLIA and COVID-19[77][78]:
- CLIA regulations remain firmly in effect during the U.S.-declared public health emergency; a Section 1135 waiver, under the Social Security Act, that modifies or suspends CLIA requirements is not within the authorizing jurisdiction of the CLIA program. Additionally, CMS in general does not have the authority "to grant waivers or exceptions that are not established in statute or regulation."
- Laboratories choosing to use temporary testing sites for remotely (from home or another temporary location) viewing and reporting on cytology slides and images may do so if certain defined conditions are met. (Consult the memo for those defined conditions.)
- Proficiency testing (PT) is still required if a CLIA-certified lab is still performing testing and issuing patient results. However, should a PT provider need to postpone, suspend, or cancel a proficiency testing event, "[l]aboratories will not be penalized for lack of PT results ... so long as the cancelation is documented (including the notification from the PT program), and PT is conducted in a timely manner after the public health emergency ends. However, labs should consider performing their own self-assessment to ensure reliable testing."
- Alternate specimen collection devices (e.g., viral transport media, flocked nasopharyngeal swabs) used outside the manufacturer's instructions still require the establishment of performance specifications and assay validation prior to patient use. The FDA provides additional guidance on this topic.
- Laboratories performing laboratory developed tests (LDTs) are still required to be CLIA-certified and meet the requirements for high complexity testing. However, if the state government of such a laboratory has opted to take responsibility for authorizing an LDT (in order to expedite COVID-19 testing), then engagement with the FDA is not required.
- "CMS will temporarily exercise enforcement discretion under CLIA for the duration of the COVID-19 public health emergency for the use of authorized SARS-CoV-2 molecular and antigen POC tests on asymptomatic individuals outside of the test’s authorization."[79]
- As of May 2021[80], a CLIA specialty or subspecialty has not yet been assigned to COVID-19 testing authorized under the EUA pathway. Testing may be performed by laboratories based on intended use and by CMS specialty and subspecialty assigned to similar FDA-cleared or -approved tests with similar characteristics to the EUA being used.
2.4 Current test methods and their differences
NOTE: Information shown here may rapidly become outdated given how quickly response to pandemic testing can change. A full attempt to keep the content relevant will be made.
2.4.1 Background on the laboratory testing environment
Before continuing, it should be noted that many elements of the prior-mentioned COVID-19 testing guidance have governmental public health laboratories in mind. However, as the scale of the epidemic grew in 2020, and into 2021, the need for commercial laboratories and assay developers to get involved with efforts towards increasing analytical testing throughput—through a more rigorous public-private partnership—became abundantly clear.[81][82][83][84] At times during the pandemic, laboratory turnaround times have been slow due to a variety of factors, from lack of in-house laboratory resources to handle high test volumes and a slower-than-expected ramping up of test kit production[81][83][84], to actually getting diagnostic assays that are more rapid (yet still accurate) in their diagnosis, simpler to use, and useable at the point of care.[85][86] The good news is these rapid point-of-care molecular test kits are now becoming more readily available around the globe, including the United States, thanks in part to the U.S. Food and Drug Administration's Emergency Use Authorization (EUA) process.
Despite at-home kits becoming more available, quick and effective laboratory-based testing remains important, particularly as the delta variant continues to put strains on some U.S. states and their testing efforts into the late summer of 2021.[87][88] As such, it remains important to compare the U.S. laboratory testing environments of public health and large commercial testing labs with those of small, in-office clinical labs. In the U.S., all but research-based laboratory testing of human specimens is regulated under CLIA, including public health laboratories. Of the more than 297,000 non-exempt CLIA-registered labs in the U.S., only 38,742 or 13.0 percent of them are certified to perform moderate- and high-complexity testing as of August 2021.[89] Your public health labs and commercial diagnostic labs fall into this category, with investments in the personnel, training, certifications, and equipment to conduct those sorts of tests. Contrast this with the small yet numerous physician office laboratories (POLs) and how they operate. As of August 2021, some 42.4 percent of non-exempt CLIA-certified laboratories in the United States are POLs.[89] Located in an ambulatory or outpatient care setting, these labs test specimens from human patients to assist with the diagnosis, treatment, or monitoring of a patient condition. Testing in the clinical lab generally depends on three common methodologies to meet those goals: comparing the current value of a tested substance to a reference value, examining a specimen with microscopy, and detecting the presence of infection-causing pathogens.[90]
These POL's operate in a different environment than your average public health laboratory or reference lab that receives, processes, and reports on specimens en masse. The POL is typically a smaller operation, performing simple laboratory testing that can produce useful diagnostic data cheaply and rapidly. Rather than performing advanced pathology and molecular diagnostic procedures that require specific equipment and expertise, the POL typically focuses on blood chemistry, urinalysis, and other testing domains that don't require significant resources and provide rapid results. This can be seen in Centers for Medicare and Medicaid Services statistics reported in August 2021 that show 69.3 percent of non-exempt POLs in the U.S. are certified to provide CLIA-waived tests[89], "simple tests with a low risk for an incorrect result."[75]
As of September 2021, with 1. all but several handfuls of the current EUAed molecular in vitro diagnostic COVID-19 test kits being limited to moderate- and high-complexity CLIA labs[91] (the FDA claims that EUAed SARS-CoV-2 tests authorized for "use at the point of care" are considered CLIA-waived tests[92]), and 2. serology testing still being considered moderate- to high-complexity in nature[91], a significant majority of clinical laboratories are left with only a handful of CLIA-waived options for assisting with the effort to test the U.S. population for SARS-CoV-2 infection. Given the rapid rate of change at multiple levels of government and society and the continual spreading of misinformation[93][94][95], it's important to remember these fundamental differences in laboratories when trying to explain to someone why even in September 2021 they will still find it challenging to go to their primary care physician and get tested for SARS-CoV-2 in the doctor's office. Should researchers develop and the FDA provide EUAs for more CLIA-waived point-of-care assays, these differences may become less noticeable, and more people will be able to be tested.
2.4.2 PCR-based methods
As of September 2021, the U.S. Food and Drug Administration (FDA) has issued 260 EAUs for all types of molecular in vitro diagnostic test kits. Two hundred and eight of those 260 are only authorized to be used in CLIA-certified high-complexity laboratories, and 19 are rated for both moderate- and high-complexity. When looking at the makeup of the EUAs, a huge majority of those tests use some form of RT-PCR methods, with most using real-time or qualitative versions of RT-PCR (qRT-PCR). CLIA-waived RT-PCR tests are still rare, however. Eight PCR-based kits from five manufacturers have an additional authorization for point-of-care (POC) use (and thus CLIA-waived use) when used with their authorized POC devices[91]:
- BioFire Diagnostics' BioFire Respiratory Panel 2.1-EZ
- Cepheid's Xpert Omni SARS-CoV-2
- Cepheid's Xpert Xpress SARS-CoV-2
- Cepheid's Xpert Xpress SARS-CoV-2/Flu/RSV
- Mesa Biotech's Accula SARS-Cov-2 Test
- Roche Molecular Systems' cobas SARS-CoV-2
- Roche Molecular Systems' cobas SARS-CoV-2 & Influenza A/B
- Visby Medical's COVID-19 Point of Care Test
Of course, there are many more test kits than those approved in the United States. The Foundation for Innovative New Diagnostics (FIND) is currently "collating an overview of all SARS-CoV-2 tests commercially available or in development for the diagnosis of COVID-19."[96] As of September 2021, their site shows nearly 280 commercialized manual NAAT tests around the world (most being RT-PCR), with 30 in development. The AdVeritasDx test and controls database is also useful.[97]
2.4.3 LFA and isothermal amplification methods
LFAs are currently rare, but due to their advantages of being quick and useable at the point of care, some healthcare professionals have suggested that as a format for antigen and antibody (serology) testing, they could positively change the testing landscape.[19][98][99] As of September 2021, 25 of 88 serology tests that have received EUAs by the FDA are explicitly labeled as being LFAs, with 12 of those 25 being approved for POC/CLIA-waived use.[100] An article by Sheridan in Nature Biotechnology highlights a handful of others developed around the world (see their Table 1).[19] FIND shows more than 400 commercialized rapid diagnostic immunoassay tests around the world, though it's not clear how many of them actually LFAs (from their list, only 14 are explicitly stated as being LFA).[96] While LFAs have increasingly been approved around the world in 2021[100][96], it remains a question whether or not we continue to see more of them, at least in the United States.[98]
Also of note are isothermal amplification methods. Abbott's ID NOW and Cue Health's Cue COVID-19 and Cue COVID-19 Test for Home and OTC tests (the latter being the first isothermal amplification test approved for home use) are described by the FDA as using "isothermal nucleic acid amplification technology for the qualitative detection of SARS-CoV-2 viral nucleic acids."[101][102][103] Isothermal amplification tends to be an easier process to manage due to being able to keep amplification at a constant temperature.[104] In fact, Abbott has stated its EUAed ID NOW COVID-19 test can be completed within five minutes.[105] However, May 2020 findings by New York University put the test's accuracy into question. On July 1, 2020, an FDA spokesperson allegedly indicated receipt of 126 reports of "adverse events" concerning the test.[106] In 2020, some 393 complaints were reported to the FDA, with 1,492 complains being reported in 2021 (through July 31) according to an FDA MAUDE (Manufacturer and User Facility Device Experience) search.[107] The FDA was reportedly investigating the data and working with Abbott to have additional studies performed on the test's accuracy in 2020.[108] In October 2020, Abbott released additional study data showing overall sensitivity of 93.3% and specificity of 98.4%, emphasizing the ID NOW's best use with samples taken within seven days of symptom onset.[109] On August 27, 2021, the FDA re-issued its EUA for the ID NOW with updated in silico inclusivity analysis results (among other things)[110], but it's not clear if the FDA is continuing to work with Abbott on the test's accuracy claims.
