Journal:Differentiating cannabis products: Drugs, food, and supplements
| Full article title | Differentiating cannabis products: Drugs, food, and supplements |
|---|---|
| Journal | Frontiers in Pharmacology |
| Author(s) | Salehi, Arash; Puchalski, Keely; Shokoohinia, Yalda; Zolfaghari, Behzad; Asgary, Sedigheh |
| Author affiliation(s) | Isfahan University of Medical Sciences, Southwest College of Naturopathic Medicine |
| Primary contact | Email: sedighehasgary at gmail dot com |
| Editors | Lupica, Carl R. |
| Year published | 2022 |
| Volume and issue | 13 |
| Article # | 906038 |
| DOI | 10.3389/fphar.2022.906038 |
| ISSN | 1663-9812 |
| Distribution license | Creative Commons Attribution 4.0 International |
| Website | https://www.frontiersin.org/articles/10.3389/fphar.2022.906038/full |
| Download | https://www.frontiersin.org/articles/10.3389/fphar.2022.906038/pdf (PDF) |
Abstract
"Hemp" refers to non-intoxicating, low delta-9 tetrahydrocannabinol (Δ9-THC) cultivars of Cannabis sativa L. "Marijuana" refers to cultivars with high levels of Δ9-THC, the primary psychoactive cannabinoid found in the plant and a federally controlled substance used for both recreational and therapeutic purposes. Although marijuana and hemp belong to the same genus and species, they differ in terms of chemical and genetic composition, production practices, product uses, and regulatory status. Hemp seed and hemp seed oil have been shown to have valuable nutritional capacity. Cannabidiol (CBD), a non-intoxicating phytocannabinoid with a wide therapeutic index and acceptable side effect profile, has demonstrated high medicinal potential in some conditions. Several countries and states have facilitated the use of THC-dominant medical cannabis for certain conditions, while other countries continue to ban all forms of cannabis regardless of cannabinoid profile or low psychoactive potential.
Today, differentiating between hemp and marijuana in the laboratory is no longer a difficult process. Certain thin-layer chromatography (TLC) methods can rapidly screen for cannabinoids, and several gas and liquid chromatography techniques have been developed for precise quantification of phytocannabinoids in plant extracts and biological samples. Geographic regulations and testing guidelines for cannabis continue to evolve. As they are improved and clarified, we can better employ the appropriate applications of this uniquely versatile plant from an informed scientific perspective.
Keywords: cannabis, cannabidiol, CBD, hemp, marijuana, tetrahydrocannabinol, THC
Introduction
Cannabis sativa L. is a member of the Cannabaceae family, a small family of annual herbaceous plants that includes the widely cultivated genus, Humulus (hops), and eight other genera.[1] The terms "hemp" and "marijuana" both refer to plants derived from the Cannabis sativa L. species, but from different cultivars or chemotypes having either low or high delta-9 tetrahydrocannabinol (Δ9-THC) content, respectively. The general term "cannabis" includes both hemp and marijuana types of plants.[2] Cannabis has been utilized by several populations for millennia. Central and southeast Asia are considered plausible origins.[3] It has been claimed that the application of the herb for various purposes dates to 12,000 years ago based on Neolithic evidence found in Taiwan.[4] Hemp is reputed to be the oldest cultivated fiber plant[5], and hemp seed and seed oil have historically been used as food.[6] Marijuana has long been utilized for recreational as well as medical purposes.[7] The oldest report of the medical use of cannabis dates to 5,000 years ago when it was used for the treatment of fatigue, rheumatism, and malaria.[8] The Assyrians, Egyptians, Indians, Persians, Greeks, and Romans are all reported to have used cannabis for medical purposes.[9]
Classifications for different cannabis species continues to be a highly debated topic amongst taxonomists and botanists.[10] Although some experts recognize three different species of Cannabis—C. sativa, C. indica, and C. ruderalis[11]—and other experts only one monospecific species (C. sativa L.) with two subspecies (subsp. sativa and subsp. indica [Lam.])[12][13], neither of these nomenclature systems accurately reflects the diversity and complexity of modern cannabis plants. Because cannabis species have been cultivated globally over many years to exhibit nearly indistinguishable phenotypes with overlapping genotypes, Cannabis sativa L. varieties are now most accurately identified by cultivar or chemotype, including specific cannabinoid profiling, and even subtyping.[14] (Recently proposed suggestions for chemotyping will be discussed later in this article.)
Although it has valuable nutritional capacity, hemp material was initially cultivated mainly to produce textiles and ropes. Due to expanded applications of synthetic fibers over the past two centuries, its cultivation decreased precipitously. In recent years, hemp regained popularity due to the rediscovery of its nutritional benefits, economic value, and variety of medicinal uses.[6] Despite the resurgence of hemp, ongoing efforts to curtail recreational drug use have prompted several countries and states to restrict the cultivation of cannabis varieties with high concentrations of Δ9-THC, the primary psychoactive metabolite of the plant.[15]
For the past several decades, a lack of understanding and proper differentiation between hemp- and marijuana-type plants has slowed the development of cannabis research on the potential health benefits of the plant.[6] And despite having clearer definitions, improper use of these terms is still regularly seen in the literature. It is pertinent to clarify terminology and provide useful tools and legal definitions to differentiate between food, drug, and supplement derivatives of cannabis.
In this review we focus mainly on the question, “What is essential for defining Cannabis as a food, supplement, or drug?” We also investigate the nutritional potential of hempseed and seed oil, the medicinal benefits of certain phytocannabinoids (one of the predominant phytochemical groups in Cannabis) as drugs and supplements, and the regulatory status of cannabis and cannabis products across the globe. Finally, we review several analytical techniques for the detection and quantification of cannabinoids in cannabis extracts, samples, and other products.
Definitions, production practices, and uses
Cannabis terminology has been a significant source of confusion for many. In public opinion at large, marijuana is often viewed as a plant purposed solely for recreational use, which has overshadowed the high value of medical cannabis, hemp-type plants, and hemp products in nutrition, the health and wellness industry, scientific and medical communities, and the global economy.[16] Because of this, it is increasingly important to implement clear literature-based nomenclature, legal definitions, and recommendations of agencies specializing in this controversial plant. Here we explore some of the current definitions and nomenclature.
Fundamentally, "marijuana" refers to cultivars of cannabis with psychoactive potential used for both recreational and therapeutic purposes, and "hemp" refers to non-intoxicating cultivars of cannabis with various end uses, including in fabrics and textiles, nutraceuticals, pharmaceuticals, food products, beverages, oral and topical self-care products, veterinary products, and other manufactured and industrial goods. The term "industrial hemp," in some cases, may be used interchangeably with "hemp."[2][17]
With the passage of the Agriculture Improvement Act of 2018 (i.e., the 2018 Farm Bill), hemp is now legally defined as "the plant species Cannabis sativa L. and any part of that plant, including the seeds thereof and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a total delta-9 tetrahydrocannabinol concentration of not more than 0.3 percent on a dry weight basis."[18] Meanwhile, the Controlled Substances Act of 1970 (CSA) still broadly describes marijuana without using any limits on Δ9-THC or any other cannabinoid, defined as "all parts of the plant Cannabis sativa L., whether growing or not; the seeds thereof; the resin extracted from any part of such plant; and every compound, manufacture, salt, derivative, mixture, or preparation of such plant, its seeds, or resin."[19] Noticeably, this definition of marijuana excludes the mature stalks of Cannabis sativa L. and products derived from the stalks (e.g., fiber), as well as sterilized seeds incapable of germination, and any certain preparations of viable seeds, such as oil or cake, provided those preparations do not contain resin.
The enactment of the 2018 Farm Bill effectively removed hemp from the legal definition of marijuana originally set forth in the CSA in 1970. The CSA had classified cannabis, including hemp, marijuana, and all associated cannabinoids, as a Schedule I controlled substance. The removal of hemp from controlled substance status allowed for agricultural cultivation of hemp plants on U.S. soil as well as greater federal oversight of hemp in commerce, including commodities like food and supplements. Although the separation of hemp from marijuana allowed significantly more freedom for hemp growers and product manufacturers—as well as researchers, clinicians, and consumers—the change also led to some new aspects of confusion, specifically surrounding use of the isolated compound cannabidiol (CBD). (We will later explore the nuances of CBD applications in food, pharmaceuticals, and supplements.)
The terms "hemp" and "marijuana" are both affiliated with lengthy cultural and political histories, and in the case of marijuana, polarizing ones. Today, these terms are no longer fully adequate to describe the numerous varieties and phytochemical complexities found in modern day cannabis, nor accurately describe their potential applications. In attempts to improve existing cannabis nomenclature, the United States Pharmacopeia (USP) Cannabis Expert Panel (CEP) suggested in 2020 that due to cannabis’s complex secondary metabolome and highly variable distribution of chemical constituents, three broad categories should be adopted for classifying cannabis based on phytocannabinoid chemotype: 1) THC-dominant chemotype; 2) intermediate chemotype with both THC and CBD; and 3) CBD-dominant chemotype.[14] They also proposed that these categories could be further subcategorized by other cannabinoids and/or mono- and sesquiterpene profiles as we continue to learn more about the therapeutic potential of these other compounds in humans.[14]
Hemp and marijuana plants generally share a common genetic trait pool with some predictable functional genetic variations; however, distinct genetic variations have also been observed amongst cannabis cultivars (particularly hemp cultivars) spanning the entire genome, not just genes related to THC or cannabinoid synthesis.[10] Single nucleotide variant analysis demonstrates separation between THC and CBD.[20] In most cases, the quantitative ratio of the phytochemicals tetrahydrocannabinolic acid (THCA) and cannabidiolic acid (CBDA) (THCA:CBDA) may effectively be used for differentiating cannabis cultivars as either THC- or CBD-dominant (or intermediate), as THCA:CBDA ratios strongly reflect underlying genetic nuances in cannabinoid synthase enzymes (THCA and CBDA synthases) and their sequencing, loci distribution, patterns of expression (functional vs. non-functional), and affinities for cannabigerolic acid (CBGA), their shared substrate.[21] Weiblen et al.[21] also proposed that all three types of cannabis chemotypes (THC-dominant, CBD-dominant, or intermediate) likely contain multiple linked loci with both THCA and CBDA synthase enzymes; however, identifying the presence of a non-functional CBDA synthase may directly indicate THC-dominance in a plant, as this non-functional enzyme has been selected for over many years where marijuana cultivation has thrived. This novel finding, if proven reliable as an identification method, would allow for rapid screening of THC- vs. CBD-dominant plant material at the genetic level prior to any plant cultivation.
Based on the genetic biosynthetic pathways of THC and CBD, Fernandez et al.[22] were able to report a systematic genetic prediction of Cannabis chemotypes of 62 European agricultural hemp cultivars. They suggested three main chemotype groupings for Cannabis sativa L., similar to the chemotypes proposed by the USP CEP[22]:
- Chemotype I, or THC-dominant varieties, in which the ratio of CBD/THC is low (0.00–0.05) and characterized by a THC level >0.3% (in the U.S.; dry weight of reproductive parts in the female plant at flowering time)
- Chemotype II, or THC-intermediate types, which have both THC and CBD in a content ratio (CBD/THC) of about 0.5–3.0
- Chemotype III, or CBD-predominant varieties, in which the ratio of CBD/THC is 15–25 and, in the U.S., contains <0.3% THC concentration (dry weight of reproductive part in female plants at flowering time)
These novel nomenclature systems eliminate much of the confusion surrounding hemp and marijuana, and they provide a destigmatized, more objective way of discussing Cannabis for certain applications. These classifications are particularly helpful when discussing the female inflorescence material of Cannabis plants, which is cultivated for many different chemotypes and cannabinoid concentrations (both THC- and CBD-dominant plants) for therapeutic use. For clarity, we will continue to use the three USP categories suggested above (THC-dominant, THC-intermediate, and CBD-dominant), where applicable, for the rest of this article.
In addition to the many chemotypes of plant material, there are also differences in production practices applied to cannabis. Based on different applications, hemp is generally cultivated for three different crops: fiber, seed, and CBD-dominant inflorescence. In contrast, THC-dominant plants are grown solely for their inflorescence for both medicinal and recreational use, as the inflorescence contains the most phytocannabinoids (i.e., Δ9-THC).[2] Generally, all phytocannabinoids in cannabis exist in resin from the secretory glandular trichomes on the flowering tops of female plants. The numbers of these trichomes are fewer in male plants.[23] For this reason, female flowers are more valuable for phytocannabinoid production, while male plants are more appropriate for fiber production. Cannabis growers cultivating female plants for their phytocannabinoid production (generally CBD-, THC-, or CBD/THC co-dominant chemotypes) usually remove all male plants to prevent pollinating and seed yielding. Conversely, only male plants are grown to produce hemp fiber and seeds. Some hemp farmers producing fiber and seeds try to prevent flowering to encourage taller stalk growth and less branching[2], whereas those cultivating hemp for CBD will promote growth of the plant’s phytocannabinoid-rich flowering tops.