Specific isothermal amplification techniques called loop-mediated isothermal amplification (LAMP) and reverse transcription LAMP (RT-LAMP) are beginning to emerge as options for COVID-19 testing. For example, Talis Biomedical is developing the Talis One COVID-19 system for point-of-care testing. It has received National Institutes of Health's Rapid Acceleration of Diagnostics (RADx) funding[111] and, should it receive its EUA (as of August 2021, it was still awaiting FDA authorization[112]), is expected to be among the first U.S.-approved RT-LAMP tests for COVID-19.[113] In November 2020, the first LAMP-based, prescription "collect and test" at-home kit—the Lucira COVID-19 All-In-One Test Kit—was approved by the FDA for emergency use.[68] Globally, examining FIND's list of nearly 280 commercialized manual NAAT tests around the world, five of them are explicitly shown to be some form of LAMP test. Multiple preprints on medRxiv and bioRxiv, as well as published papers, suggest that RT-LAMP could provide rapid results for SARS-CoV-2 testing.[114][115][116][117][118] However, it's apparent that adoption of LAMP as a COVID-19 test technique has been slow at best overall.
2.4.4 Blood serum
Blood serum or serology assays come in three common varieties: LFA, enzyme-linked immunosorbent assay (ELISA), or neutralization assay.[59] As discussed prior, LFAs are intended to be rapid point-of-care tools for qualitatively testing body fluids for patient antibodies or viral antigen. The ELISA is, in contrast, a more lab-bound method which produces results that are qualitative or quantitative. In the context of COVID-19 testing, ELISA tests for the presence of patient antibodies in a given specimen based upon whether or not an interaction is observed with the viral proteins present on the test plate. However, even if antibodies are present, ELISA isn't able to tell a clinician if those antibodies are able to protect against future infection. Neutralization assays are the lengthiest to complete, taking from three upwards to five days.[59] This is largely due to the fact that the assay depends on culturing cells that encourage growth of the target virus. Afterwards, introduced patient antibodies, if present, will fight to prevent viral infection of cells. This process is performed in decreasing concentrations, giving the clinician an opportunity to "visualize and quantify how many antibodies in the patient serum are able to block virus replication."[59] In contrast to ELISA, a neutralization assay is able to determine if a patient's antibodies are actively fighting against the target virus, even after recovering from the infection. In November 2020, the FDA granted an EUA to the first ELISA-based serology test to detect nuetralizing antibodies from recent or prior SARS-CoV-2 infection.[119]
Johns Hopkins' Center for Health Security appears to be tracking serology-based COVID-19 tests that are in development or have been approved in various parts of the globe. However, for the most up-to-date list of serology tests that have received EUAs in the United States, the FDA's EUA list appears to be the best source. As of September 2021, the FDA shows 88 serology assays approved for diagnostic use in the U.S. Of those 88, sixteen are explicitly listed as being ELISA-based.
A review of Johns Hopkins' tracking list showed more LFA-based tests among those approved in other parts of the world. Among their list of those still in development, an LFA stands out for integrating CRISPR detection.[120] CRISPR (clustered regularly interspaced short palindromic repeats) represents bacterial and archaeal DNA sequences derived from DNA fragments of previous infection. This genetic material can then be used as an activator of biomarkers when attached RNA "guides" find a match with target viral RNA in patient specimen.[121] This CRISPR-based LFA, called DETECTR, was further described in a paper published in October 2020, with the authors concluding it could be used "as a complementary technically independent approach to qRT-PCR, thereby increasing the testing capacity of medical microbiological laboratories and relieving the existent PCR-platforms for routine non-SARS-CoV-2 diagnostic testing."[122]
2.4.5 Antigen tests
An antigen is a substance—often a protein but may also be an environmental like a virus—that provokes the immune system to produce an antibody against it.[123] As such, another approach to testing for the presence of a virus in a specimen is to test for the antigen rather than the antibody. An antigen test is useful as a repeated surveillance test, but it has drawbacks as a one-time diagnostic test.[124][125][126] For COVID-19 and other viral infections, an antigen test has the advantage that specimen collection can typically be done with a simple nasal swab rather than a more invasive nasopharyngeal swab. Another advantage, on one hand, is that antigen testing is more rapid and convenient because the extraction and amplification steps of PCR are not used. On the other, antigen testing is less sensitive for the same reason: you test only what's there (rather than amplifying the amount for greater sensitivity).[125][127]
A theory increasingly gaining traction, however, is that "[a] higher frequency of testing makes up for poor sensitivity.”[125][126][128] Several researchers have shared pre-print and published research suggesting this outcome[125]:
Larremore and his colleagues have modeled the benefits of more frequent tests, even ones that are less accurate than today’s. Fast tests repeated every three days, with isolation of people who test positive, prevents 88% of viral transmission compared with no tests; a more sensitive test used every two weeks reduced viral transmission by about 40%, they report in a 27 June preprint on medRxiv. Paltiel and his colleagues reached much the same conclusion when they modeled a variety of testing regimes aimed at safely reopening a 5000-student university. In a 31 July paper in JAMA Network Open, they found that, with 10 students infected at the start of the semester, a test that identified only 70% of positive cases, given to every student every two days, could limit the number of infections to 28 by the end of the semester. Screening every seven days allowed greater viral spread, with the model predicting 108 infections.
As such, the utility of antigen testing, despite its lower sensitivity, appears to be surveillance situations where a large group of individuals who are at risk can be screened at regularly scheduled intervals of two to four days. The end result, in theory, would be few people who are target-positive would be missed, positives could be isolated and verified with a more sensitive test, and more target-positive people would be identified and isolated before reaching peak infectivity.[125][129] To be clear, it's not a perfect solution, but as Harvard epidemiologist Michael Mina and Boston University economist Laurence Kotlikoff suggest, "[w]e need the best means of detecting and containing the virus, not a perfect test no one can use."[129] In August 2020, a coalition of six U.S. state governors bought into that concept and agreed to work together with the Rockefeller Foundation, as well as the Quidel Corporation and Becton, Dickinson and Company, which had received FDA EUAs to market antigen tests for SARS-CoV-2.[128][130] As of September 2021, thirty-four FDA EUAs for antigen tests have been issues; 28 of those 34 include allowances for CLIA-waived testing, and 10 were authorized for home use.[131]
2.4.6 Testing alternatives and challenges
We’ve never faced this before, where clinical labs needed to very quickly be able to ramp up a test so fast.[132]
- Jennifer Doudna, Executive Director of the Innovative Genomics Institute, University of California, Berkeley
Though the dismantling and fund-cutting (proposed and real) of government programs designed to protect the populace from pandemics—as well as shortfalls in funding overall[133]—have likely hobbled local, national, and global response to COVID-19[134][135][136][137], it should be recognized that this pandemic may arguably represent a once-in-a-century type of event.[138][139] That said, even the most well-prepared governments would still face challenges in quickly learning about, controlling, and developing therapies for a novel disease agent. Shortages in supplies, workers, funding, and other resources are inevitably caused with a pandemic as people across all types of infrastructure fall ill.[133][140] This requires the additional human elements of adaptability, drive, and shared knowledge to find new and alternative solutions to fighting the challenges inherent to fighting against a novel disease.
See for example a non-peer-reviewed paper published on bioRxiv in early April 2020, where Schmid-Burgk et al. pointed out that though RT-PCR methods are the most common for currently testing for SARS-CoV-2, "global capacity for testing using these approaches, however, has been limited by a combination of access and supply issues for reagents and instruments." They proposed "a novel protocol that would allow for population-scale testing using massively parallel RT-LAMP by employing sample-specific barcodes." They claimed that a single heating step, pooled processing, and parallel sequencing with computational analysis would allow for the testing and tacking of "tens of millions of samples." Though the protocol was not validated with clinical samples, and concerns about sensitivity levels of RT-LAMP (an isothermal nucleic acid amplification technique that allows for RNA amplification) have been raised, the authors' work exemplifies the immediacy and ingenuity that has gone into finding workable solutions to a once-a-century problem.[114]
Another example of ingenuity in the face of difficult circumstances can be found at the University of California, Berkeley. At the beginning of the pandemic, its Innovative Genomics Institute (IGI) rapidly repurposed a 2,500-square-foot scientific lab into an automated diagnostic laboratory that could initially process more than 1,000 patient samples per day, with the ability to ramp up to 3,000 per day thanks to robotics and a streamlined workflow. Partnering with dozens of people from Thermo Fisher Scientific, Salesforce, Third Wave Analytics, and Hamilton Corp., the lab focused on not only turnaround time but also accuracy of results through automation. Their continued success, of course, still relies on a steady supply of reagents and related supplies from Thermo Fisher.[132][141]
Since the pandemic's start, others have also expressed concerns about the global supply of reagents necessary to test for SARS-CoV-2. Successful testing using RT-PCR requires two different enzymes: reverse transcriptase, for converting RNA to DNA, and polymerase, for amplifying the converted DNA. These enzymes and other reagent components may be instrument-specific, and at least one component has to be sympathetic to detection of the target virus' RNA. Little of this can be prepared without a proper sequence of the virus in question. Dr. Ronald Leonard, president and medical director of Cytocheck Laboratory and medical director of the Labette Health hospital, expressed the difficulties associated with reagent manufacturing thusly[142]:
With the instant demand for SARS-CoV-2 testing, the manufacturing process had to start from scratch for the SARS-CoV-2 specific components, and this did cause a lag time before reagents were available. The increased demand coupled with the decision to only allocate reagents to two national laboratories, some state health departments, and to "hot spots" has compounded the difficulty for laboratories like ours to obtain the necessary reagents to perform the testing.
Reports of COVID-19 test reagent shortages from various sources have appeared since March 2020.[143][144][145][146][147][148][149][130][129][150][151][152] Over time, those shortages even extended to supplies for non-COVID-19 tests such as those for strep throat, bronchitis, mycobacterial infections, urinary-tract infections, fungal infections, and sexually transmitted infections.[150][151][152] In the face of these supply challenges, some have taken matters into their own hands. Noting Irish laboratories' difficulties sourcing lysis buffer (for isolating molecules of interest and keeping them stable), Cork Institute of Technology's Dr. Brigid Lucey worked with several other virologists and microbiologists early in the pandemic, as well as with pharmaceutical company Eli Lilly, to produce a custom-formulated yet high-quality lysis buffer for not only Irish laboratories but also other countries can take advantage of. "We are happy to share what we found with other countries and it’s important our scientists retain their skills to make this kind of formulation because we may need to do this again in the future if we get other pandemics," she said.[146]
Another challenge lies in the accuracy of serology-based antibody tests, let alone how much they actually tell us about immunity. FierceBiotech's Conor Hale touched upon this in late April 2020[153]:
Compared to molecular tests—which sequence and match the RNA of the novel coronavirus to produce a result—the FDA has described antibody tests that gauge the body’s immune system response as a less-complicated endeavor that could proceed without review, dubbed “regulatory flexibility” by Commissioner Stephen Hahn. This policy shift has led to confusion, with some antibody test developers falsely claiming their tests are FDA-approved or could diagnose COVID-19 at home. Still others have sold outrightly fraudulent tests online.
At least in the U.S., these problems are compounded by company participation in test validation of EUAs being voluntary.[153] As a late April 2020 memorandum from Congress put it: "FDA is unable to validate the accuracy of antibody tests that are already on the market, and companies are ignoring requests from the Department of Health and Human Services (HHS) to voluntarily submit their tests for validation ... FDA has failed to police the coronavirus serological antibody test market, has taken no public enforcement action against any company, and has not conveyed any clear policy on serological tests..."[154] The entire memorandum is revealing in the challenges of attempting to relax social distancing measures under the pretense of the effectiveness of antibody testing. Entities such as the University of California - San Francisco[155] and the University of California - Berkeley[156] early on emphasized the importance of elements such as sensitivity, specificity, proper training, and the unknowns of the predictive ability of the test. Even assays running under trusted platforms such as PCR can reveal issues. For example, Thermo Fisher Scientific's TaqPath COVID019 Combo Kit, approved for EUA in March 2020, received scrutiny and updates to address issues with false-positive and -negative results[157][158], highlighting the difficulty of, at least early on in the pandemic, balancing the need for rapidly approving test kits for emergency use while also maintaining some semblance of oversight regarding their effectiveness and proper use. This has been further hampered by an August 2020 decision that stated the FDA could not require laboratory developed tests (LDTs) to be submitted for an EUA. As the Pew Charitable Trust's Liz Richardson argued in September 2021, reversing this decision "would enable labs to continue bringing innovative and effective new tests to market quickly while empowering FDA to protect the public from faulty products that allow infection to spread."[159]
Finally, while point-of-care testing, including at-home testing, has made strides in 2021[131], their need remains apparent. Dozens of at-home tests remained in development (many of them antigen tests) at the end of 2020, and even now a major barrier is found in making them both accurate and easy for untrained people to understand and use.[160][161] And as more at-home tests continue to receive EUAs in 2021, they are still not without concern, particularly in regards to ensuring that at-home test results get reported properly and rapidly.[160][162] One such early example at the end of 2020 was Lucira Health's LAMP-based Lucira COVID-19 All-In-One Test Kit, which was approved as a prescription-based at-home test, but with the stipulation that the prescribing healthcare provider would be responsible for reporting results.[68] Such a requirement arguably can and should be put in place for at-home antigen-based testing as well.[160]
As the pandemic churns through its second year, it's more important than ever for leaders across government, healthcare, and the media to continue to not spread misinformation and not make decisions based on poor scientific evidence. It will take organized efforts from multiple stakeholders—such as that found with a June 2020 NIH workshop on expanding and improving COVID-19 antibody tests[163]—to continue to find responsible solutions to the challenges we still face with this pandemic.
References
- ↑ AdvaMedDx (2013). "Introduction to Molecular Diagnostics: The Essentials of Diagnostics Series". pp. 19. http://www.epemed.org/online/www/content2/108/469/3172/listdownloads/3175/507/ENG/dxinsights.pdf. Retrieved 06 September 2021.
- ↑ Gregg, A.R. (2018). "Expanded Carrier Screening". Obstetrics and Gynecology Clinics of North America 45 (1): 103–112. doi:10.1016/j.ogc.2017.10.005. PMID 29428278.
- ↑ Chokoshvili, D.; Vears, D.F.; Borry, P. (2018). "Reproductive autonomy in expanded carrier screening: More than meets the eye?". Expert Review of Molecular Diagnostics 18 (12): 993–94. doi:10.1080/14737159.2018.1544496. PMID 30394810.
- ↑ Edgerly, C.H.; Weimer, E.T. (2018). "The Past, Present, and Future of HLA Typing in Transplantation". Methods in Molecular Biology 1802: 1–10. doi:10.1007/978-1-4939-8546-3_1. PMID 29858798.
- ↑ Loda, M. (1994). "Polymerase chain reaction-based methods for the detection of mutations in oncogenes and tumor suppressor genes". Human Pathology 25 (6): 564–71. doi:10.1016/0046-8177(94)90220-8. PMID 7912220.
- ↑ Ninfa, A.J.; Ballou, D.P.; Benore, M. (2009). Fundamental Laboratory Approaches for Biochemistry and Biotechnology. Wiley. pp. 408–410. ISBN 9780470087664. https://books.google.com/books?id=k6_XQwAACAAJ&pg=PA408. Retrieved 08 April 2020.
- ↑ Hamborsky, J.; Kroger, A.; Wolfe, C., ed. (2015). Epidemiology and Prevention of Vaccine-Preventable Diseases (13th ed.). Centers for Disease Control and Prevention. https://www.cdc.gov/vaccines/pubs/pinkbook/index.html. Retrieved 08 April 2020.
- ↑ Buckingham, L. (2019). "Chapter 11: Detection and Identification of Microorganisms". Molecular Diagnostics: Fundamentals, Methods and Clinical Applications (3rd ed.). F.A. Davis Company. pp. 301–343. ISBN 9780803699540. https://books.google.com/books?hl=en&lr=&id=dJWNDwAAQBAJ&pg=301.
- ↑ Kim, H.; Kang, N.; An, K. et al. (2017). "MRPrimerV: A database of PCR primers for RNA virus detection". Nucleic Acids Research 45 (D1): D475–81. doi:10.1093/nar/gkw1095. PMC PMC5210568. PMID 27899620. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5210568.
- ↑ Makin, S. (5 February 2020). "How Coronaviruses Cause Infection—from Colds to Deadly Pneumonia". Scientific American. https://www.scientificamerican.com/article/how-coronaviruses-cause-infection-from-colds-to-deadly-pneumonia1/. Retrieved 08 April 2020.
- ↑ Rohde, R. (31 January 2020). "2019 Novel Coronavirus (2019-nCoV) Update: Uncoating the Virus". American Society for Microbiology. https://asm.org/Articles/2020/January/2019-Novel-Coronavirus-2019-nCoV-Update-Uncoating. Retrieved 08 April 2020.
- ↑ Hadaya, J.; Schumm, M.; Livingston, E.H. (2020). "Testing Individuals for Coronavirus Disease 2019 (COVID-19)". JAMA. doi:10.1001/jama.2020.5388. PMID 32236503.
- ↑ 13.0 13.1 13.2 13.3 13.4 Koczula, K.M.; Gallotta, A. (2016). "Lateral flow assays". Essays in Biochemistry 60 (1): 111–20. doi:10.1042/EBC20150012. PMC PMC4986465. PMID 27365041. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4986465.