Historically, there were significant visual differences between hemp and marijuana plants. Modern cultivated plants appear more phenotypically similar when grown for cannabinoid production, but some distinctions can still be made. Cultivated hemp for fiber and seed is usually characterized by a single tall stalk with few leaves and branches due to high-density cultivation in order to prevent branching, measuring up to 4.5 meters tall. In contrast, the flowering, phytocannabinoid-rich Cannabis plants (i.e., THC-dominant, THC-intermediate, or CBD-dominant chemotypes) are usually characterized by short, tightly clustered, bushy, low-density cultivation with many leaves and branches (in order to develop buds and flowers), and are only 1.2 meters tall.[2]
Chemical and genetic composition
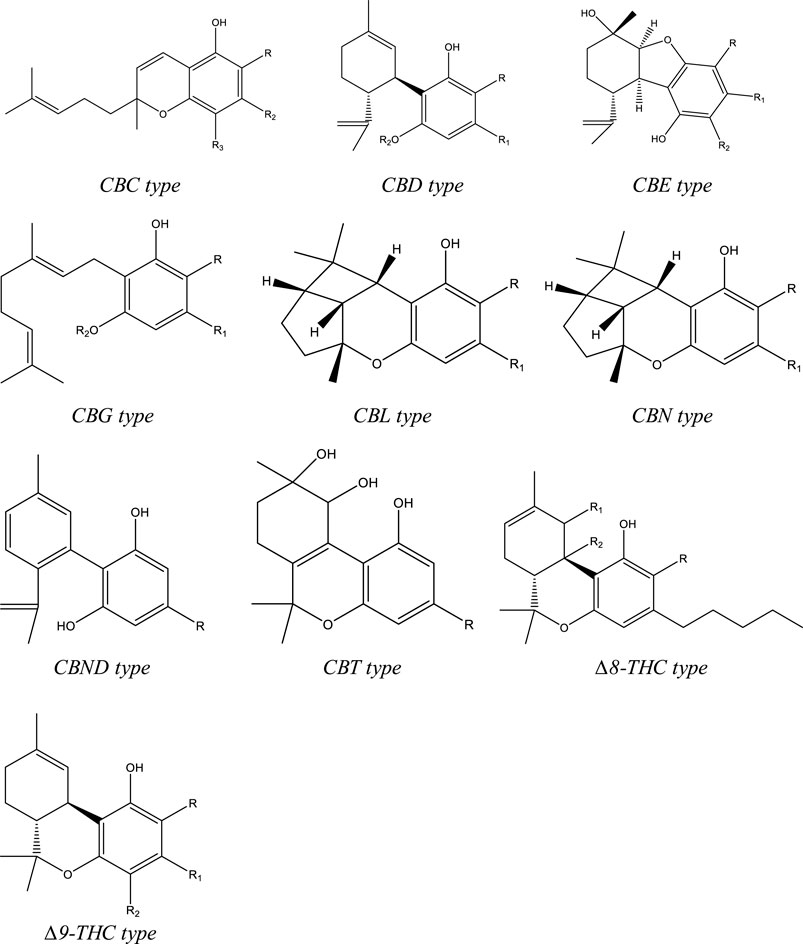
To date, more than 500 compounds have been identified from Cannabis sativa L., out of which 125 compounds are phytocannabinoids. Structurally these compounds have a C-21 terpenophenolic backbone consisting of an alkyl resorcinol with a monoterpene moiety. Eleven types are described for these cannabinoids based on their structure: Δ9-THC, CBD, (−)-Δ8-trans-tetrahydrocannabinol (Δ8-THC), cannabigerol (CBG), cannabinol (CBN), cannabichromene (CBC), cannabielsoin (CBE), cannabicyclol (CBL), cannabinodiol (CBND), cannabitriol (CBT), and miscellaneous-type cannabinoids (Figure 1). Among the phytocannabinoids, 25 of them are reported to be classified as Δ9-THC types and 10 have been elucidated as CBD types.[24][25] Apart from the cannabinoids, four major categories of non-cannabinoid-type compounds are present, including alkaloids, flavonoids, non-cannabinoid phenols, and terpenes.[25]
|
Although marijuana is technically defined as having greater than just 0.3% Δ9-THC concentration by dry weight in the U.S., today these plants are often bred to have Δ9-THC levels averaging as high as 10–30%.[17] Modern hemp plants cultivated for CBD production range anywhere from 5–15% total CBD.[26] The number of cannabis plants cultivated for high CBD:THC ratios has grown exponentially in the past decade. Some researchers are finding that specific CBD-dominant chemovars have a CBDA synthase (CBDAS) gene that synthesizes not only CBDA, the precursor of CBD, but also synthesizes some THCA as a side product.[26] With accumulation of CBD, where CBDAS is present, there may be concomitant accumulation of THC that exceeds most legal thresholds prior to the time that the plant accumulates the desired 10% or more total CBD.[26] This illustrates the importance of chemovar selection and genetic testing prior to large scale cultivation.
Phytocannabinoids are the most abundant constituents in cannabis. Isoprenoids, also called terpenoids, are the second most abundant constituents, numbering over 200 total terpenes.[25][27] Typically found in cannabis trichomes at about 10%, and in flowers at about 1%, some chemotypes have been bred to contain as much as 3.5% terpene content or higher in the flowers.[28][29] Although relatively modest in concentration to the cannabinoids, terpenes (mainly mono- and sesquiterpenes) are another important class of compounds. Terpenes are of increasing interest to researchers, healthcare practitioners, and the natural products industry as both isolated therapeutic agents and as enhancing compounds in holistic cannabis formulas, designed to engage what has recently been referred to as the entourage effect.[30] Variation in terpene content is likely responsible for the more sedative vs. stimulating effects of different cannabis cultivars, a notion that had historically been attributed to indica vs. sativa varieties.
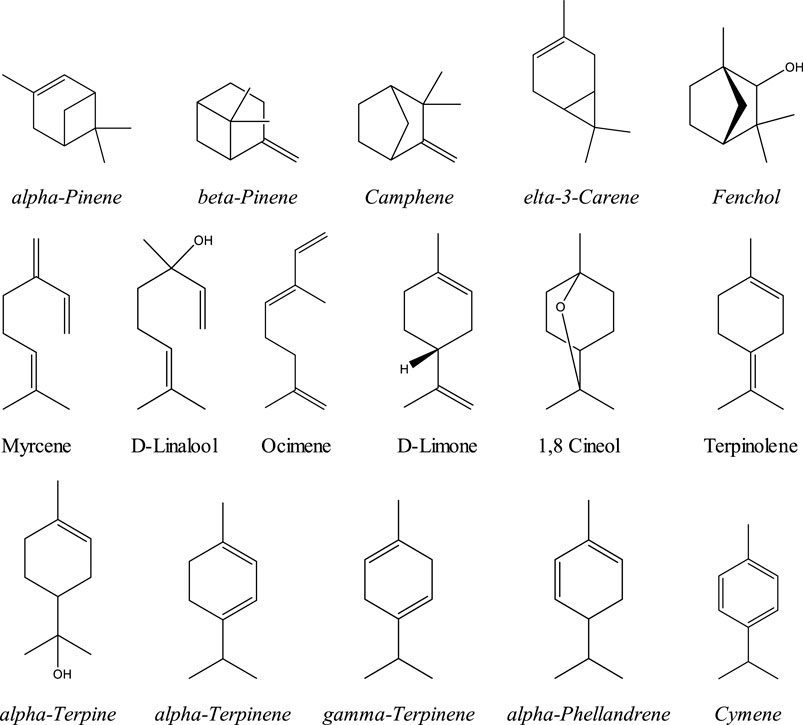
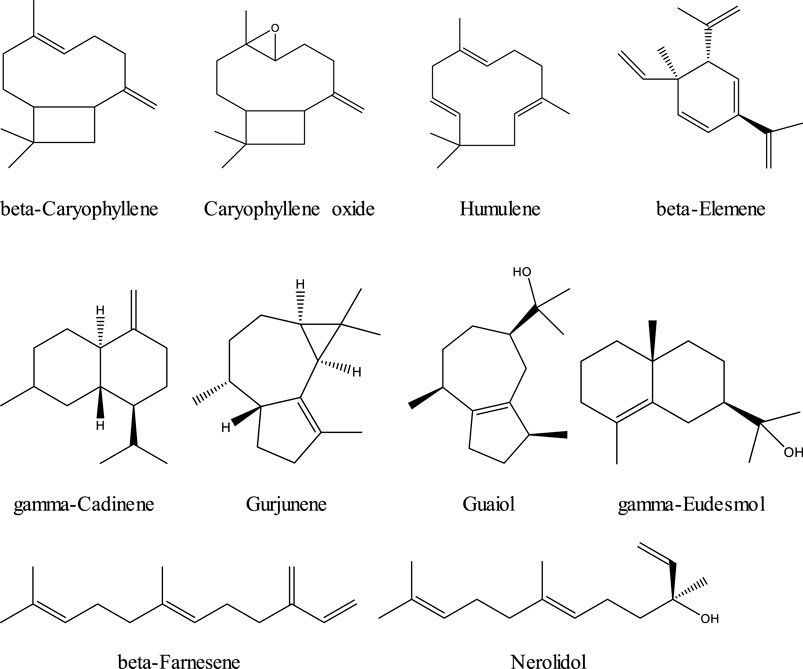
Terpenes are a family of organic compounds which are biosynthetically produced by isoprene units found mainly in plants. Certain cannabis terpenes have been identified and studied in plants such as tea tree, lavender, thyme, basil, Pinus sp., frankincense, and citrus fruits like lemon and mandarin.[31][32] Monoterpenes and sesquiterpenes of cannabis (the volatile compounds) mainly exist in the essential oil, which is isolated by hydrodistillation.[25] Among terpenes, the monoterpene β-myrcene is believed to have the highest prevalence in modern cannabis chemovars in the U.S.[31] The 16 commonly encountered monoterpenes from three classes (acyclic, monocyclic, and bicyclic monoterpenes) are shown in Figure 2. The most common terpene that exists in cannabis extracts is the sesquiterpene β-caryophyllene. Sesquiterpenes have a 15-carbon skeleton. The most abundant sesquiterpenes of cannabis are shown in Figure 3.[31] However, other sources cite additional terpenes as most predominant based on samples from more specific geographic locations.[25][33]
|
|
Pharmacological effects
Cannabis is known to affect nearly every system in the human body, including but not limited to the central and peripheral nervous systems, as well as the endocrine, immune, gastrointestinal, and musculoskeletal systems. Thousands of articles and over 100 clinical studies have been published on the pharmacodynamics and bioactive effects of cannabis on human psychology, appetite, cognition, sleep, and pain.[31] Only in this century are we are beginning to understand the complexities of these pharmacological actions and interactions, which are largely due to the actions of cannabis on the human endocannabinoid system (ECS) and the “entourage effect” or bioactive synergisms between phytocannabinoids and other compounds within the plant.
The largest body of cannabis research prior to the past two or three decades focused primarily on the Δ9-THC as a medicinal and recreational drug and emphasized its psychoactive attributes and safety profile. In recent years, CBD has garnered more substantial research interest as a non-intoxicating but equally potent therapeutic compound. Structurally, Δ9-THC was elucidated in 1964 as the main psychoactive compound in cannabis. In 1988, Huestis et al. discovered that THC exerted its psychoactive effects through agonizing cannabinoid receptor 1 (CBR1).[34] CBR1 is a G protein coupled receptor (GPCR) distributed widely throughout the central nervous system (CNS). A few years later, Munro et al. identified a second cannabinoid receptor, CBR2, that also exists in the CNS (in lower concentrations than CB1R), but in greater concentrations in the peripheral nervous system (PNS) and immune system.[34] Two endogenous ligands (endocannabinoids) were later discovered to bind to these cannabinoid receptors: N-arachidonoyl ethanolamide (AEA) and 2-arachidonoyl glycerol (2-AG).[34] AEA and 2-AG are physiologic lipid-based retrograde neurotransmitters that interact with the ECS receptors and proteins in a similar manner to exogenous phytocannabinoids THC and CBD.
The pathways by which exogenous cannabinoids such as Δ9-THC and CBD interact with the ECS are complex and only partially understood. Definitively, THC has been shown to directly bind CB1 and CB2 receptors, exerting psychoactive and other neurologically-mediated effects. CBD does not directly bind CB1 or CB2 receptors, exhibiting a low affinity for both, but can antagonize synthetic agonists of cannabinoid receptors, suggesting a negative allosteric modulatory effect.[35][36] Although CBD does not agonize CBR1 or CBR2, it exhibits several cannabinoid receptor-independent activities, including direct agonism of TRPV1 and 5-HT1A receptors, enhancement of adenosine receptor signaling, upregulation of peroxisome proliferator activated receptor gamma (PPARγ) protein expression, and suppression of GPR55 receptor activity.[31] CBD is non-intoxicating with a wide therapeutic index and acceptable side effect profile, and has pre-clinically exerted antipsychotic, intestinal anti-prokinetic, neuroprotective, anti-proliferative, anti-ischemic, vasorelaxant, analgesic, anxiolytic, anti-inflammatory, and antiepileptic effects.[37][38] CBD also has been shown to negate the unpleasant side effects of THC—including anxiety, psychosis, tachycardia, and drowsiness—through negative allosteric modulation of CB1 receptors.[39]
Other prominent cannabinoids of pharmaceutical interest include CBG, CBC, CBN, tetrahydrocannabivarin (THCV), cannabidivarin (CBDV), and the acidic cannabinoid precursors THCA and CBDA. Most of these cannabinoids are still being investigated in preliminary in vitro and animal model research studies; however, some mechanisms of action have been elucidated and reported in the literature. CBG, for example, has weak affinity for CB1 and CB2 receptors, but is a potent α2-adrenoreceptor agonist, an efficient serotonin 5-HT1A receptor antagonist, and might activate a number of other dominant receptors involved in heat/cold sensitization, pain, and inflammation via antagonism of TRPV8 receptors and stimulation of TRPV1, TRPV2, TRPA1, TRPV3, and TRPV4.[40][41][42][43] CBC can have profound therapeutic effects on inflammation and pain through CB2 receptor activity and stimulation and desensitization of TRP ankyrin-type 1 (TRPA1) cation channels, interactions with TRPV3 and TRPV4 cation channels, and desensitization of TRPV2 and TRPV4 channels.[44][45] CBC has also been shown to relieve pain in mice by augmenting the analgesic effects of THC when the two are co-administered[45][46][47], and it can increase concentrations and prolong the duration of endocannabinoids like anandamide through its interactions with TRP channels.[42][44][48] CBN is being investigated for several topical therapeutic targets for skin conditions like psoriasis, burns, and MRSA infections, via inhibition of keratinocyte proliferation, TRPV2 agonism, and direct antimicrobial activity, respectively.[49][50][51][52] Additionally, CBN is an important marker compound of THC degradation in plant material, an artifact that occurs naturally with storage and may be expedited by high heat exposure as the oxidation byproduct of THC.[53]
THCV, a propyl analogue of THC, exhibits concentration-dependent activity at CB1 receptors and may perform as an agonist or act as an antagonist, accordingly.[54] THCV demonstrates anticonvulsant attributes in mouse pyriform cortex and cerebellum[55], and it may promote weight loss and other cardiometabolic benefits.[56] CBDV has been reported to have significant anticonvulsant activity and is potentially of equal therapeutic value to CBD in treating epilepsy, especially focal/partial onset seizures.[57] CBDV also activates or blocks a wide range of cation channels, depending on the concentration and experimental conditions, including TRPA1, TRPM8, and TRPV channels 1–4. Of additional importance, CBDV inhibits the cellular uptake of anandamide and inhibits endocannabinoid degradation through modulating enzymes diacylglycerol lipase and N-acylethanolamine-hydrolyzing acid amidase (NAAA).[58]
THCA is the acidic precursor of THC, representing up to 90% of total THC in the plant prior to prolonged storage, UV exposure, or heat.[59] Like THC, THCA is an agonist of CB1 and CB2 receptors, but there is discrepancy as to which (THC or THCA) has greater affinity.[60][61][62] Although several cannabinoids have been shown to deposit in brain tissue[63][64], THCA seems to have limited access to the CNS or ability to cross the blood brain barrier (BBB), likely due to its carboxylic acid group.[59] This means it also does not elicit any psychoactivity. Preliminary research suggests THCA exhibits antineoplastic, neuroprotective, and anti-inflammatory activities via modulation of COX pathways, TRP cation channels, and other immunomodulatory and cell signaling pathways. CBDA, the acidic precursor to CBD, shares the same enhancing activity of CBD at 5-HT1A receptors, even up to 10 times higher than the activity of CBD, but does not show agonism or antagonism at CB1 receptors.[65][66] CBDA can also inhibit COX1 and COX2, and, at low concentrations (between 1 and 10 μM), targets GPR55, TRPA1, TRPV1, and TRPM8.[67]
In terms of pharmacological effects of cannabis, where there is strong support for cannabinoids, there is an equal amount of hesitancy in embracing the therapeutic effects of terpenoids. Although low in concentration in plant material and final cannabis preparations, several aromatic terpenes, especially the monoterpenoids, have been shown to have remarkably potent physiologic effects when administered in humans and animals via inhalation in small amounts.[68][69][70] Terpenes are lipophilic, allowing them to penetrate the skin and the blood brain barrier.[71] The subsequent activity of terpenes in the CNS seems to be part pharmacological and part psychological, where the psychological effects actually predominate when fragrances are inhaled and the sense of smell is stimulated via G-protein coupled (odorant) receptors.[72] In addition, when scent processing of odor information is prevented, pharmacological effects that might be overridden when fragrances are inhaled become more evident.[72] Though most often studied for inducing stimulant or sedative psychoactive effects on the nervous system, terpenes also exhibit marked pharmacological versatility outside of the nervous system. Some additional biological effects are related to their antioxidant, antiviral, antinociceptive, antiphlogistic (anti-inflammatory), and drug penetration enhancing effects.[72] These effects are mediated by neuronal and muscle cell ion channels, interactions with cell membranes, second messenger systems, and enzymes.[72][73] B-myrcene, one of the most prevalent terpenes found in all nearly all cannabis plants, increases CB1 receptor saturation, thereby enhancing the psychoactive effects of THC.[74] Linalool—a terpene also found in mint, lavender, and cinnamon—may increase serotonin and dopamine levels in the brain.[74] a-Pinene binds GABAA-Benzodiazepine receptors (GABAA-BZD) in the brains of rat models, acting as a partial modulator, and it may also display acetylcholinesterase inhibitor activity, either alone or in combination with other terpenes commonly found in the same plants.[75] β-Caryophyllene is a potent selective agonist for CB1 receptors which, synergistically, could potentiate the antipruritic and gastro-protective activities of THC and the anti-inflammatory effects of CBD.[31][76]
Finally, hemp food products can also induce some important pharmacological effects. Phytol, a major diterpene from hemp seed oil, has been shown to have anticancer and antioxidant activities.[77] Several studies suggest antihypertensive effects of hydrolyzed hemp seed proteins due to their inhibitory effects on angiotensin-converting enzyme and renin.[78] There is also a patented hemp seed flour which was claimed to have an HDL boosting effect and may be beneficial in preventing cardiovascular disease.[79]
Food derivatives
Many food products can be derived from hemp, most of which are made from hemp seed. Common hemp seed food products include bulk or packaged raw hulled hemp seeds, hemp seed oil, hemp protein concentrates or powders, and hemp milk, among others. The use of hemp in food products has continued to increase in popularity since the 2018 Farm Bill removed hemp from the CSA. The bill also separated hemp from marijuana and allowed for the interstate transportation of seeds, plants, and processed hemp products.
As previously stated, hemp refers to Cannabis sativa with less than 0.3% THC by dry weight. For legal and safety reasons, the level of THC must also be determined in final hemp products and included in the label.[80] Oral intake of as low as 2.5 mg THC per day can cause intoxicating effects in adults.[81] Germany's Federal Institute for Consumer Health Protection and Veterinary Medicine (BgVV) recommended a maximum limit of 1–2 μg/kg THC per day.[82] Consumption of higher doses in food products should be avoided due to health threatening consequences. To be precautionary, it is important to consider variations in an individual’s sensitivity, drug metabolism, and food-drug interactions. It should also be noted that hemp ingredients from plant parts other than the seeds (including the flower) are not allowed as food ingredients. Non-seed ingredients are prohibited regardless of whether they are added to the food item by the grower/producer, manufacturer, retailer, or by the consumer.
Hemp seeds are a highly nutritious food. Due to the high nutritional value of hemp seed, it could be a complete food source for mankind. Each 100 grams of hemp seed provides 500–600 Kcal energy.[83] Hemp seed usually contains 25–35% lipids, 20–25% proteins, and 20–30% carbohydrates, as well as vitamins (thiamine, riboflavin, pyridoxine, vitamin E and C), minerals (mainly magnesium and iron), flavonoids, tocopherols (alpha, beta, gamma, and delta tocopherols), terpenes, phytosterols, and bioactive peptides.[84]
A very low but detectable amount of THC is present in hemp seeds, which mainly exists on the seed surface and seems to be a contaminant from glandular trichomes at harvest time. Cultivating and handling processes should be done in such a way that prevents contamination with high levels of THC.[81] Like THC, CBD is also found in relatively low amounts in the seed oil.[85]
Hemp seed oil includes a large percentage of polyunsaturated fatty acids (PUFA) (72–83%) primarily consisting of linoleic (omega-6) and linolenic (omega-3) acids. The ratio of omega-6:omega-3 in the oil is 3:1, which explains its protective potential against cardiovascular disease, osteoporosis, and eczema.[80][86] From a nutritive point of view, hemp seed oil is much richer than soybean oil.[17] Human daily nutritional needs of fats can be provided by only three tablespoons of oil.[85] Furthermore, hemp seed oil shows a great antioxidant potential. Tocopherol isomers which exist to a large extent in the oil, as well as terpenes and polyphenols (mainly flavonoids), are responsible for this activity.[80][87][88] Hemp seed oil also contains a noteworthy amount of phytosterols. It has been noted that plant sterols and stanols exert low-density lipoprotein (LDL) lowering effects in humans.[89]
It is important not to heat the seeds during oil extraction. Unrefined and cold pressed oil is preferred. Cooking the oil produces toxic trans-fatty acids and should be avoided. In addition, unsaturated oil is vulnerable to oxidation, therefore the oil has a very short shelf life and must be protected from air, heat, and light.[17]
Hemp seed protein is abundant in edestin and albumin, which makes it easily digestible, and provides a beneficial supply of essential amino acids, particularly arginine and glutamic acid.[90][91] Hemp seed flour is free from gluten and is not known to have any known allergens, hence it does not cause any problem in individuals with celiac disease.[86]
Of the many nutritious hemp-derived food products, consumption of hemp seed oil is the most popular. In addition to the more common hemp seed oil, flour, and protein powder, the market of hemp food products continues to expand and now includes a range of novel products from sauces, butter, pasta, and cheese, to energy bars, chocolate, ice cream, and sweets.[17][92]
It should be noted that CBD, the cannabinoid typically found in highest concentration in the inflorescence of hemp plants cultivated for medicinal use, is naturally present, along with other cannabinoids and phytochemicals, in hemp products made from Cannabis inflorescence. There is, however, little to no CBD naturally found in hemp food products like seeds or seed oil, flour, or protein. With rising interest in the beneficial therapeutic effects of CBD and the passage of the 2018 Farm Bill, many retailers began selling CBD-infused foods, beverages, and personal care items, presuming CBD also fell under hemp-based food and supplement regulations. However, CBD was approved as the active ingredient in a seizure medication called Epidiolex in June of 2018. As the active ingredient in a pharmaceutical drug, CBD cannot legally be used as an additive in food or supplements in the U.S. The European Commission, however, has put CBD-containing supplements in the novel foods category.[93] CBD-dominant full-spectrum or broad-spectrum hemp products (<0.3% THC) with naturally occurring cannabinoids, including CBD, are still able to be consumed as food or supplements, but using CBD in any other context is not allowed. CBD and CBD-infused products have remained in the market despite FDA claims of the illegality of CBD supplements, and consumers should be aware of these distinctions. Daily intake of 1–2 mg/kg CBD is implausible to be unsafe[38]; however, after identifying concerns of hepatotoxicity, this limitation was done to reduce potential life-threatening adverse effects and prevent serious drug or supplement interactions.[94]
Pharmaceutical drug derivatives
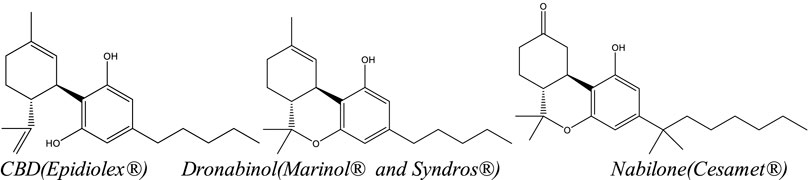
The U.S. Food and Drug Administration (FDA) has approved four cannabis-related pharmaceutical products to date: one natural, Cannabis-derived drug product—Epidiolex—which contains highly purified CBD, and three synthetic THC-related products—Marinol (dronabinol), Syndros (dronabinol), and Cesamet (nabilone) (Figure 4). Although both drugs containing dronabinol (Marinol and Syndros) are synthetic, dronabinol is also a naturally occurring compound found in Cannabis. In contrast, nabilone is not naturally occurring; it is a man-made drug with structural similarities to THC. In addition to the four cannabis-related products available in the U.S., a product called nabiximols (Sativex), a naturally sourced full-spectrum botanical extract with ∼1:1 mixture of THC:CBD in an oromucosal spray, is currently marketed legally in over 25 countries, including Mexico, Canada, Australia, the United Kingdom, and parts of Europe.[95][96] A Dutch-based company, Bedrocan, also offers five different GMP-certified, pharmaceutical-grade cannabis products with varying THC:CBD ratios (i.e., THC-dominant, THC-intermediate, and CBD-dominant) with complete terpene profiles. Bedrocan’s products are available in Australia, South Africa, most of Europe, parts of the United Kingdom, and Sweden.[96] Finally, a CBD-based transdermal gel called Zygel (Zynerba) is being tested in various stages of clinical trials for the treatment of Fragile X syndrome and other neuropsychiatric conditions, including autism spectrum disorder (ASD) and 22q.[97]
|
Cannabinoid drugs each fall under a different level of controlled substance regulation (regulations initiated by the CSA and enforced by the DEA), based on each drug’s risk for abuse and physical and/or psychological dependence.[98] A drug will occasionally change in status with new legislation. Marinol (dronabinol) was originally a Schedule II controlled substance, and is now listed under Schedule III, indicating it has lower risk for abuse than Schedule I or II substances, and has risk of low-to-moderate physical dependency or high psychological dependency if abused.[98] As controlled substances, Syndros (dronabinol) and Cesamet (nabilone) are both under Schedule II. Epidiolex, a drug containing <0.1% THC, was originally classified in Schedule I, but after the 2018 Farm Bill was enacted, it was reduced to Schedule V, the least restrictive class of controlled substances.[99] In April of 2020, Epidiolex was entirely descheduled and is no longer subject to the tracking and monitoring requirements of the CSA. The term "medical marijuana" does not refer to any pharmaceutical drug, but rather use of the whole cannabis plant (with >0.3% THC) as medicine in any product (dried flower, resin, tincture, capsule, etc.) or delivery system (inhalation, oral, sublingual, topical, etc.).[100] Despite medical and recreational marijuana becoming increasingly legal in several states, medical marijuana is still federally illegal and is classified as a Schedule I controlled substance. Physicians may write prescriptions for medical marijuana for patients with certain state-approved medical conditions, which vary widely by state.[100] Hemp, referring to whole plant cannabis with <0.3% THC, was officially removed from the CSA in 2018, so it is no longer a controlled substance and is now regulated under the category of products "generally regarded as safe" (GRAS).