- ↑ Liu, Z.; Hu, J.; Qu, Z.; Xu, F. (2018). "Chapter 8: Paper-Based Immunoassays". In Vashist, S.K.; Luong, J.H.T.. Handbook of Immunoassay Technologies: Approaches, Performances, and Applications. Academic Press. pp. 183–202. ISBN 9780128117620. https://books.google.com/books?id=jSk0DwAAQBAJ&pg=PA183. Retrieved 08 April 2020.
- ↑ Jauset-Rubio, M.; Svobodová, M.; Mairal, T. et al. (2016). "Ultrasensitive, rapid and inexpensive detection of DNA using paper based lateral flow assay". Scientific Reports 6: 37732. doi:10.1038/srep37732. PMC PMC5123575. PMID 27886248. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5123575.
- ↑ Foysal, K.H.; Seo, S.E.; Kim, M.J. et al. (2019). "Analyte Quantity Detection from Lateral Flow Assay Using a Smartphone". Sensors 19 (21): 4812. doi:10.3390/s19214812. PMC PMC6864604. PMID 31694281. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6864604.
- ↑ Hanafiah, K.M.; Arifin, N.; Bustami, T. et al. (2017). "Development of Multiplexed Infectious Disease Lateral Flow Assays: Challenges and Opportunities". Diagnostics 7 (3): 51. doi:10.3390/diagnostics7030051. PMC PMC5617951. PMID 28880218. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5617951.
- ↑ Ngom, B.; Guo, Y.; Wang, X.; Bi, D. (2010). "Development and application of lateral flow test strip technology for detection of infectious agents and chemical contaminants: A review". Analytical and Bioanalytical Chemistry 397: 1113–1135. doi:10.1007/s00216-010-3661-4. PMID 20422164.
- ↑ 19.0 19.1 19.2 Sheridan, C. (23 March 2020). "Fast, portable tests come online to curb coronavirus pandemic". Nature Biotechnology - News. doi:10.1038/d41587-020-00010-2. https://www.nature.com/articles/d41587-020-00010-2. Retrieved 08 April 2020.
- ↑ McKie, R. (9 December 2017). "Scientists trace 2002 Sars virus to colony of cave-dwelling bats in China". The Guardian. https://www.theguardian.com/world/2017/dec/10/sars-virus-bats-china-severe-acute-respiratory-syndrome. Retrieved 03 April 2020.
- ↑ Normile, D. (2004). "Mounting Lab Accidents Raise SARS Fears". Science (5671): 659–61. doi:10.1126/science.304.5671.659. PMID 15118129.
- ↑ New York State Department of Health (February 2004). "Laboratory Testing for SARS". State of New York. https://www.health.ny.gov/diseases/communicable/sars/sars_reporting/attachment_6_dear_doctor_lab.htm. Retrieved 03 April 2020.
- ↑ 24.0 24.1 24.2 24.3 24.4 Centers for Disease Control and Prevention (21 May 2004). "Public Health Guidance for Community-Level Preparedness and Response to Severe Acute Respiratory Syndrome (SARS), Version 2 - Supplement F: Laboratory Guidance" (PDF). Centers for Disease Control and Prevention. https://www.cdc.gov/sars/guidance/f-lab/downloads/F-lab-full.pdf. Retrieved 03 April 2020.
- ↑ 25.0 25.1 Knobler, S.; Mahmoud, A.; Lemon, S. et al., ed. (2004). "Appendix C: In the absence of SARS-CoV transmission worldwide: Guidance for surveillance, clinical and laboratory evaluation, and reporting". Learning from SARS: Preparing for the Next Disease Outbreak. National Academies Press. pp. 292–302. doi:10.17226/10915. ISBN 9780309182157.
- ↑ 26.0 26.1 World Health Organization (23 January 2004). "WHO SARS International Reference and Verification Laboratory Network: Policy and Procedures in the Inter-Epidemic Period". World Health Organization. http://www.who.int/csr/resources/publications/en/SARSReferenceLab.pdf. Retrieved 03 April 2020.
- ↑ Liang, G.; Chen, Q.; Xu, J. et al. (2004). "Laboratory Diagnosis of Four Recent Sporadic Cases of Community-acquired SARS, Guangdong Province, China". Emerging Infectious Diseases 10 (10): 1774–81. doi:10.3201/eid1010.040445. PMC PMC3323270. PMID 15504263. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3323270.
- ↑ Lequin, R.M. (2005). "Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA)". Clinical Chemistry 51 (12): 2415–18. doi:10.1373/clinchem.2005.051532. PMID 16179424.
- ↑ 29.0 29.1 Loeffelholz, M.J.; Tang, T.-W. (2020). "Laboratory diagnosis of emerging human coronavirus infections – The state of the art". Emerging Microbes & Infections 9 (1): 747–56. doi:10.1080/22221751.2020.1745095. PMID 32196430.
- ↑ Bernard-Stoecklin, S.; Nikolay, B.; Assiri, A. et al. (2019). "Comparative Analysis of Eleven Healthcare-Associated Outbreaks of Middle East Respiratory Syndrome Coronavirus (Mers-Cov) from 2015 to 2017". Scientific Reports 9: 7385. doi:10.1038/s41598-019-43586-9. PMC PMC6517387. PMID 31089148. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6517387.
- ↑ Banerjee, A.; Kulcsar, K.; Misra, V. et al. (2019). "Bats and Coronaviruses". Viruses 11 (1): E41. doi:10.3390/v11010041. PMC PMC6356540. PMID 30634396. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6356540.
- ↑ Alshukairi, A.N.; Zheng, J.; Zhao, J. et al. (2018). "High Prevalence of MERS-CoV Infection in Camel Workers in Saudi Arabia". mBio 9 (5): e01985-18. doi:10.1128/mBio.01985-18. PMC PMC6212820. PMID 30377284. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6212820.
- ↑ 33.0 33.1 Centers for Disease Control and Prevention (2 August 2019). "CDC Laboratory Testing for Middle East Respiratory Syndrome Coronavirus (MERS-CoV)". Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/mers/lab/lab-testing.html. Retrieved 04 April 2020.
- ↑ Centers for Disease Control and Prevention (2 August 2019). "Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens from Persons Under Investigation (PUIs) for Middle East Respiratory Syndrome Coronavirus (MERS-CoV) – Version 2.1". Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/mers/guidelines-clinical-specimens.html. Retrieved 04 April 2020.
- ↑ 35.0 35.1 35.2 35.3 World Health Organization (January 2018). "Laboratory testing for Middle East Respiratory Syndrome Coronavirus: Interim guidance". WHO/MERS/LAB/15.1/Rev1/2018. World Health Organization. https://www.who.int/csr/disease/coronavirus_infections/mers-laboratory-testing/en/. Retrieved 04 April 2020.
- ↑ Al-Abdely, H.M.; Midgley, C.M.; Alkhamis, A.M. et al. (2019). "Middle East Respiratory Syndrome Coronavirus Infection Dynamics and Antibody Responses among Clinically Diverse Patients, Saudi Arabia". Emerging Infectious Diseases 25 (4): 753-766. doi:10.3201/eid2504.181595.
- ↑ Kelly-Cirino, C.; Mazzola, L.T.; Chua, A. et al. (2019). "An updated roadmap for MERS-CoV research and product development: focus on diagnostics". BMJ Global Health 4 (Suppl. 2): e001105. doi:10.1136/bmjgh-2018-001105. PMC PMC6361340. PMID 30815285. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6361340.
- ↑ 38.0 38.1 38.2 Wat, D. (2004). "The common cold: A review of the literature". European Journal of Internal Medicine 15 (2): 79–88. doi:10.1016/j.ejim.2004.01.006. PMID 15172021.
- ↑ 39.0 39.1 39.2 39.3 39.4 39.5 Babady, N.E.; England, M.R.; Jurcic Smith, K.L. et al. (2018). "Multicenter Evaluation of the ePlex Respiratory Pathogen Panel for the Detection of Viral and Bacterial Respiratory Tract Pathogens in Nasopharyngeal Swabs". Journal of Clinical Microbiology 56 (2): e01658-17. doi:10.1128/JCM.01658-17. PMC PMC5786739. PMID 29212701. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5786739.
- ↑ Jenison, R. (30 November 2016). "Rapid lab tests can help reduce antibiotic resistance". STAT. https://www.statnews.com/2016/11/30/antibiotic-resistance-molecular-diagnostics/. Retrieved 03 April 2020.
- ↑ Roy, K. (26 September 2018). "Rapid test for viral infections reduces unnecessary antibiotic prescribing". Healio. https://www.healio.com/news/infectious-disease/20180926/rapid-test-for-viral-infections-reduces-unnecessary-antibiotic-prescribing. Retrieved 06 September 2021.
- ↑ Mahoney, J.B. (2008). "Detection of Respiratory Viruses by Molecular Methods". Clinical Microbiology Reviews 21 (4): 716–47. doi:10.1128/CMR.00037-07. PMC PMC2570148. PMID 18854489. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2570148.