In the U.S., dronabinol (Marinol) was the first FDA approved cannabinoid drug in 1985. By 1999, annual sales of Marinol had reached over $20 billion, and it was primarily being used (>80% of patients) in the treatment of HIV and AIDS as an appetite stimulant.[98] Marinol is an oil-based capsule taken orally and, since 1992, has been approved to treat two different conditions: chemotherapy-induced nausea and vomiting (CINV) in cancer patients unresponsive to traditional antiemetics and cachexia in AIDS wasting syndrome.[98] Preliminary clinical trials suggest dronabinol may also be beneficial for patients with obstructive sleep apnea (OSA) and chronic pain, but further evidence is needed.[101] For some patients suffering with chemo-induced nausea and vomiting, oral capsules are an impractical delivery system due to swallowing difficulties, long absorption times (approximately two hours), and high variability in intraindividual absorption and bioavailability.[102][103] Syndros, a liquid delivery form of dronabinol, was approved in July 2016 as an easier-to-swallow administration option with lower intraindividual variability, faster absorption time (nine minutes vs. two hours), and the option to titrate the dosage to clinical effect.[102] Albeit more consistent and tolerable than capsules, because of its liquid delivery system, Syndros may be more easily abused as a drug than Marinol, as it can be converted for inhalation (thus, the Schedule II status).
Although many systematic reviews and meta analyses have verified dronabinol’s appetite stimulatory and potent antiemetic effects, both alone and in combination with traditional antiemetics like Odensatron[95][101][104][105], others suggest dronabinol may be no more effective in treating CINV than modern conventional antiemetics and may also have more side effects.[106] In addition, many patients reportedly prefer smoking or vaping naturally derived THC-containing products to using synthetic THC medications.[99][103][107][108] Some patients also seem to benefit more from combination THC-CBD products[99][109] and/or whole plant medical marijuana with its synergistic cannabinoids, terpenes, and flavonoids than using THC alone. It is important to note that FDA-approved cannabinoid medications have better dosage regulation and quality control than most natural cannabis products that are state-regulated. These approved medications are backed by in-depth multi-stage clinical trials evaluating their safety and efficacy, which is not true for most medical marijuana products. Due to historical difficulties in studying cannabis as a Schedule I controlled substance, a lot more research is needed in this area.[99][108]
The same year that dronabinol was approved (1985), a second oral THC-related drug, nabilone (Cesamet) (Figure 4) was also approved for the treatment of CINV unresponsive to conventional antiemetics and anorexia in AIDS patients.[110][111] As a synthetic analog of THC (Figure 4), nabilone has enough chemical structural differences from THC that it cannot be converted to THC after ingestion and is not detected as a positive for THC in urinary toxicology screenings.[110] As a potent cannabinoid receptor agonist, nabilone is reported to have a faster absorption time, stronger potency, increased bioavailability, and longer duration of action compared to dronabinol, thus requiring lower and less frequent dosing.[110] In addition to its approved uses, early research suggests nabilone may also be useful in treating chronic and neuropathic pain, insomnia, cannabis withdrawal and relapse, posttraumatic stress disorder (PTSD), chronic anxiety, and any combination of these frequently comorbid conditions.[34][110][112]
In June of 2018, Epidiolex (an oral solution containing 100 mg/ml CBD and <0.1% THC) was approved in the U.S., initially for seizures associated with two rare and drug-resistant conditions—Lennox-Gastaut Syndrome and Dravet Syndrome—in patients ≥2 years of age (updated to ≥1 year of age in 2020). In August of 2020, Epidiolex was also approved for use in tuberous sclerosis complex (TSC) seizures in patients ≥1 year of age.[95][113] CBD, the primary active constituent in Epidiolex, is metabolized largely through cytochrome P450 (CYP450) enzymes, is a substrate of CYP3A4 and CYP2C19, and also affects the CYP2C family.[114] Several studies raise concerns regarding drug-drug interactions with CBD and the potential hepatotoxicity of CBD with substantial doses, such as those given with Epidiolex.[114] Hepatotoxicity has been demonstrated through liver transaminase elevation and increased liver weights of rhesus monkeys, as well as histopathological evidence.[114][115][116][117] The application of therapeutic doses in human trials has suggested that hepatotoxicity doesn’t occur in all cases and heavily relies on setting, dose, and timing.[118] The benefits, in nearly all cases, are determined to outweigh the risks.[114]
The same manufacturer of Epidiolex—UK-based GW Pharmaceuticals—also manufactures a medicinal cannabis product called Sativex (nabiximols) for the treatment of moderate to severe spasticity in multiple sclerosis patients who have not responded adequately to other MS medications.[119][120] Unlike any of the above cannabinoid drugs with a single purified or synthesized cannabinoid, Sativex comprises a standardized whole plant Cannabis extract with controlled amounts of not just Δ9-THC and CBD (2.7 and 2.5 mg per 100 uL single spray, respectively), but also minor cannabinoids, terpenoids, flavonoids, fatty acids, and sterols.[119] Patients generally self-titrate the medication, starting with a single spray and increasing up to 12 sprays daily over the course of two weeks. Several systematic reviews of Sativex in uncontrolled MS-related spasticity suggest it has long term benefit in reducing spasticity, especially as adjunctive treatment, and is also generally well tolerated and safe, does not typically result in dependency or abuse, and has low risk for negative psychoactive effects.[119][120][121][122]
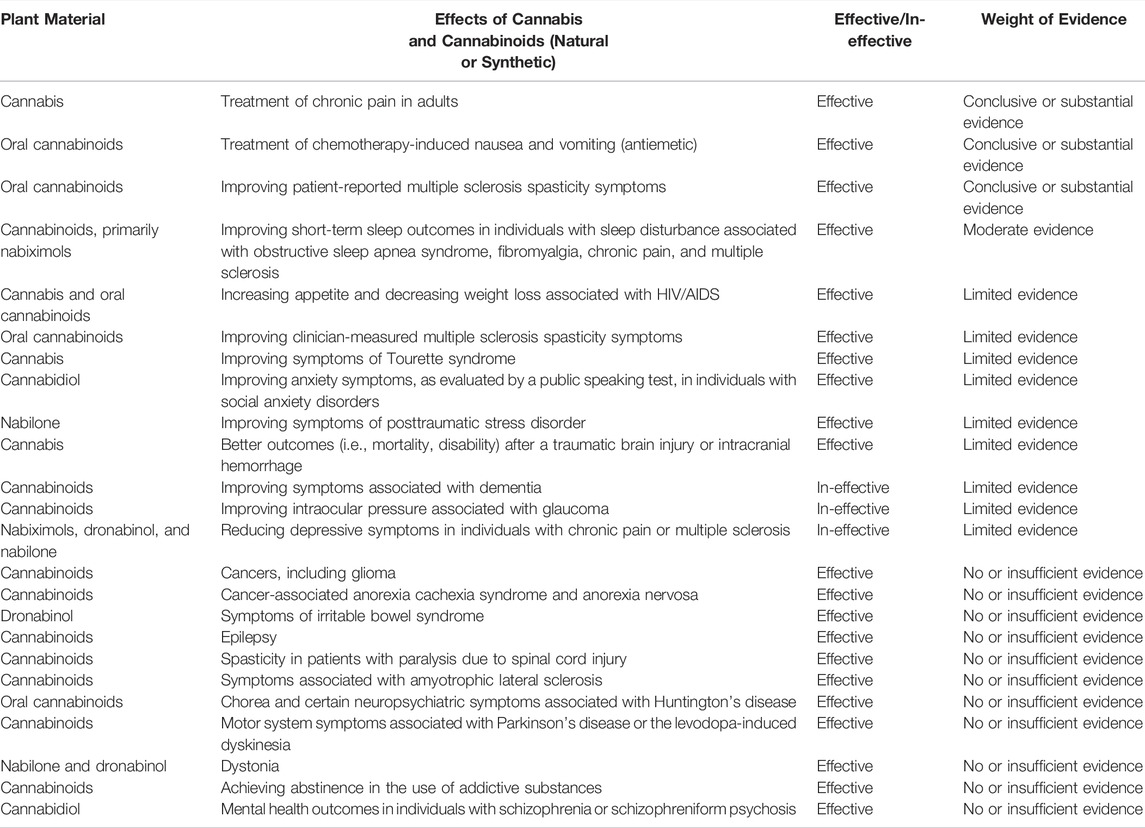
In 2017, the committee on health effects of marijuana from the National Academies of Sciences, Engineering, and Medicine (U.S.) by investigating more than 10.700 abstracts, published an evidence-based review of the health effects of cannabis and cannabinoids. Regarding the efficacy of cannabis and cannabinoids (either natural or synthetic), the weight of evidence was categorized mainly as "conclusive evidence," "substantial evidence," "moderate evidence," "limited evidence," and “no or insufficient evidence” based on the clinical trials (Table 1).[123]
|
Regulatory status
Before the late 1950s, the production of hemp was encouraged by the U.S. Department of Agriculture (USDA).[2] After introducing THC as the psychotropic constituent of Cannabis sativa L., authorities started to make policies against the expansion of cultivating all types of the cannabis plant without distinguishing between low- and high-THC varieties.[6] Cannabis was internationally banned by the Single Convention on Narcotic Drugs in 1961 and placed in Schedule I, along with substances like heroin. Schedule I controlled substances are concluded to be highly addictive, have great potential for abuse, and/or have capacity for use as precursors for other illegal drugs. In addition, the Convention on Psychotropic Substances in 1971 listed THC in Schedule I due to its potential for abuse, threat to public health, and low therapeutic potential. In 1988, the United Nations Convention against Illicit Traffic in Narcotic Drugs and Psychotropic Substances prohibited "the production, manufacture, supply, distribution, sale, transportation, import, or export of any narcotic drug or psychotropic substance." It also prohibited possession of these substances and cultivation of plants under the legislation of states parties.[13] As a result, the importance of scientific and medical research regarding cannabis was marginalized.[15]
Today, the regulatory aspect of cannabis that is of greatest concern in most jurisdictions is the employment of strict THC limits to determine which plants fall under "hemp" status and can be legally cultivated, sold, and marketed as such. A limit of 0.3% THC (dry weight basis) in "young, vigorous leaves of relatively mature plants" was a guideline arbitrarily adopted by Small et al.[12] for this purpose, and many countries, including the U.S., have established this same criterion of a 0.3% THC limit. The range for THC limits across the globe currently spans between 0.2 and 1.0%, but most countries have chosen 0.2% as the upper limit. Relatively few jurisdictions, including the Swiss government, have increased the legal limit for hemp cultivars to as high as 1.0% THC, a much higher percent than the rest of the European Union (E.U.), which currently limits THC content to just 0.2%. The E.U. legalized the cultivation of Cannabis sativa L. plants for industrial applications in 2013, limiting trade to varieties with less than 0.2% THC dry weight of leaves and flowering parts.[124] The E.U. had initially set the upper limit for industrial hemp to 0.5% THC in 1984, then reduced it to 0.3% in 1987, and finally to 0.2% in 1999. In Canada, cultivating hemp varieties with THC content below 0.3% dry weight of leaves and flowering parts was allowed in 1998. Today, Canada is the largest producer and exporter of hemp products, in particular, hemp-based food and ingredients, around the world.[125] The hemp-producing market of the E.U. is second only to Canada. Countries including France, Netherlands, Lithuania, and Romania are among the largest producers in the E.U.[125] The E.U. also certifies specific hemp varieties of seeds in the Common Catalogue of Varieties of Agricultural Plant Species and grants subsidies to farmers using these catalogued seeds as long as they remain <0.2% THC.[126]
In each of the above cases, measurements of THC concentrations are usually required to include not just THC, but also THC's carboxylated precursor THCA and sometimes its non-enzymatic oxidation byproduct CBN.[14][26][126] THCA is more prevalent than THC in fresh cannabis plant material, as well as in samples dried at low temperature that have not been in storage for long.[126] When THCA is naturally decarboxylated to THC, there is a loss of molecular mass, so calculations for %w/w (%mg of cannabinoid/100 mg herbal drug) for total THC concentration should include the following ratio of 0.877% THCA:
- % of Total Δ9-THC = (% Δ9-THCA x 0.877) + % Δ9-THC + % CBN
Similarly, when calculating total CBD concentration, the following should be performed to account for the decarboxylation reaction of CBDA to CBD:
- % of Total CBD = (% CBDA x 0.877) + % CBD
The preceding paragraphs specifically discuss hemp regulations, which likely define the most important aspect of cannabis governance today; however, this unique plant presents other regulatory challenges, such as the legalization of medical and/or recreational THC-dominant cannabis, determining which conditions are eligible for medical THC use, discrepancies between federal and state cannabis regulations, and confusion around the use of isolated cannabinoids, like CBD, which are present in full- and broad-spectrum hemp extracts.
In terms of recreational cannabis legalization, Latin America has been the world’s pioneer in facilitating regulations for recreational THC-dominant cannabis. Uruguay was the first country in the world which legalized production, distribution, sale, and consumption of high-THC cannabis and its derivatives for purposes other than medical and scientific uses.[127] Cannabis markets in Uruguay were allowed to sell their products for medicinal, scientific, industrial, and recreational purposes under several considerations.[15] After regulatory changes in 2017, Germany was one of the first countries to pronounce that both public health services and private medical insurances should cover medical cannabis treatments.[15] Select countries like Portugal have decriminalized ("eliminating criminal penalties for the unauthorized consumption and possession (typically of amounts small enough to be for personal use only) of a controlled substance"[127] the use of marijuana. In Spain, possession of marijuana was decriminalized too, but not its sale. In the Netherlands, use of marijuana has been de facto decriminalized (“not applying statutes that penalize the production, distribution, or consumption of a substance to the fullest extent”).[127] Also in the Netherlands, Cannabis manufacturer Bedrocan has expanded to procure vast ownership over medicinal cannabis production in the country and produces, standardizes, and exports large amounts of cannabis products with different THC and CBD concentrations. More strict policies still apply in parts of Asia, as medical cannabis has remained prohibited in Iran, Japan, Vietnam, and Pakistan. However, in India, several legal provisions for use of medical cannabis have been permitted.[15][128]
In the U.S. although marijuana remains a Schedule I controlled substance under federal law—which bans the manufacture, distribution, dispensation, and possession of marijuana—individual states have elected to legalize marijuana for recreational use apart from federal guidelines.[125] In the U.S., marketing of all cannabis-derived products and CBD was banned until a few years ago based on their Schedule I status and concerns about their side effects. The 2018 Farm Bill modified the definition of hemp so that hemp was no longer a controlled substance. Hemp cultivation in the U.S. is now under USDA-approved license, while growing marijuana is generally prohibited because it is considered to have drug-abuse potential.[2] Regardless of federal restrictions, select states allow for the legal cultivation of cannabis plants both for medicinal and recreational purposes. Dietary supplements of CBD remain federally illegal, but the FDA has primarily taken action only against marketers making disease treatment claims. After approving Epidiolex, which contains the active ingredient CBD, for certain epilepsy conditions, CBD was inevitably assigned pharmaceutical drug status and could no longer be treated like a dietary supplement.[118]
Analytical methods
Based on International Narcotic Control Board (INCB) reports, total global production of cannabis increased from 1.3 tons in 2000 to 100.2 tons in 2015.[129] Third-party cannabis testing laboratories are responsible for analysis and quality control of this massive amount of raw material, as well as any final product containing cannabis, whether THC-dominant, THC-intermediate, or CBD-dominant. In addition to phytocannabinoid potency testing, which is typically done via high-performance liquid chromatography (HPLC), many cannabis labs also now commonly test for terpene content (typically done via gas chromatography–mass spectrometry [GC-MS]), as well as contaminants such as pesticides, heavy metals (cannabis is a hyperaccumulator), mycotoxins, and residual solvents on derivatives and edibles. For any final cannabis products, in addition to the above analyses, many labs also test for water activity, moisture content, and homogeneity to determine potential for microbial growth and an equal distribution of cannabinoids within the product, respectively. Testing requirements are overseen by specific regulating bodies where cannabis is being grown, traded, or consumed. Labs regularly consult with growers, insurers, producers, distributors, and retailers to ensure final products are labeled and marketed accurately and are safe for consumers. In addition to the above tests, some other tests labs occasionally perform include product stability, flavonoid and lipid concentrations, gender testing, nutrient testing, and leachability studies.
The breadth of all things cannabis lab testing is beyond the scope of this article. Our focus will be on phytocannbinoid profiling to determine CBD- vs. THC-dominant chemovars, as well as the pros and cons of the most common analysis platforms.
Though different analytical methods and guidelines exist, differentiating between CBD- and THC-dominant cannabis chemotypes is no longer a difficult process due to increased knowledge of biochemical and biomolecular characteristics of Cannabis sativa L. and the development of instruments and analytical methods that facilitate detection and exact quantification of cannabinoid content.[6][130] The United Nations suggested identifying cannabinoids fast and easily with color tests such as the Fast Corinth V salt test, the Fast Blue B salt test, and the Rapid Duquenois test. Thin-layer chromatography (TLC) in both normal and reverse phase systems, besides visualizing with Fast Blue B salt as a reagent, may be used to detect cannabinoids generally. TLC is typically used for fast screening of cannabinoids and does not provide further information; in addition, it has low specificity and sensitivity, and false-positive results may be seen.[9][13][131] An accurate, reproducible, and rapid high-performance TLC (HPTLC) method was developed for quantitative analysis of total THC quantification (decarboxylated THCA + THC + CBN) along with a qualitative fingerprint of the other primary neutral cannabinoids (CBD, THCV, CBG, and CBC), with results comparable to that of an HPLC fingerprint.[130][132] HPTLC may be helpful in screening for high levels of THC in hemp and hemp products.
Gas chromatography (GC) is one of the oldest and most common methods used for quantifying cannabinoids. The method is accomplished in a short period (under 20 minutes) at 300°C with low polarity stationary phases (e.g., 5% diphenyl and 95% dimethyl polysiloxan). Cannabinoids are partly present in the plant in their acidic forms. Quantification of acidic cannabinoids with GC is impossible because they undergo decarboxylation during passage through the hot injection port; hence, it is necessary for cannabinoids to be derivatized prior to GC. Derivatizing also results in better peak shape and more volatility.[9][133] Flame ionization detectors (FIDs) and mass spectrometry (MS) are commonly used as detectors for the recognition and quantification of cannabinoids, coupled with GC. GC-MS is the most researched and reviewed platform for analytical cannabinoid profiling to date.[131][134][135] GC-FID provides a more accurate and cheaper quantification than GC-MS because GC-MS requires expensive and not always available equivalent deuterated standards.[131] Due to its higher efficacy, helium gas generally is preferred over nitrogen and hydrogen in studies as a carrier gas in GC-MS.[136] Several other GC methods have been reported. Some of these include GC technology and starch gel electrophoresis[137], multiple reaction monitoring (MRM) GC coupled with triple quadrupole MS[138], gas chromatography–vacuum ultraviolet spectroscopy (GC-VUV)[139], and two-dimensional gas chromatography.[140]
HPLC is gaining popularity as the botanical chemical fingerprint method of choice, including for phytocannabinoid profiling.[130] The most commonly analyzed cannabinoids include ∆9-THC, THCA, CBD, CBDA, CBN, CBG, and CBC. Using C18 columns for the stationary phase allows for high resolution and separation of cannabinoids. The addition of 0.1% formic acid to methanol or water for the mobile phase improves peak shapes and enhances resolution.[9][131] It is possible to quantify both neutral and acidic cannabinoids with HPLC (i.e., THC and THCA), due to low-temperature operation. In contrast, HPLC is not appropriate for the analysis of volatile compounds such as terpenes, which are better analyzed via GC-MS.[136] MS and ultraviolet (UV) are commonly coupled with HPLC as a detector.[136] UV is cheaper and more straightforward than MS.[9] Neutral cannabinoids show a UV absorption peak around 220 nm, and acidic cannabinoids peak in the range of 270–310 nm.[141][142] MS may provide higher specificity than UV, which makes it possible to analyze complex matrices of cannabis.[131] To yield further information and improve specificity, the diode array detector (DAD) has been suggested by some authors.[9] HPLC-DAD differentiates between Cannabis sativa L. chemotypes based on different absorption spectra.[143] In order to differentiate between CBD and CBG, using HPLC-MS is preferred over HPLC-UV because UV does not provide enough resolution in this case, with certainty, in above 10% concentration of extract.[136][144] Some other methods have also been shown to have different advantages in the quantification of cannabinoids, including liquid chromatography–tandem mass spectrometry (LC-MS/MS) combined with atmospheric-pressure chemical ionization (APCI)[145], HPLC-MS/MS[146], and ultra-high-performance liquid chromatography–quadrupole time-of-flight (UPLC-qTOF).[136]
Many labs prefer either GC- or LC-based methods for cannabinoid testing. According to a recent comprehensive literature review of the past four decades of analytical methods reported for cannabinoid profiling in over 220 scientific papers, Stefkov et al.[130] states that GC- and LC-based methods, at this point in time, exhibit comparable accuracy, selectivity, linearity, sensitivity, and precision. Both are used routinely and in investigational analysis of cannabis and cannabis products. HPTLC may soon prove to be superior to either one of these (GC techniques or HPLC) due to the possibilities of lower running costs, quicker analysis run time, automation of sample application and spotting, and ability to analyze multiple samples simultaneously.
In addition to developing a highly sensitive, precise, and affordable instrumental analysis, the identification of plant materials, sample selection, and preparation should be taken into account.
Macroscopic and microscopic features are primarily used to identify the plant material. Differential identification is accomplished to figure out the presence of closely related species or other adulterations. HPTLC fingerprint could complete the macroscopic and microscopic techniques, as it provides important data about the major cannabinoids. The USP general chapters <563>, <203>, and <1064> describe general procedures, which could be useful in the identification of botanicals.[14] A proper sample of plant material should represent the characteristics of the whole batch to ensure reproducibility in the results of quantification analyses. A non-homogenized or stratified batch may lead to under- or overestimation of cannabinoid content (due to higher levels of cannabinoids in glandular hair trichomes).[14] The USP general chapter <561> explains a valid sampling procedure for botanical drugs. The procedure is based on the weight of the holding container. Contents should be mixed for less than 1 kg containers. For 1–5 kg containers, sampling should be performed by withdrawing an equal amount of plant material from the upper, middle, and lower parts of the container. For above 5 kg containers, three samples must be taken from each of the upper, middle and lower parts.[147]
Sample preparation varies and depends mainly on the aim of analysis and sample form (solid, liquid, or gas).[148] For non-biological samples, which range from raw materials and cannabis extracts to semi- (mixture of cannabinoids) or fully-purified (single cannabinoid) materials and formulated cannabis preparations (e.g., edible oil or capsule), analysis of cannabinoids is done to determine the chemical profile of cannabinoids. Cannabis extraction is the process of isolating cannabinoids or terpenes from cannabis trichomes with the purpose of producing medical or recreational supplements and is necessary before any quantification or purification. This process involves several consecutive procedures as follows[148]:
- Harvesting the plant material
- Trimming the flowers
- Drying the flowers in the absence of sunlight to avoid chemical degradation
- Milling the flowers to decrease particle size
- Immersing the flowers in a solvent (constituents release into the solvent)
- Winterizing the solution (a deep-freezing procedure with the aim of the sedimentation of a viscous black residue called wax)
- Filtrating the solution to remove wax from the extract
- Removing all of the solvent using a rotary evaporator
Further purification methods, might be followed according to the final goal, such as crystallization or chromatography. Polar solvents are routinely used for the solvent extraction of semi-polar cannabinoids or terpenes from the flower trichomes, e.g., using ethanol, methanol, isopropanol, or dimethyl ether.[149]
Biological specimens (blood, plasma, serum, urine, oral fluid, breath, and hair) typically are analyzed for research purposes and forensic toxicological and drug abuse investigations.[150] Several solvent-based extraction approaches are used for solid samples to extract the marker analytes (cannabinoids in this case) and remove any byproducts. Liquid-phase extraction (LPE), solid-phase extraction (SPE), and solvent extraction assisted with microwave, ultrasonic, or pressure methods have been proposed for solid sample preparation. Solid-phase microextraction (SPME) is widely employed for hair samples due to increased automation and potential for high throughput. Liquid samples are mostly involved in the detection of drug abuse cases. Similar to solid samples, liquid samples must be processed for isolation of cannabinoids and elimination of interferences. SPE and liquid-Liquid extraction (LLE)—in addition to recent microextraction methods, including solid-phase microextraction, packed sorbent microextraction, dispersive solid-phase extraction, dispersive liquid-liquid microextraction, and hollow fiber-based liquid-phase microextraction—are used for this purpose. As for gas samples, a sample of air is typically prepared through capillary microextraction. For aerosols, liquid-phase extraction is utilized after collecting airborne particles.[148]
Discussion
Confusion and inappropriate use of the terms "hemp" and "marijuana" is a great limitation in regards to broader acceptance and widespread use of the cannabis plant. Distinctions between hemp and marijuana extend beyond definitions and concentrations of THC, as they differ in chemical and genetic composition, production practices, and especially in their use as medicine, food, and in industrial applications. Hundreds of preclinical and clinical studies have been done to evaluate the efficacy and safety of phytocannabinoids like CBD in different conditions, but the body of evidence from high-quality clinical studies still lacks robust conclusions about CBD’s safety and efficacy in treating diseases other than epilepsy.