- ↑ Subbaraman, N.; Callaway, E. (23 March 2020). "Coronavirus tests: Researchers chase new diagnostics to fight the pandemic". Nature - News Explainer. doi:10.1038/d41586-020-00827-6. https://www.nature.com/articles/d41586-020-00827-6. Retrieved 05 April 2020.
- ↑ Apuzzo, M.; Gebrekidan, S. (20 March 2020). "Can’t Get Tested? Maybe You’re in the Wrong Country". The New York Times. https://www.nytimes.com/2020/03/20/world/europe/coronavirus-testing-world-countries-cities-states.html. Retrieved 05 April 2020.
- ↑ Hindsley, G. (28 March 2020). "The Lost Month: How a Failure to Test Blinded the U.S. to COVID-19". The New York Times. https://www.nytimes.com/2020/03/28/us/testing-coronavirus-pandemic.html. Retrieved 05 April 2020.
- ↑ Denison, M.R. (2004). "Coronavirus Research: Keys to Diagnosis, Treatment, and Prevention of SARS". Learning from SARS: Preparing for the Next Disease Outbreak. Institute of Medicine. pp. 137–72. doi:10.17226/10915. ISBN 9780309182157. https://www.nap.edu/read/10915/chapter/5.
- ↑ Ceccarelli, M.; Berretta, M.; Venanzi Rullo, E. et al. (2020). "Differences and similarities between Severe Acute Respiratory Syndrome (SARS)-CoronaVirus (CoV) and SARS-CoV-2. Would a rose by another name smell as sweet?". European Review for Medical and Pharmacological Sciences 24 (5): 2781-2783. doi:10.26355/eurrev_202003_20551. PMID 32196628.
- ↑ Wilder-Smith, A.; Chiew, C.J.; Lee, V.J. (2020). "Can we contain the COVID-19 outbreak with the same measures as for SARS?". The Lancet Infectious Diseases. doi:10.1016/S1473-3099(20)30129-8. PMC PMC7102636. PMID 32145768. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7102636.
- ↑ World Health Organization (10 January 2020). "Laboratory testing of human suspected cases of novel coronavirus (nCoV) infection: Interim guidance 10 January 2020". WHO/2019-nCoV/laboratory/2020.1. World Health Organization. https://apps.who.int/iris/bitstream/handle/10665/330374/WHO-2019-nCoV-laboratory-2020.1-eng.pdf. Retrieved 05 April 2020.
- ↑ 50.0 50.1 50.2 World Health Organization, et al. (11 September 2020). "Diagnostic testing for SARS-CoV-2". WHO/2019-nCoV/laboratory/2020.6. World Health Organization. https://apps.who.int/iris/handle/10665/334254. Retrieved 18 November 2020.
- ↑ Terry, M. (3 April 2020). "Cellex and Mayo Clinic Launch Tests to Determine COVID-19 Immunity from Previous Exposure". BioSpace. https://www.biospace.com/article/fda-approves-1st-covid-19-antibody-test/. Retrieved 05 April 2020.
- ↑ 52.0 52.1 52.2 "In Vitro Diagnostics EUAs". U.S. Food and Drug Administration. 20 August 2020. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/vitro-diagnostics-euas#individual-serological. Retrieved 23 August 2020.
- ↑ 53.0 53.1 Centers for Disease Control and Prevention (2 August 2021). "Overview of Testing for SARS-CoV-2". Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/hcp/testing-overview.html. Retrieved 06 September 2021.
- ↑ 54.0 54.1 54.2 Centers for Disease Control and Prevention (26 February 2021). "Interim Guidelines for Collecting and Handling of Clinical Specimens for COVID-19 Testing". Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-nCoV/lab/guidelines-clinical-specimens.html. Retrieved 06 September 2021.
- ↑ 55.0 55.1 55.2 Public Health England (29 March 2021). "COVID-19: Guidance for sampling and for diagnostic laboratories". U.K Government. https://www.gov.uk/government/publications/wuhan-novel-coronavirus-guidance-for-clinical-diagnostic-laboratories. Retrieved 06 September 2021.
- ↑ 56.0 56.1 56.2 56.3 56.4 56.5 56.6 56.7 Public Health Laboratory Network (22 June 2021). "PHLN guidance on laboratory testing for SARS-CoV-2 (the virus that causes COVID-19)". Department of Health, Australian Government. https://www.health.gov.au/resources/publications/phln-guidance-on-laboratory-testing-for-sars-cov-2-the-virus-that-causes-covid-19. Retrieved 06 September 2021.
- ↑ Centers for Disease Control and Prevention (27 April 2020). "Evaluating and Testing Persons for Coronavirus Disease 2019 (COVID-19)". Centers for Disease Control and Prevention. Archived from the original on 01 May 2020. https://web.archive.org/web/20200501002841/https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-criteria.html. Retrieved 08 July 2020.
- ↑ Branswell, H. (4 April 2020). "CDC launches studies to get more precise count of undetected Covid-19 cases". STAT. https://www.statnews.com/2020/04/04/cdc-launches-studies-to-get-more-precise-count-of-undetected-covid-19-cases/. Retrieved 05 April 2020.
- ↑ 59.0 59.1 59.2 59.3 59.4 Center for Health Security (26 August 2021). "Serology tests for COVID-19". Johns Hopkins University. https://www.centerforhealthsecurity.org/covid-19TestingToolkit/serology/Serology-based-tests-for-COVID-19.html. Retrieved 06 September 2021.
- ↑ 60.0 60.1 Wang, W.; Xu, Y.; Gao, R. et al. (2020). "Detection of SARS-CoV-2 in Different Types of Clinical Specimens". JAMA. doi:10.1001/jama.2020.3786. PMC PMC7066521. PMID 32159775. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7066521.
- ↑ Yu, F.; Yan, L.; Wang, N. et al. (2020). "Quantitative Detection and Viral Load Analysis of SARS-CoV-2 in Infected Patients". Clinical Infectious Diseases: ciaa345. doi:10.1093/cid/ciaa345. PMID 32221523.
- ↑ Public Health Laboratory Network (3 September 2020). "PHLN guidance for serological testing in COVID-19" (Word). Department of Health, Australian Government. https://www.health.gov.au/sites/default/files/documents/2020/09/phln-guidance-for-serological-testing-in-covid-19-phln-guidance-on-serological-testing-in-covid-19.docx. Retrieved 06 September 2021.
- ↑ Xu, R.; Cui, B.; Duan, X. et al. (2020). "Saliva: Potential diagnostic value and transmission of 2019-nCoV". International Journal of Oral Science 12: 11. doi:10.1038/s41368-020-0080-z.
- ↑ Wyllie, A.L.; Fournier, J.; Casanovas-Massana, A. et al. (2020). "Saliva or Nasopharyngeal Swab Specimens for Detection of SARS-CoV-2". New England Journal of Medicine 383: 1283–6. doi:10.1056/NEJMc2016359. PMC PMC7484747. PMID 32857487. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7484747.
- ↑ Vault Health (14 April 2020). "Vault Health Launches First-of-its-Kind Saliva-based FDA EUA Approved Test for COVID-19". PR Newswire. https://www.prnewswire.com/news-releases/vault-health-launches-first-of-its-kind-saliva-based-fda-eua-approved-test-for-covid-19-301039633.html. Retrieved 01 May 2020.
- ↑ Fox, A. (12 October 2020). "EUAs for Laboratory Developed COVID-19 Tests Will No Longer Be Reviewed by the FDA". JDSupra. https://www.jdsupra.com/legalnews/euas-for-laboratory-developed-covid-19-91593/. Retrieved 18 November 2020.
- ↑ 67.0 67.1 67.2 Centers for Disease Control and Prevention (13 November 2020). "Guidance for SARS-CoV-2 Point-of-Care Testing". Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/lab/point-of-care-testing.html. Retrieved 07 September 2021.
- ↑ 68.0 68.1 68.2 Romo, V. (17 November 2020). "FDA Approves 1st At-Home Coronavirus Test". NPR. https://www.npr.org/sections/coronavirus-live-updates/2020/11/17/936055284/fda-approves-first-at-home-coronavirus-test. Retrieved 18 November 2020.
- ↑ "In Vitro Diagnostics EUAs - Antigen Diagnostic Tests for SARS-CoV-2". U.S. Food and Drug Administration. 7 September 2021. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2. Retrieved 07 September 2021.
- ↑ American Association for Clinical Chemistry (12 November 2020). "Proper Self-collection of Nasal Swabs Critical for Accurate COVID-19 Testing". Lab Tests Online. American Association for Clinical Chemistry. https://labtestsonline.org/news/proper-self-collection-nasal-swabs-critical-accurate-covid-19-testing. Retrieved 18 November 2020.
- ↑ "Lucira COVID-19 All-In-One Test Kit - Instruction for Use" (PDF). Lucira Health. 2020. https://www.fda.gov/media/143808/download. Retrieved 18 November 2020.
- ↑ Beale, S. (29 June 2020). "Multiple Studies Raise Questions About Reliability of Clinical Laboratory COVID-19 Diagnostic Tests". Dark Daily. https://www.darkdaily.com/2020/06/29/multiple-studies-raise-questions-about-reliability-of-clinical-laboratory-covid-19-diagnostic-tests/. Retrieved 07 September 2021.