Due to the complex nature of the ECS and its role on several physiologic functions in the body such as mood, memory, inflammation, and appetite, there are still many uncertainties about the way exogenous phytocannabinoids interact with the human ECS, and many areas that still need further research.
In addition, there is an ongoing need for legislative and regulatory intervention in some countries in order to provide a platform for developing research about cannabis and related industries. Making policies that promote the advantages of cannabis and prevent its misuse is necessary, and policymakers should attempt to increase public knowledge about cannabis safety based on current research. Most of the primary issues with cannabis could likely be resolved through identifying and quantifying intoxicating compounds in both original plant material and cannabis products, agreement on universal nomenclature, reporting and labeling guidelines, and continued education for all parties involved, including farmers, insurers, policy makers, scientists, physicians, and consumers. Instrumental analytical methods and genetic testing will play a crucial role in achieving this goal.
Despite all of the recent advances, several cannabis topics remain to be addressed. For example, researchers still have room to explore the advantages of natural plant extracts over synthetic drugs, clarify legal use of full spectrum hemp products vs. low- and high-dose CBD-containing products, purify CBD in prescription medication, and explore the many benefits of lesser known terpenes and minor cannabinoids. Nevertheless, due to expanding knowledge about cannabis’s value in medicine and industry, as well as recent changes in legislation allowing for easier access to this plant, novel research findings are continuing to grow and spread around the world.
Acknowledgements
The authors thank Roy Upton (the founder, executive director, and editor of the American Herbal Pharmacopoeia [AHP]) for technical assistance and critical review.
Author contributions
SA conceived the presented idea and managed the team. AS and KP wrote the article with Co-operation. YS, SA, and BZ reviewed the article critically and they substantially contribute to the final manuscripts.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- ↑ Jin, Jian‐Jun; Yang, Mei‐Qing; Fritsch, Peter W.; Velzen, Robin; Li, De‐Zhu; Yi, Ting‐Shuang (1 July 2020). "Born migrators: Historical biogeography of the cosmopolitan family Cannabaceae" (in en). Journal of Systematics and Evolution 58 (4): 461–473. doi:10.1111/jse.12552. ISSN 1674-4918. https://onlinelibrary.wiley.com/doi/10.1111/jse.12552.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Johnson, R. (22 March 2019). "Defining Hemp: A Fact Sheet" (PDF). Congressional Research Service. https://www.everycrsreport.com/files/20190322_R44742_1b0195c6aa7e2cad29256c85a8574347c1ee833d.pdf.
- ↑ Stevens, Chris J; Murphy, Charlene; Roberts, Rebecca; Lucas, Leilani; Silva, Fabio; Fuller, Dorian Q (1 October 2016). "Between China and South Asia: A Middle Asian corridor of crop dispersal and agricultural innovation in the Bronze Age" (in en). The Holocene 26 (10): 1541–1555. doi:10.1177/0959683616650268. ISSN 0959-6836. PMC PMC5125436. PMID 27942165. http://journals.sagepub.com/doi/10.1177/0959683616650268.
- ↑ Li, Hui-Lin (1 July 1974). "The origin and use of cannabis in eastern asia linguistic-cultural implications" (in en). Economic Botany 28 (3): 293–301. doi:10.1007/BF02861426. ISSN 0013-0001. http://link.springer.com/10.1007/BF02861426.
- ↑ Cherney, Jerome; Small, Ernest (12 November 2016). "Industrial Hemp in North America: Production, Politics and Potential" (in en). Agronomy 6 (4): 58. doi:10.3390/agronomy6040058. ISSN 2073-4395. http://www.mdpi.com/2073-4395/6/4/58.
- ↑ 6.0 6.1 6.2 6.3 6.4 Farinon, Barbara; Molinari, Romina; Costantini, Lara; Merendino, Nicolò (29 June 2020). "The Seed of Industrial Hemp (Cannabis sativa L.): Nutritional Quality and Potential Functionality for Human Health and Nutrition" (in en). Nutrients 12 (7): 1935. doi:10.3390/nu12071935. ISSN 2072-6643. PMC PMC7400098. PMID 32610691. https://www.mdpi.com/2072-6643/12/7/1935.
- ↑ Piluzza, G.; Delogu, G.; Cabras, A.; Marceddu, S.; Bullitta, S. (1 December 2013). "Differentiation between fiber and drug types of hemp (Cannabis sativa L.) from a collection of wild and domesticated accessions" (in en). Genetic Resources and Crop Evolution 60 (8): 2331–2342. doi:10.1007/s10722-013-0001-5. ISSN 0925-9864. http://link.springer.com/10.1007/s10722-013-0001-5.
- ↑ Abel, Ernest L. (2013) (in en). Marihuana: The First Twelve Thousand Years. Boston, MA: Springer New York. pp. 289. doi:10.1007/978-1-4899-2189-5. ISBN 978-1-4899-2191-8. http://link.springer.com/10.1007/978-1-4899-2189-5.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 Leghissa, Allegra; Hildenbrand, Zacariah L.; Schug, Kevin A. (1 January 2018). "A review of methods for the chemical characterization of cannabis natural products" (in en). Journal of Separation Science 41 (1): 398–415. doi:10.1002/jssc.201701003. https://onlinelibrary.wiley.com/doi/10.1002/jssc.201701003.
- ↑ 10.0 10.1 Sawler, Jason; Stout, Jake M.; Gardner, Kyle M.; Hudson, Darryl; Vidmar, John; Butler, Laura; Page, Jonathan E.; Myles, Sean (26 August 2015). Tinker, Nicholas A.. ed. "The Genetic Structure of Marijuana and Hemp" (in en). PLOS ONE 10 (8): e0133292. doi:10.1371/journal.pone.0133292. ISSN 1932-6203. PMC PMC4550350. PMID 26308334. https://dx.plos.org/10.1371/journal.pone.0133292.
- ↑ Pollio, Antonino (1 December 2016). "The Name of Cannabis : A Short Guide for Nonbotanists" (in en). Cannabis and Cannabinoid Research 1 (1): 234–238. doi:10.1089/can.2016.0027. ISSN 2378-8763. PMC PMC5531363. PMID 28861494. http://www.liebertpub.com/doi/10.1089/can.2016.0027.
- ↑ 12.0 12.1 Small, Ernest; Cronquist, Arthur (1 August 1976). "A Practical and Natural Taxonomy for Cannabis" (in en). TAXON 25 (4): 405–435. doi:10.2307/1220524. ISSN 0040-0262. https://onlinelibrary.wiley.com/doi/abs/10.2307/1220524.
- ↑ 13.0 13.1 13.2 United Nations Office on Drugs and Crime (November 2013). "The International Drug Control Conventions" (PDF). United Nations. https://www.unodc.org/documents/commissions/CND/Int_Drug_Control_Conventions/Ebook/The_International_Drug_Control_Conventions_E.pdf.
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 Sarma, Nandakumara D.; Waye, Andrew; ElSohly, Mahmoud A.; Brown, Paula N.; Elzinga, Sytze; Johnson, Holly E.; Marles, Robin J.; Melanson, Jeremy E. et al. (24 April 2020). "Cannabis Inflorescence for Medical Purposes: USP Considerations for Quality Attributes" (in en). Journal of Natural Products 83 (4): 1334–1351. doi:10.1021/acs.jnatprod.9b01200. ISSN 0163-3864. https://pubs.acs.org/doi/10.1021/acs.jnatprod.9b01200.
- ↑ 15.0 15.1 15.2 15.3 15.4 Aguilar, S.; Gutiérrez, V.; Sánchez, L. et al. (April 2018). "Medicinal cannabis policies and practices around the world" (PDF). International Drug Policy Consortium. http://fileserver.idpc.net/library/Medicinal%20cannabis%20briefing_ENG_FINAL.PDF.
- ↑ Cadena, A. (10 September 2018). "Hemp vs Marijuana: The Difference Explained". CBDOrigin. https://cbdorigin.com/hemp-vs-marijuana/.
- ↑ 17.0 17.1 17.2 17.3 17.4 Crini, Grégorio; Lichtfouse, Eric; Chanet, Gilles; Morin-Crini, Nadia (1 September 2020). "Applications of hemp in textiles, paper industry, insulation and building materials, horticulture, animal nutrition, food and beverages, nutraceuticals, cosmetics and hygiene, medicine, agrochemistry, energy production and environment: a review" (in en). Environmental Chemistry Letters 18 (5): 1451–1476. doi:10.1007/s10311-020-01029-2. ISSN 1610-3653. https://link.springer.com/10.1007/s10311-020-01029-2.
- ↑ Drug Enforcement Administration, Department of Justice (21 August 2020). "Implementation of the Agriculture Improvement Act of 2018". Federal Register. National Archives. https://www.federalregister.gov/documents/2020/08/21/2020-17356/implementation-of-the-agriculture-improvement-act-of-2018.
- ↑ Legal Information Institute. "21 U.S. Code § 802 - Definitions". Cornell Law School. https://www.law.cornell.edu/uscode/text/21/802#16.
- ↑ van Bakel, Harm; Stout, Jake M; Cote, Atina G; Tallon, Carling M; Sharpe, Andrew G; Hughes, Timothy R; Page, Jonathan E (2011). "The draft genome and transcriptome of Cannabis sativa" (in en). Genome Biology 12 (10): R102. doi:10.1186/gb-2011-12-10-r102. ISSN 1465-6906. PMC PMC3359589. PMID 22014239. http://genomebiology.biomedcentral.com/articles/10.1186/gb-2011-12-10-r102.
- ↑ 21.0 21.1 Weiblen, George D.; Wenger, Jonathan P.; Craft, Kathleen J.; ElSohly, Mahmoud A.; Mehmedic, Zlatko; Treiber, Erin L.; Marks, M. David (1 December 2015). "Gene duplication and divergence affecting drug content in Cannabis sativa" (in en). New Phytologist 208 (4): 1241–1250. doi:10.1111/nph.13562. ISSN 0028-646X. https://onlinelibrary.wiley.com/doi/10.1111/nph.13562.
- ↑ 22.0 22.1 Borroto Fernandez, Eduviges; Peterseil, Verena; Hackl, Gerald; Menges, Sonja; Meijer, Etienne; Staginnus, Christina (1 May 2020). "Distribution of Chemical Phenotypes (Chemotypes) in European Agricultural Hemp ( Cannabis sativa L.) Cultivars" (in en). Journal of Forensic Sciences 65 (3): 715–721. doi:10.1111/1556-4029.14242. ISSN 0022-1198. https://onlinelibrary.wiley.com/doi/10.1111/1556-4029.14242.
- ↑ Hill, Andrew J.; Williams, Claire M.; Whalley, Benjamin J.; Stephens, Gary J. (1 January 2012). "Phytocannabinoids as novel therapeutic agents in CNS disorders" (in en). Pharmacology & Therapeutics 133 (1): 79–97. doi:10.1016/j.pharmthera.2011.09.002. https://linkinghub.elsevier.com/retrieve/pii/S016372581100180X.
- ↑ Hanuš, Lumír Ondřej; Meyer, Stefan Martin; Muñoz, Eduardo; Taglialatela-Scafati, Orazio; Appendino, Giovanni (2016). "Phytocannabinoids: a unified critical inventory" (in en). Natural Product Reports 33 (12): 1357–1392. doi:10.1039/C6NP00074F. ISSN 0265-0568. http://xlink.rsc.org/?DOI=C6NP00074F.
- ↑ 25.0 25.1 25.2 25.3 25.4 Radwan, Mohamed M.; Chandra, Suman; Gul, Shahbaz; ElSohly, Mahmoud A. (8 May 2021). "Cannabinoids, Phenolics, Terpenes and Alkaloids of Cannabis" (in en). Molecules 26 (9): 2774. doi:10.3390/molecules26092774. ISSN 1420-3049. PMC PMC8125862. PMID 34066753. https://www.mdpi.com/1420-3049/26/9/2774.
- ↑ 26.0 26.1 26.2 26.3 Stack, George M.; Toth, Jacob A.; Carlson, Craig H.; Cala, Ali R.; Marrero‐González, Mariana I.; Wilk, Rebecca L.; Gentner, Deanna R.; Crawford, Jamie L. et al. (1 April 2021). "Season‐long characterization of high‐cannabinoid hemp ( Cannabis sativa L.) reveals variation in cannabinoid accumulation, flowering time, and disease resistance" (in en). GCB Bioenergy 13 (4): 546–561. doi:10.1111/gcbb.12793. ISSN 1757-1693. https://onlinelibrary.wiley.com/doi/10.1111/gcbb.12793.
- ↑ Russo, Ethan B (1 August 2011). "Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects: Phytocannabinoid-terpenoid entourage effects" (in en). British Journal of Pharmacology 163 (7): 1344–1364. doi:10.1111/j.1476-5381.2011.01238.x. PMC PMC3165946. PMID 21749363. https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.2011.01238.x.
- ↑ Potter, D. (March 2009). "The Propagation, Charactersation and Optimisation of Cannabis sativa L as a Phytopharmaceutical" (PDF). King's College London. https://extractionmagazine.com/wp-content/uploads/2018/06/THE-PROPAGATION-CHARACTERISATION-AND-OPTIMISATION-OF-CANNABIS-SATIVA-L-AS-A-PHYTOPHARMACEUTICAL.pdf.
- ↑ Fischedick, Justin Thomas; Hazekamp, Arno; Erkelens, Tjalling; Choi, Young Hae; Verpoorte, Rob (1 December 2010). "Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes" (in en). Phytochemistry 71 (17-18): 2058–2073. doi:10.1016/j.phytochem.2010.10.001. https://linkinghub.elsevier.com/retrieve/pii/S003194221000381X.
- ↑ Sommano, Sarana Rose; Chittasupho, Chuda; Ruksiriwanich, Warintorn; Jantrawut, Pensak (8 December 2020). "The Cannabis Terpenes" (in en). Molecules 25 (24): 5792. doi:10.3390/molecules25245792. ISSN 1420-3049. PMC PMC7763918. PMID 33302574. https://www.mdpi.com/1420-3049/25/24/5792.
- ↑ 31.0 31.1 31.2 31.3 31.4 31.5 Russo, Ethan B.; Marcu, Jahan (2017), "Cannabis Pharmacology: The Usual Suspects and a Few Promising Leads" (in en), Advances in Pharmacology (Elsevier) 80: 67–134, doi:10.1016/bs.apha.2017.03.004, ISBN 978-0-12-811232-8, https://linkinghub.elsevier.com/retrieve/pii/S1054358917300273
- ↑ Del Prado-Audelo, María Luisa; Cortés, Hernán; Caballero-Florán, Isaac H.; González-Torres, Maykel; Escutia-Guadarrama, Lidia; Bernal-Chávez, Sergio A.; Giraldo-Gomez, David M.; Magaña, Jonathan J. et al. (13 August 2021). "Therapeutic Applications of Terpenes on Inflammatory Diseases". Frontiers in Pharmacology 12: 704197. doi:10.3389/fphar.2021.704197. ISSN 1663-9812. PMC PMC8414653. PMID 34483907. https://www.frontiersin.org/articles/10.3389/fphar.2021.704197/full.
- ↑ Fischedick, Justin T. (1 January 2017). "Identification of Terpenoid Chemotypes Among High (−)- trans -Δ 9 - Tetrahydrocannabinol-Producing Cannabis sativa L. Cultivars" (in en). Cannabis and Cannabinoid Research 2 (1): 34–47. doi:10.1089/can.2016.0040. ISSN 2378-8763. PMC PMC5436332. PMID 28861503. http://www.liebertpub.com/doi/10.1089/can.2016.0040.
- ↑ 34.0 34.1 34.2 34.3 Lader, Malcolm (1 January 2009). "Addiction and the pharmacology of cannabis: implications for medicine and the law". Medicine, Science and the Law 49 (1): 1–17. doi:10.1258/rsmmsl.49.1.1. ISSN 0025-8024. https://doi.org/10.1258/rsmmsl.49.1.1.
- ↑ Freeman, Abigail M.; Petrilli, Katherine; Lees, Rachel; Hindocha, Chandni; Mokrysz, Claire; Curran, H. Valerie; Saunders, Rob; Freeman, Tom P. (1 December 2019). "How does cannabidiol (CBD) influence the acute effects of delta-9-tetrahydrocannabinol (THC) in humans? A systematic review" (in en). Neuroscience & Biobehavioral Reviews 107: 696–712. doi:10.1016/j.neubiorev.2019.09.036. https://linkinghub.elsevier.com/retrieve/pii/S0149763419305615.
- ↑ Premoli, Marika; Aria, Francesca; Bonini, Sara Anna; Maccarinelli, Giuseppina; Gianoncelli, Alessandra; Pina, Silvia Della; Tambaro, Simone; Memo, Maurizio et al. (1 May 2019). "Cannabidiol: Recent advances and new insights for neuropsychiatric disorders treatment" (in en). Life Sciences 224: 120–127. doi:10.1016/j.lfs.2019.03.053. https://linkinghub.elsevier.com/retrieve/pii/S0024320519302176.
- ↑ Noreen, Nida; Muhammad, Faqir; Akhtar, Bushra; Azam, Farooq; Anwar, Muhammad Irfan (2018). "Is Cannabidiol a Promising Substance for New Drug Development? A Review of its Potential Therapeutic Applications" (in en). Critical Reviews in Eukaryotic Gene Expression 28 (1): 73–86. doi:10.1615/CritRevEukaryotGeneExpr.2018021528. ISSN 1045-4403. http://www.dl.begellhouse.com/journals/6dbf508d3b17c437,642ad759707fc517,35856ff546e47ff9.html.
- ↑ 38.0 38.1 Cerino, Pellegrino; Buonerba, Carlo; Cannazza, Giuseppe; D'Auria, Jacopo; Ottoni, Ermete; Fulgione, Andrea; Di Stasio, Antonio; Pierri, Biancamaria et al. (1 February 2021). "A Review of Hemp as Food and Nutritional Supplement" (in en). Cannabis and Cannabinoid Research 6 (1): 19–27. doi:10.1089/can.2020.0001. ISSN 2578-5125. PMC PMC7891210. PMID 33614949. https://www.liebertpub.com/doi/10.1089/can.2020.0001.
- ↑ Laprairie, R B; Bagher, A M; Kelly, M E M; Denovan-Wright, E M (1 October 2015). "Cannabidiol is a negative allosteric modulator of the cannabinoid CB 1 receptor: Negative allosteric modulation of CB 1 by cannabidiol" (in en). British Journal of Pharmacology 172 (20): 4790–4805. doi:10.1111/bph.13250. PMC PMC4621983. PMID 26218440. https://onlinelibrary.wiley.com/doi/10.1111/bph.13250.
- ↑ Cascio, Mg; Gauson, La; Stevenson, La; Ross, Ra; Pertwee, Rg (1 January 2010). "Evidence that the plant cannabinoid cannabigerol is a highly potent α2-adrenoceptor agonist and moderately potent 5HT1A receptor antagonist: Novel pharmacological actions of cannabigerol" (in en). British Journal of Pharmacology 159 (1): 129–141. doi:10.1111/j.1476-5381.2009.00515.x. PMC PMC2823359. PMID 20002104. https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.2009.00515.x.
- ↑ De Petrocellis, Luciano; Di Marzo, Vincenzo (1 March 2010). "Non-CB1, Non-CB2 Receptors for Endocannabinoids, Plant Cannabinoids, and Synthetic Cannabimimetics: Focus on G-protein-coupled Receptors and Transient Receptor Potential Channels" (in en). Journal of Neuroimmune Pharmacology 5 (1): 103–121. doi:10.1007/s11481-009-9177-z. ISSN 1557-1890. http://link.springer.com/10.1007/s11481-009-9177-z.
- ↑ 42.0 42.1 De Petrocellis, Luciano; Ligresti, Alessia; Moriello, Aniello Schiano; Allarà, Marco; Bisogno, Tiziana; Petrosino, Stefania; Stott, Colin G; Di Marzo, Vincenzo (1 August 2011). "Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes: Novel pharmacology of minor plant cannabinoids" (in en). British Journal of Pharmacology 163 (7): 1479–1494. doi:10.1111/j.1476-5381.2010.01166.x. PMC PMC3165957. PMID 21175579. https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.2010.01166.x.
- ↑ Morales, Paula; Hurst, Dow P.; Reggio, Patricia H. (2017), Kinghorn, A. Douglas; Falk, Heinz; Gibbons, Simon et al.., eds., "Molecular Targets of the Phytocannabinoids: A Complex Picture", Phytocannabinoids (Cham: Springer International Publishing) 103: 103–131, doi:10.1007/978-3-319-45541-9_4, ISBN 978-3-319-45539-6, PMC PMC5345356, PMID 28120232, http://link.springer.com/10.1007/978-3-319-45541-9_4
- ↑ 44.0 44.1 De Petrocellis, L.; Orlando, P.; Moriello, A. Schiano; Aviello, G.; Stott, C.; Izzo, A. A.; Di Marzo, V. (1 February 2012). "Cannabinoid actions at TRPV channels: effects on TRPV3 and TRPV4 and their potential relevance to gastrointestinal inflammation: Plant cannabinoids and TRPV channels" (in en). Acta Physiologica 204 (2): 255–266. doi:10.1111/j.1748-1716.2011.02338.x. https://onlinelibrary.wiley.com/doi/10.1111/j.1748-1716.2011.02338.x.
- ↑ 45.0 45.1 Cascio, Maria Grazia; Pertwee, Roger G. (21 August 2014), Pertwee, Roger, ed., "Known Pharmacological Actions of Nine Nonpsychotropic Phytocannabinoids", Handbook of Cannabis (Oxford University Press): 137–156, doi:10.1093/acprof:oso/9780199662685.003.0007, ISBN 978-0-19-966268-5, https://academic.oup.com/book/27329/chapter/197029644
- ↑ Davis, W.Marvin; Hatoum, Nabil S. (1 January 1983). "Neurobehavioral actions of cannabichromene and interactions with Δ9-tetrahydrocannabinol" (in en). General Pharmacology: The Vascular System 14 (2): 247–252. doi:10.1016/0306-3623(83)90004-6. https://linkinghub.elsevier.com/retrieve/pii/0306362383900046.
- ↑ Maione, Sabatino; Piscitelli, Fabiana; Gatta, Luisa; Vita, Daniela; De Petrocellis, Luciano; Palazzo, Enza; de Novellis, Vito; Di Marzo, Vincenzo (1 February 2011). "Non-psychoactive cannabinoids modulate the descending pathway of antinociception in anaesthetized rats through several mechanisms of action: Phytocannabinoids in supraspinal pain control" (in en). British Journal of Pharmacology 162 (3): 584–596. doi:10.1111/j.1476-5381.2010.01063.x. PMC PMC3041249. PMID 20942863. https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.2010.01063.x.
- ↑ Shinjyo, Noriko; Di Marzo, Vincenzo (1 November 2013). "The effect of cannabichromene on adult neural stem/progenitor cells" (in en). Neurochemistry International 63 (5): 432–437. doi:10.1016/j.neuint.2013.08.002. https://linkinghub.elsevier.com/retrieve/pii/S0197018613002106.
- ↑ Wilkinson, Jonathan D.; Williamson, Elizabeth M. (1 February 2007). "Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis" (in en). Journal of Dermatological Science 45 (2): 87–92. doi:10.1016/j.jdermsci.2006.10.009. https://linkinghub.elsevier.com/retrieve/pii/S092318110600315X.
- ↑ Appendino, Giovanni; Gibbons, Simon; Giana, Anna; Pagani, Alberto; Grassi, Gianpaolo; Stavri, Michael; Smith, Eileen; Rahman, M. Mukhlesur (1 August 2008). "Antibacterial Cannabinoids from Cannabis sativa : A Structure−Activity Study" (in en). Journal of Natural Products 71 (8): 1427–1430. doi:10.1021/np8002673. ISSN 0163-3864. https://pubs.acs.org/doi/10.1021/np8002673.
- ↑ Qin, Ning; Neeper, Michael P.; Liu, Yi; Hutchinson, Tasha L.; Lubin, Mary Lou; Flores, Christopher M. (11 June 2008). "TRPV2 Is Activated by Cannabidiol and Mediates CGRP Release in Cultured Rat Dorsal Root Ganglion Neurons". The Journal of Neuroscience 28 (24): 6231–6238. doi:10.1523/JNEUROSCI.0504-08.2008. ISSN 0270-6474. PMC 6670541. PMID 18550765. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6670541/.
- ↑ Russo, Ethan B. (21 August 2014), Pertwee, Roger, ed., "The Pharmacological History of Cannabis", Handbook of Cannabis (Oxford University Press): 23–43, doi:10.1093/acprof:oso/9780199662685.003.0002, ISBN 978-0-19-966268-5, https://academic.oup.com/book/27329/chapter/197025364
- ↑ Upton, R.; Craker, L.; ElSohly, M. et al. (2014). "Cannabis inflorescence : cannabis spp. ; standards of identity, analysis, and quality control". American Herbal Pharmacopoeia and Therapeutic Compendium. American Herbal Pharmacopoeia. pp. 65. ISBN 9781929425372.
- ↑ Pertwee, R G (1 January 2008). "The diverse CB 1 and CB 2 receptor pharmacology of three plant cannabinoids: Δ 9 -tetrahydrocannabinol, cannabidiol and Δ 9 -tetrahydrocannabivarin: Δ 9 -THC, CBD and Δ 9 -THCV" (in en). British Journal of Pharmacology 153 (2): 199–215. doi:10.1038/sj.bjp.0707442. PMC PMC2219532. PMID 17828291. http://doi.wiley.com/10.1038/sj.bjp.0707442.
- ↑ Hill, Andrew J.; Weston, Samantha E.; Jones, Nicholas A.; Smith, Imogen; Bevan, Sarah A.; Williamson, Elizabeth M.; Stephens, Gary J.; Williams, Claire M. et al. (1 August 2010). "Δ⁹-Tetrahydrocannabivarin suppresses in vitro epileptiform and in vivo seizure activity in adult rats". Epilepsia 51 (8): 1522–1532. doi:10.1111/j.1528-1167.2010.02523.x. ISSN 1528-1167. PMID 20196794. https://pubmed.ncbi.nlm.nih.gov/20196794.