- ↑ 73.0 73.1 73.2 West, C.P.; Montori, V.M.; Sampathkumar, P. (2020). "COVID-19 Testing: The Threat of False-Negative Results". Mayo Clinic Proceedings 95 (6): 1127–29. doi:10.1016/j.mayocp.2020.04.004. PMC PMC7151274. PMID 32376102. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7151274.
- ↑ "HIPAA and COVID-19". Department of Health and Human Services. 11 May 2020. https://www.hhs.gov/hipaa/for-professionals/special-topics/hipaa-covid19/index.html. Retrieved 18 May 2020.
- ↑ 75.0 75.1 75.2 Centers for Disease Control and Prevention (6 August 2018). "Clinical Laboratory Improvement Amendments (CLIA): Test complexities". https://www.cdc.gov/clia/test-complexities.html. Retrieved 09 April 2020.
- ↑ "CLIA Categorizations". U.S. Food and Drug Administration. 25 February 2020. https://www.fda.gov/medical-devices/ivd-regulatory-assistance/clia-categorizations. Retrieved 09 April 2020.
- ↑ 77.0 77.1 Centers for Medicare and Medicaid Services (17 December 2020). "Frequently Asked Questions (FAQs), CLIA Guidance During the COVID-19 Emergency" (PDF). https://www.cms.gov/files/document/frequently-asked-questions-faqs-clia-guidance-during-covid-19-emergency-updated-12-17-2020.pdf. Retrieved 07 September 2021.
- ↑ Wright, D.R. (26 March 2020). "Clinical Laboratory Improvement Amendments (CLIA) Laboratory Guidance During COVID-19 Public Health Emergency" (PDF). Centers for Medicare and Medicaid Services. https://www.cms.gov/files/document/qso-20-21-clia.pdf-0. Retrieved 18 May 2020.
- ↑ "Updated CLIA SARS-CoV-2 Molecular and Antigen Point of Care Test Enforcement Discretion" (PDF). Centers for Medicare and Medicaid Services. 7 December 2020. https://www.cms.gov/files/document/clia-sars-cov-2-point-care-test-enforcement-discretion.pdf. Retrieved 07 September 2021.
- ↑ "CAP Responds to Your COVID-19 Questions". College of American Pathologists. 2021. https://www.cap.org/laboratory-improvement/news-and-updates/cap-responds-to-your-covid-19-questions. Retrieved 07 September 2021.
- ↑ 81.0 81.1 Madrigal, A.C.; Meyer, R. (31 March 2020). "Private Labs Are Fueling a New Coronavirus Testing Crisis". The Atlantic. https://www.theatlantic.com/health/archive/2020/03/next-covid-19-testing-crisis/609193/. Retrieved 07 April 2020.
- ↑ Hale, C. (17 March 2020). "FDA opens the gates to commercial coronavirus testing without agency review". FierceBiotech. https://www.fiercebiotech.com/medtech/fda-opens-gates-to-commercial-coronavirus-testing-without-agency-review. Retrieved 07 April 2020.
- ↑ 83.0 83.1 Appleby, J. (28 March 2020). "Why It Takes So Long To Get Most COVID-19 Test Results". NPR - Health Shots. https://www.npr.org/sections/health-shots/2020/03/28/822869504/why-it-takes-so-long-to-get-most-covid-19-test-results. Retrieved 07 April 2020.
- ↑ 84.0 84.1 Ryan-Mosley, T. (5 April 2020). "Why some covid-19 tests in the US take more than a week". MIT Technology Review. https://www.technologyreview.com/2020/04/05/998460/covid-19-test-results-faster-commercial-labs-delays-coronavirus/. Retrieved 07 September 2021.
- ↑ Nguyen, T.; Bang, D.D.; Wolff, A. (2020). "2019 novel coronavirus disease (COVID-19): Paving the road for rapid detection and point-of-care diagnostics". Micromachines 11 (3): 306. doi:10.3390/mi11030306. PMID 32183357.
- ↑ Yang, T.; Wang, Y.-C.; Shen, C.-F.; Cheng, C.-M. (2020). "Point-of-care RNA-based diagnostic device for COVID-19". Diagnostics 10 (3): 165. doi:10.3390/diagnostics10030165.
- ↑ Scott, D. (1 September 2021). "Why can’t America fix its Covid-19 testing problems?". Vox. https://www.vox.com/coronavirus-covid19/2021/9/1/22642745/us-covid-19-test-numbers-delta-variant. Retrieved 06 September 2021.
- ↑ Zoga, D. (3 September 2021). "NBC 5 Responds: Searching for a COVID-19 Test? You're Not Alone". NBC DFW. https://www.nbcdfw.com/news/nbc-5-responds/nbc-5-responds-searching-for-a-covid-19-test-youre-not-alone/2734134/. Retrieved 06 September 2021.
- ↑ 89.0 89.1 89.2 Centers for Medicare and Medicaid Services, Division of Clinical Laboratory and Quality (August 2021). "CLIA Update - August 2021" (PDF). https://www.cms.gov/Regulations-and-Guidance/Legislation/CLIA/Downloads/statupda.pdf. Retrieved 07 September 2021.
- ↑ Garrels, M.; Oatis, C.S. (2014). Laboratory and Diagnostic Testing in Ambulatory Care: A Guide for Healthcare Professionals (3rd ed.). Elsevier Health Sciences. pp. 368. ISBN 9780323292368. https://books.google.com/books?id=LM9sBQAAQBAJ. Retrieved 09 April 2020.
- ↑ 91.0 91.1 91.2 "In Vitro Diagnostics EUAs - Molecular Diagnostic Tests for SARS-CoV-2". U.S. Food and Drug Administration. 7 September 2021. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-molecular-diagnostic-tests-sars-cov-2. Retrieved 07 September 2021.
- ↑ U.S. Food and Drug Administration (25 March 2021). "FAQs on Diagnostic Testing for SARS-CoV-2". U.S. Food and Drug Administration. https://www.fda.gov/medical-devices/emergency-situations-medical-devices/faqs-diagnostic-testing-sars-cov-2. Retrieved 07 September 2021.
- ↑ Simonite, T. (24 March 2020). "The Professors Who Call ‘Bullshit’ on Covid-19 Misinformation". Wired. https://www.wired.com/story/professors-call-bullshit-covid-19-misinformation/. Retrieved 09 April 2020.
- ↑ Suciu, P. (8 April 2020). "During COVID-19 Pandemic It Isn't Just Fake News But Seriously Bad Misinformation That Is Spreading On Social Media". Forbes. https://www.forbes.com/sites/petersuciu/2020/04/08/during-covid-19-pandemic-it-isnt-just-fake-news-but-seriously-bad-misinformation-that-is-spreading-on-social-media/. Retrieved 09 April 2020.
- ↑ Soucheray, S. (15 July 2021). "Surgeon General warns of COVID-19 misinformation". CIDRAP. University of Minnesota. https://www.cidrap.umn.edu/news-perspective/2021/07/surgeon-general-warns-covid-19-misinformation. Retrieved 07 September 2021.
- ↑ 96.0 96.1 96.2 Foundation for Innovative New Diagnostics (27 July 2024). "SARS-CoV-2 Diagnostic Pipeline". Foundation for Innovative New Diagnostics. https://www.finddx.org/covid-19/pipeline/. Retrieved 18 November 2020.
- ↑ AdVeritasDx (2020). "The SARS-CoV-2 Test & Controls Database". https://adveritasdx.com/. Retrieved 18 November 2020.
- ↑ 98.0 98.1 Bistricean, C. (1 April 2020). "COVID-19 Testing for the Physician Office Laboratory (POL) - CovidLiMS.com". Medium. https://medium.com/@lablynx/covid-19-testing-for-the-physician-office-laboratory-pol-covidlims-com-bf1615e071ab. Retrieved 10 April 2020.
- ↑ Dickens, J.F. (3 April 2020). "Coronavirus testing: How it works – Questions answered". Reaction. https://reaction.life/coronavirus-testing-how-it-works-questions-answered/. Retrieved 10 April 2020.
- ↑ 100.0 100.1 "In Vitro Diagnostics EUAs - Serology and Other Adaptive Immune Response Tests for SARS-CoV-2". U.S. Food and Drug Administration. 3 September 2021. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-serology-and-other-adaptive-immune-response-tests-sars-cov-2. Retrieved 07 September 2021.
- ↑ Hinton, D.M. (27 March 2020). "ID NOW COVID-19" (PDF). U.S. Food and Drug Administration. https://www.fda.gov/media/136522/download. Retrieved 10 April 2020.
- ↑ Hinton, D.M. (10 June 2020). "Cue COVID-19 Test" (PDF). U.S. Food and Drug Administration. https://www.fda.gov/media/138823/download. Retrieved 08 July 2020. }}
- ↑ Hinton, D.M. (5 March 2021). "Cue COVID-19 Test for Home and Over The Counter (OTC) Use" (PDF). U.S. Food and Drug Administration. https://www.fda.gov/media/146467/download. Retrieved 07 September 2021.
- ↑ Zanoli, L.M.; Spoto, G. (2013). "Isothermal Amplification Methods for the Detection of Nucleic Acids in Microfluidic Devices". Biosensors 3 (1): 18–43. doi:10.3390/bios3010018. PMC PMC4263587. PMID 25587397. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4263587.
- ↑ "Detect COVID-19 in as Little as 5 Minutes". Abbott. 27 March 2020. https://www.abbott.com/corpnewsroom/product-and-innovation/detect-covid-19-in-as-little-as-5-minutes.html. Retrieved 10 April 2020.
- ↑ Devine, C. (3 July 2020). "Coronavirus test used by White House has questionable accuracy". CNN Politics. https://www.cnn.com/2020/07/03/politics/coronavirus-white-house-test-abbott/index.html. Retrieved 08 July 2020.
- ↑ "MAUDE - Manufacturer and User Facility Device Experience". U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfMAUDE/Search.cfm. Retrieved 07 September 2021. "Search for "ID NOW COVID-19" in Brand Name"
- ↑ Perrone, M. (14 May 2020). "FDA probes accuracy issue with Abbott’s rapid virus test". Associated Press. https://apnews.com/c8ab010e8e02dfe7beb34a5e5df11279. Retrieved 19 May 2020.
- ↑ Taylor, N.P. (7 October 2020). "Abbott, on defense, details embattled rapid COVID-19 test results". MedTechDive. https://www.medtechdive.com/news/abbott-on-defense-id-now-coronavirus-test-postmarket-study/586579/. Retrieved 18 November 2020.
- ↑ Hinton, D.M. (27 August 2021). "ID NOW COVID-19" (PDF). U.S. Food and Drug Administration. https://www.fda.gov/media/136522/download. Retrieved 07 September 2021.
- ↑ National Institutes of Health (31 July 2020). "NIH delivering new COVID-19 testing technologies to meet U.S. demand". News Releases. National Institutes of Health. https://www.nih.gov/news-events/news-releases/nih-delivering-new-covid-19-testing-technologies-meet-us-demand. Retrieved 12 August 2020.
- ↑ O'Connor, L. (11 August 2021). "Despite MDx Product Delays, Talis Biomedical Expecting 'Meaningful Revenue Ramp in 2022'". 360 Dx. Archived from the original on 11 August 2021. https://web.archive.org/web/20210811210316/https://www.360dx.com/business-news/despite-mdx-product-delays-talis-biomedical-expecting-meaningful-revenue-ramp-2022. Retrieved 07 September 2021.
- ↑ "The Talis Advantage". Talis Biomedical. https://talis.bio/technology/. Retrieved 13 August 2020.
- ↑ 114.0 114.1 Schmid-Burgk, J.L.; Li, D.; Feldman, D. et al. (2020). "LAMP-Seq: Population-Scale COVID-19 Diagnostics Using Combinatorial Barcoding". bioRxiv. doi:10.1101/2020.04.06.025635.
- ↑ Lamb, L.E.; Barolone, S.N.; Ward, E. et al. (2020). "Rapid Detection of Novel Coronavirus (COVID-19) by Reverse Transcription-Loop-Mediated Isothermal Amplification". medRxiv. doi:10.1101/2020.02.19.20025155.
- ↑ Kellner, M.J.; Ross, J.J.; Schnabl, J. et al. (2020). "A rapid, highly sensitive and open-access SARS-CoV-2 detection assay for laboratory and home testing". bioRxiv. doi:10.1101/2020.06.23.166397.
- ↑ Yu, L.; Wu, S.; Hao, X. et al. (2020). "Rapid Detection of COVID-19 Coronavirus Using a Reverse Transcriptional Loop-Mediated Isothermal Amplification (RT-LAMP) Diagnostic Platform". Clinical Chemistry 66 (7): 975–77. doi:10.1093/clinchem/hvaa102. PMC PMC7188121. PMID 32315390. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7188121.
- ↑ Park, G.-S.; Ku, K.; Baek, S.-H. et al. (2020). "Development of Reverse Transcription Loop-Mediated Isothermal Amplification Assays Targeting Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)". Journal of Molecular Diagnostics 22 (6): 729–35. doi:10.1016/j.jmoldx.2020.03.006. PMC PMC7144851. PMID 32276051. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7144851.
- ↑ Food and Drug Administration (6 November 2020). "Coronavirus (COVID-19) Update: FDA Authorizes First Test that Detects Neutralizing Antibodies from Recent or Prior SARS-CoV-2 Infection". Food and Drug Administration. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-first-test-detects-neutralizing-antibodies-recent-or. Retrieved 18 November 2020.
- ↑ "A protocol for rapid detection of the 2019 novel coronavirus SARS-CoV-2 using CRISPR diagnostics: SARS-CoV-2 DETECTR" (PDF). Mammoth Biosciences. 2 March 2020. https://mammoth.bio/wp-content/uploads/2020/03/Mammoth-Biosciences-A-protocol-for-rapid-detection-of-SARS-CoV-2-using-CRISPR-diagnostics-DETECTR.pdf. Retrieved 09 April 2020.
- ↑ "CRISPR’s powers unleashed for disease detection". Nature - Research Highlights. 16 February 2018. https://www.nature.com/articles/d41586-018-02200-0. Retrieved 09 April 2020.
- ↑ Brandsma, E.; Verhagen, H.J.M.P.; van de Laar, T.J.W. et al. (2020). "Rapid, sensitive and specific SARS coronavirus-2 detection: A multi-center comparison between standard qRT-PCR and CRISPR based DETECTR". The Journal of Infectious Diseases In Print: jiaa641. doi:10.1093/infdis/jiaa641. PMC PMC7665660. PMID 33038252. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7665660.
- ↑ "Antigen". MedlinePlus. U.S. National Library of Medicine. https://medlineplus.gov/ency/article/002224.htm. Retrieved 07 August 2020.
- ↑ Anderson, K. (6 August 2020). "5 Investigates: Concerns about current use of rapid antigen tests for COVID-19". WCVB 5 ABC. https://www.wcvb.com/article/5-investigates-concerns-about-current-use-of-rapid-antigen-tests-for-covid-19/33538332. Retrieved 07 August 2020.
- ↑ 125.0 125.1 125.2 125.3 125.4 Service, R.F. (2020). "Radical shift in COVID-19 testing needed to reopen schools and businesses, researchers say". Science. doi:10.1126/science.abe1546.
- ↑ 126.0 126.1 Kremer, R. (7 August 2020). "UW System Orders 350,000 COVID-19 Tests". Urban Milwaukee. https://urbanmilwaukee.com/2020/08/07/uw-system-orders-350000-covid-19-tests/. Retrieved 07 August 2020.
- ↑ Guglielmi, G. (2020). "The explosion of new coronavirus tests that could help to end the pandemic". Nature 583: 506–09. doi:10.1038/d41586-020-02140-8.
- ↑ 128.0 128.1 Clark, C. (6 August 2020). "COVID Antigen Tests: Coming to Case Counts Near You?". MedPage Today. https://www.medpagetoday.com/infectiousdisease/covid19/87930. Retrieved 07 August 2020.
- ↑ 129.0 129.1 129.2 Courage, K.H. (31 July 2020). "Should we be testing fewer people to stop the spread of Covid-19?". Vox. https://www.vox.com/2020/7/31/21336212/covid-19-test-results-delays. Retrieved 05 August 2020.
- ↑ 130.0 130.1 Mervosh, S.; Fernandez, M. (4 August 2020). "‘It’s Like Having No Testing’: Coronavirus Test Results Are Still Delayed". The New York Times. https://www.nytimes.com/2020/08/04/us/virus-testing-delays.html. Retrieved 05 August 2020.
- ↑ 131.0 131.1 "In Vitro Diagnostics EUAs - Antigen Diagnostic Tests for SARS-CoV-2". U.S. Food and Drug Administration. 7 September 2021. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2. Retrieved 07 September 2021.
- ↑ 132.0 132.1 Terry, M. (30 March 2020). "CRISPR Pioneer Jennifer Doudna Expects Automated COVID-19 Test Lab to Begin Testing Next Week". BioSpace. https://www.biospace.com/article/crispr-discoverer-jennifer-doudna-launches-covid-19-test-lab/. Retrieved 10 April 2020.
- ↑ 133.0 133.1 Sands, P.; Mundaca-Shah, C.; Dzau, V.J. (2016). "The Neglected Dimension of Global Security — A Framework for Countering Infectious-Disease Crises". The New England Journal of Medicine 374 (13): 1281–87. doi:10.1056/NEJMsr1600236. PMID 26761419.
- ↑ Morris, C. (26 February 2020). "Trump administration budget cuts could become a major problem as coronavirus spreads". Fortune. https://fortune.com/2020/02/26/coronavirus-covid-19-cdc-budget-cuts-us-trump/. Retrieved 10 April 2020.
- ↑ Specter, M. (17 March 2020). "The Coronavirus and the Gutting of America’s Public-Health System". The New Yorker. https://www.newyorker.com/news/daily-comment/coronavirus-and-the-gutting-of-americas-public-health-system. Retrieved 10 April 2020.
- ↑ Roos, R. (25 May 2007). "Congress cut pandemic funds before passing spending bill". CIDRAP - News & Perspective. https://www.cidrap.umn.edu/news-perspective/2007/05/congress-cut-pandemic-funds-passing-spending-bill. Retrieved 10 April 2020.
- ↑ Schnirring, L. (20 December 2007). "Congress slashes pandemic preparedness funding". CIDRAP - News & Perspective. https://www.cidrap.umn.edu/news-perspective/2007/12/congress-slashes-pandemic-preparedness-funding. Retrieved 10 April 2020.
- ↑ Gates, B. (2020). "Responding to Covid-19 — A Once-in-a-Century Pandemic?". The New England Journal of Medicine. doi:10.1056/NEJMp2003762. PMID 32109012.
- ↑ Shontell, A. (10 April 2020). "Melinda Gates: This is not a once-in-a-century pandemic. 'We will absolutely have more of these.' The billionaire philanthropist predicts a timeline for going back to normal". Business Insider. https://www.businessinsider.com/melinda-gates-coronavirus-interview-vaccine-timeline-2020-4. Retrieved 10 April 2020.
- ↑ Madhav, N.; Oppenheim, B.; Gallivan, M. et al. (2017). "Chapter 17: Pandemics: Risks, Impacts, and Mitigation". In Jamison, D.T.; Gelband, H.; Horton, S. et al.. Disease Control Priorities: Improving Health and Reducing Poverty (3rd ed.). The World Bank. ISBN 9781464805288. https://www.ncbi.nlm.nih.gov/books/NBK525302/.
- ↑ Sanders, R. (30 March 2020). "UC Berkeley scientists spin up a robotic COVID-19 testing lab". Berkeley News. https://news.berkeley.edu/2020/03/30/uc-berkeley-scientists-spin-up-a-robotic-covid-19-testing-lab/. Retrieved 10 April 2020.
- ↑ Nolting, R. (10 April 2020). "Local testing delayed by lack of reagents". Parsons Sun. https://www.parsonssun.com/news/coronavirus/article_99193646-7aec-11ea-a7c5-b700f82332db.html. Retrieved 10 April 2020.
- ↑ Herper, M.; Branswell, H. (10 March 2020). "Shortage of crucial chemicals creates new obstacle to U.S. coronavirus testing". STAT. https://www.statnews.com/2020/03/10/shortage-crucial-chemicals-us-coronavirus-testing/. Retrieved 10 April 2020.
- ↑ Hale, C. (18 March 2020). "Qiagen aims to more than quadruple its COVID-19 reagent production in 6 weeks". Fierce Biotech. https://www.fiercebiotech.com/medtech/qiagen-aims-to-more-than-quadruple-its-covid-19-reagent-production-6-weeks. Retrieved 10 April 2020.
- ↑ Mehta, A. (3 April 2020). "Mystery surrounds UK claim of Covid-19 test reagent ‘shortage’". Chemistry World. https://www.chemistryworld.com/news/mystery-surrounds-uk-claim-of-covid-19-test-reagent-shortage/4011457.article. Retrieved 07 September 2021.
- ↑ 146.0 146.1 Roche, B. (8 April 2020). "Irish scientists develop reagent in effort to ease Covid-19 testing delays". The Irish Times. https://www.irishtimes.com/news/science/irish-scientists-develop-reagent-in-effort-to-ease-covid-19-testing-delays-1.4223897. Retrieved 10 April 2020.
- ↑ Padma, T.V. (13 May 2020). "Efforts to combat Covid-19 in India hit by imported reagent shortages". Chemistry World. https://www.chemistryworld.com/news/efforts-to-combat-covid-19-in-india-hit-by-imported-reagent-shortages/4011718.article#/. Retrieved 19 May 2020.
- ↑ David, E.; Farber, S.E. (20 June 2020). "Survey shows resources for COVID-19 diagnostic testing still limited months later". ABC News. https://abcnews.go.com/Health/survey-shows-resources-covid-19-diagnostic-testing-limited/story?id=71341885. Retrieved 08 July 2020.
- ↑ Johnson, K. (2 July 2020). "NC Labs Facing Shortages In COVID-19 Testing Chemicals". Patch. https://patch.com/north-carolina/charlotte/nc-labs-facing-shortages-covid-19-testing-chemicals. Retrieved 08 July 2020.
- ↑ 150.0 150.1 American Society for Microbiology (9 November 2020). "Supply Shortages Impacting COVID-19 and Non-COVID Testing". American Society for Microbiology. https://asm.org/Articles/2020/September/Clinical-Microbiology-Supply-Shortage-Collecti-1. Retrieved 18 November 2020.
- ↑ 151.0 151.1 Abbott, B.; Krouse, S. (9 November 2020). "Covid-19 Testing Saps Supplies Needed for Other Medical Tests". The Wall Street Journal. https://www.wsj.com/articles/covid-19-testing-saps-supplies-needed-for-other-medical-tests-11604926800. Retrieved 18 November 2020.
- ↑ 152.0 152.1 Williams, S. (21 April 2021). "Supply Shortages Hit Life Science Labs Hard". The Scientist. https://www.the-scientist.com/news-opinion/supply-shortages-hit-life-science-labs-hard-68695. Retrieved 07 September 2021.
- ↑ 153.0 153.1 Hale, C. (27 April 2020). "Congress urges FDA to better police, evaluate COVID-19 antibody tests". Fierce Biotech. https://www.fiercebiotech.com/medtech/congress-urges-fda-to-better-police-evaluate-covid-19-antibody-tests. Retrieved 01 May 2020.
- ↑ Subcommittee on Economic and Consumer Policy (24 April 2020). "Preliminary Findings of the Subcommittee’s Coronavirus Antibody Testing Investigation" (PDF). U.S. House of Representatives. https://oversight.house.gov/sites/democrats.oversight.house.gov/files/ECP%20Staff%20Report%20on%20Preliminary%20Findings%20of%20the%20Subcommittee%E2%80%99s%20Coronavirus%20Antibody%20Testing%20Investigation.pdf. Retrieved 01 May 2020.
- ↑ Farley, P.; Sanders, R. (27 April 2020). "Testing the Tests: COVID-19 Antibody Assays Scrutinized for Accuracy by UCSF, UC Berkeley Researchers". UCSF News. https://www.ucsf.edu/news/2020/04/417276/testing-tests-covid-19-antibody-assays-scrutinized-accuracy-ucsf-uc-berkeley. Retrieved 01 May 2020.
- ↑ Sanders, R. (27 April 2020). "What COVID-19 antibody tests can tell us, and what they can’t". Berkeley News. https://news.berkeley.edu/2020/04/27/what-covid-19-antibody-tests-can-tell-us-and-what-they-cant/. Retrieved 01 May 2020.
- ↑ Whooley, S. (17 August 2020). "FDA warns on false results for Thermo Fisher TaqPath COVID-19 testing kit". + Mass Device. WTWH Media LLC. https://www.massdevice.com/fda-warns-on-false-results-for-thermo-fisher-taqpath-covid-19-testing-kit/. Retrieved 23 August 2020.
- ↑ Taylor, N.P. (18 August 2020). "Thermo Fisher COVID-19 test flagged for false positive and negative results". MedTechDive. https://www.medtechdive.com/news/thermo-fisher-covid-19-test-flagged-for-false-positive-false-negative-resu/583682/. Retrieved 23 August 2020.
- ↑ Richardson, L. (2 September 2021). "With Delta Surging, FDA Needs Authority to Ensure Accuracy of COVID-19 Tests". Pew Charitable Trust. https://www.pewtrusts.org/en/research-and-analysis/articles/2021/09/02/with-delta-surging-fda-needs-authority-to-ensure-accuracy-of-covid19-tests. Retrieved 07 September 2021.
- ↑ 160.0 160.1 160.2 Wan, W. (24 October 2020). "Home tests could help in the fight against the coronavirus. So where are they?". The Washington Post. https://www.washingtonpost.com/health/2020/10/24/coronavirus-home-tests/. Retrieved 18 November 2020.
- ↑ McDermott, A. (2020). "Inner Workings: Researchers race to develop in-home testing for COVID-19, a potential game changer". PNAS 117 (42): 25956-25959. doi:10.1073/pnas.2019062117. PMC PMC7584891. PMID 32999063. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7584891.
- ↑ Crear-Perry, J. (5 November 2020). "The hidden public health hazard of rapid Covid-19 tests". STAT. https://www.statnews.com/2020/11/05/rapid-covid-19-tests-hidden-public-health-hazard/. Retrieved 18 November 2020.
- ↑ National Institutes of Health (23 June 2020). "Experts identify steps to expand and improve antibody tests in COVID-19 response". News Releases. https://www.nih.gov/news-events/news-releases/experts-identify-steps-expand-improve-antibody-tests-covid-19-response. Retrieved 08 July 2020.
Citation information for this chapter
Chapter: 2. Diagnostic testing of COVID-19 and other coronaviruses
Edition: Fall 2021
Title: COVID-19 Testing, Reporting, and Information Management in the Laboratory
Author for citation: Shawn E. Douglas
License for content: Creative Commons Attribution-ShareAlike 4.0 International
Publication date: September 2021