- ↑ Riedel, Gernot; Fadda, Paola; McKillop-Smith, Susan; Pertwee, Roger G; Platt, Bettina; Robinson, Lianne (31 March 2009). "Synthetic and plant-derived cannabinoid receptor antagonists show hypophagic properties in fasted and non-fasted mice: Cannabinoid antagonists as anorectic agents" (in en). British Journal of Pharmacology 156 (7): 1154–1166. doi:10.1111/j.1476-5381.2008.00107.x. PMC PMC2697695. PMID 19378378. https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.2008.00107.x.
- ↑ Williams, Claire M.; Jones, Nicholas A.; Whalley, Benjamin J. (21 August 2014), Pertwee, Roger, ed., "Cannabis and Epilepsy", Handbook of Cannabis (Oxford University Press): 547–563, doi:10.1093/acprof:oso/9780199662685.003.0030, ISBN 978-0-19-966268-5, https://academic.oup.com/book/27329/chapter/197043073
- ↑ Pertwee, Roger G.; Cascio, Maria Grazia (21 August 2014), Pertwee, Roger, ed., "Known Pharmacological Actions of Delta-9-Tetrahydrocannabinol and of Four Other Chemical Constituents of Cannabis that Activate Cannabinoid Receptors", Handbook of Cannabis (Oxford University Press): 115–136, doi:10.1093/acprof:oso/9780199662685.003.0006, ISBN 978-0-19-966268-5, https://academic.oup.com/book/27329/chapter/197029115
- ↑ 59.0 59.1 Moreno-Sanz, Guillermo (1 December 2016). "Can You Pass the Acid Test? Critical Review and Novel Therapeutic Perspectives of Δ 9 -Tetrahydrocannabinolic Acid A" (in en). Cannabis and Cannabinoid Research 1 (1): 124–130. doi:10.1089/can.2016.0008. ISSN 2378-8763. PMC PMC5549534. PMID 28861488. http://www.liebertpub.com/doi/10.1089/can.2016.0008.
- ↑ Verhoeckx, Kitty C. M.; Korthout, Henrie A. A. J.; van Meeteren-Kreikamp, A. P.; Ehlert, Karl A.; Wang, Mei; van der Greef, Jan; Rodenburg, Richard J. T.; Witkamp, Renger F. (1 April 2006). "Unheated Cannabis sativa extracts and its major compound THC-acid have potential immuno-modulating properties not mediated by CB1 and CB2 receptor coupled pathways". International Immunopharmacology 6 (4): 656–665. doi:10.1016/j.intimp.2005.10.002. ISSN 1567-5769. PMID 16504929. https://pubmed.ncbi.nlm.nih.gov/16504929.
- ↑ Rock, E M; Kopstick, R L; Limebeer, C L; Parker, L A (1 October 2013). "Tetrahydrocannabinolic acid reduces nausea-induced conditioned gaping in rats and vomiting in Suncus murinus: THCA, emesis and nausea" (in en). British Journal of Pharmacology 170 (3): 641–648. doi:10.1111/bph.12316. PMC PMC3792001. PMID 23889598. https://onlinelibrary.wiley.com/doi/10.1111/bph.12316.
- ↑ Rosenthaler, Sarah; Pöhn, Birgit; Kolmanz, Caroline; Nguyen Huu, Chi; Krewenka, Christopher; Huber, Alexandra; Kranner, Barbara; Rausch, Wolf-Dieter et al. (1 November 2014). "Differences in receptor binding affinity of several phytocannabinoids do not explain their effects on neural cell cultures" (in en). Neurotoxicology and Teratology 46: 49–56. doi:10.1016/j.ntt.2014.09.003. https://linkinghub.elsevier.com/retrieve/pii/S089203621400172X.
- ↑ Alozie, Sydney O.; Martin, Billy R.; Harris, Louis S.; Dewey, William L. (1 February 1980). "3H-Δ9-tetrahydrocannabinol, 3H-cannabinol and 3H-cannabidiol: Penetration and regional distribution in rat brain" (in en). Pharmacology Biochemistry and Behavior 12 (2): 217–221. doi:10.1016/0091-3057(80)90359-7. https://linkinghub.elsevier.com/retrieve/pii/0091305780903597.
- ↑ Deiana, Serena; Watanabe, Akihito; Yamasaki, Yuki; Amada, Naoki; Arthur, Marlene; Fleming, Shona; Woodcock, Hilary; Dorward, Patricia et al. (1 February 2012). "Plasma and brain pharmacokinetic profile of cannabidiol (CBD), cannabidivarine (CBDV), Δ9-tetrahydrocannabivarin (THCV) and cannabigerol (CBG) in rats and mice following oral and intraperitoneal administration and CBD action on obsessive–compulsive behaviour" (in en). Psychopharmacology 219 (3): 859–873. doi:10.1007/s00213-011-2415-0. ISSN 0033-3158. http://link.springer.com/10.1007/s00213-011-2415-0.
- ↑ Bolognini, D; Rock, Em; Cluny, Nl; Cascio, Mg; Limebeer, Cl; Duncan, M; Stott, Cg; Javid, Fa et al. (1 March 2013). "Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT 1A receptor activation: CBDA, 5-HT 1A receptor, emesis and nausea" (in en). British Journal of Pharmacology 168 (6): 1456–1470. doi:10.1111/bph.12043. PMC PMC3596650. PMID 23121618. https://onlinelibrary.wiley.com/doi/10.1111/bph.12043.
- ↑ McPartland, John M; Duncan, Marnie; Di Marzo, Vincenzo; Pertwee, Roger G (1 February 2015). "Are cannabidiol and Δ 9 -tetrahydrocannabivarin negative modulators of the endocannabinoid system? A systematic review: Mechanistic studies ( in vivo and ex vivo ) of CBD and THCV" (in en). British Journal of Pharmacology 172 (3): 737–753. doi:10.1111/bph.12944. PMC PMC4301686. PMID 25257544. https://onlinelibrary.wiley.com/doi/10.1111/bph.12944.
- ↑ Takeda, Shuso; Misawa, Koichiro; Yamamoto, Ikuo; Watanabe, Kazuhito (1 September 2008). "Cannabidiolic Acid as a Selective Cyclooxygenase-2 Inhibitory Component in Cannabis" (in en). Drug Metabolism and Disposition 36 (9): 1917–1921. doi:10.1124/dmd.108.020909. ISSN 0090-9556. http://dmd.aspetjournals.org/lookup/doi/10.1124/dmd.108.020909.
- ↑ Falk, A A; Hagberg, M T; Lof, A E; Wigaeus-Hjelm, E M; Wang, Z P (1 October 1990). "Uptake, distribution and elimination of alpha-pinene in man after exposure by inhalation." (in en). Scandinavian Journal of Work, Environment & Health 16 (5): 372–378. doi:10.5271/sjweh.1771. ISSN 0355-3140. http://www.sjweh.fi/show_abstract.php?abstract_id=1771.
- ↑ Falk, Agneta; Löf, Agneta; Hagberg, Mats; Wigaeus Hjelm, Ewa; Wang, Zhiping (1 September 1991). "Human exposure to 3-carene by inhalation: Toxicokinetics, effects on pulmonary function and occurrence of irritative and CNS symptoms" (in en). Toxicology and Applied Pharmacology 110 (2): 198–205. doi:10.1016/S0041-008X(05)80002-X. https://linkinghub.elsevier.com/retrieve/pii/S0041008X0580002X.
- ↑ Falk‐Filipsson, Agneta; Löf, Agneta; Hagberg, Mats; Hjelm, Ewa Wigaeus; Wang, Zhiping (1 January 1993). "d‐Limonene exposure to humans by inhalation: Uptake, distribution, elimination, and effects on the pulmonary function" (in en). Journal of Toxicology and Environmental Health 38 (1): 77–88. doi:10.1080/15287399309531702. ISSN 0098-4108. http://www.tandfonline.com/doi/abs/10.1080/15287399309531702.
- ↑ Buchbauer, Gerhard; Jirovetz, Leopold; Jäger, Walter; Plank, Christine; Dietrich, Hermann (1 June 1993). "Fragrance Compounds and Essential Oils with Sedative Effects upon Inhalation" (in en). Journal of Pharmaceutical Sciences 82 (6): 660–664. doi:10.1002/jps.2600820623. https://linkinghub.elsevier.com/retrieve/pii/S0022354915491518.
- ↑ 72.0 72.1 72.2 72.3 Buchbauer, Gerhard (28 December 2009), Can Baser, K; Buchbauer, Gerhard, eds., "Biological Activities of Essential Oils" (in en), Handbook of Essential Oils (CRC Press): 235–280, doi:10.1201/9781420063165-c9, ISBN 978-1-4200-6315-8, http://www.crcnetbase.com/doi/10.1201/9781420063165-c9
- ↑ Bowles, E. Joy (2003). The chemistry of aromatherapeutic oils (3rd ed ed.). Crows Nest, N.S.W: Allen & Unwin. ISBN 978-1-74114-051-4. OCLC 249327567. https://www.worldcat.org/title/mediawiki/oclc/249327567.
- ↑ 74.0 74.1 Wedman-St. Louis, Betty (2019). Cannabis as medicine. Boca Raton: Taylor & Francis. ISBN 978-0-367-15054-9.
- ↑ Salehi, Bahare; Upadhyay, Shashi; Erdogan Orhan, Ilkay; Kumar Jugran, Arun; L.D. Jayaweera, Sumali; A. Dias, Daniel; Sharopov, Farukh; Taheri, Yasaman et al. (14 November 2019). "Therapeutic Potential of α- and β-Pinene: A Miracle Gift of Nature" (in en). Biomolecules 9 (11): 738. doi:10.3390/biom9110738. ISSN 2218-273X. PMC PMC6920849. PMID 31739596. https://www.mdpi.com/2218-273X/9/11/738.
- ↑ Gertsch, Jürg (1 July 2008). "Antiinflammatory cannabinoids in diet – towards a better understanding of CB2 receptor action?: Towards a better understanding of CB 2 receptor action?" (in en). Communicative & Integrative Biology 1 (1): 26–28. doi:10.4161/cib.1.1.6568. ISSN 1942-0889. PMC PMC2633791. PMID 19704783. http://www.tandfonline.com/doi/abs/10.4161/cib.1.1.6568.
- ↑ Vetter, Walter; Schröder, Markus; Lehnert, Katja (20 June 2012). "Differentiation of Refined and Virgin Edible Oils by Means of the trans - and cis -Phytol Isomer Distribution" (in en). Journal of Agricultural and Food Chemistry 60 (24): 6103–6107. doi:10.1021/jf301373k. ISSN 0021-8561. https://pubs.acs.org/doi/10.1021/jf301373k.
- ↑ Aluko, R.E. (2017), "Hemp Seed (Cannabis sativa L.) Proteins" (in en), Sustainable Protein Sources (Elsevier): 121–132, doi:10.1016/b978-0-12-802778-3.00007-x, ISBN 978-0-12-802778-3, https://linkinghub.elsevier.com/retrieve/pii/B978012802778300007X
- ↑ Guang, H.; Wenwei, C. (19 May 2010). "Application of powder of whole cannabis sativa seeds for preparing functional food with adjuvant therapy of lowering blood fat". Google Patents. Yunnan Industrial Hemp Co. Ltd.. https://patents.google.com/patent/CN100998414B/en.
- ↑ 80.0 80.1 80.2 Rupasinghe, H. P. Vasantha; Davis, Amy; Kumar, Shanthanu K.; Murray, Beth; Zheljazkov, Valtcho D. (7 September 2020). "Industrial Hemp (Cannabis sativa subsp. sativa) as an Emerging Source for Value-Added Functional Food Ingredients and Nutraceuticals" (in en). Molecules 25 (18): 4078. doi:10.3390/molecules25184078. ISSN 1420-3049. PMC PMC7571072. PMID 32906622. https://www.mdpi.com/1420-3049/25/18/4078.
- ↑ 81.0 81.1 Ross, Samir A.; Mehmedic, Zlatko; Murphy, Timothy P.; ElSohly, Mahmoud A. (1 November 2000). "GC-MS Analysis of the Total Δ9-THC Content of Both Drug- and Fiber-Type Cannabis Seeds" (in en). Journal of Analytical Toxicology 24 (8): 715–717. doi:10.1093/jat/24.8.715. ISSN 1945-2403. http://academic.oup.com/jat/article/24/8/715/817709/GCMS-Analysis-of-the-Total-Δ9THC-Content-of-Both.
- ↑ Federal Institute for Consumer Health Protection and Veterinary Medicine (BgVV) (22 October 1997). "Einsatz von Hanf in Lebensmitteln kann gesundheitlich problematisch sein". Bundesinstitut für Risikobewertung. https://www.bfr.bund.de/de/presseinformation/1997/26/einsatz_von_hanf_in_lebensmitteln_kann_gesundheitlich_problematisch_sein-829.html.
- ↑ Rodriguez-Leyva, Delfin; Pierce, Grant N (2010). "The cardiac and haemostatic effects of dietary hempseed" (in en). Nutrition & Metabolism 7 (1): 32. doi:10.1186/1743-7075-7-32. ISSN 1743-7075. PMC PMC2868018. PMID 20409317. http://nutritionandmetabolism.biomedcentral.com/articles/10.1186/1743-7075-7-32.
- ↑ Irakli, Maria; Tsaliki, Eleni; Kalivas, Apostolos; Kleisiaris, Fotios; Sarrou, Eirini; Cook, Catherine M (17 October 2019). "Effect οf Genotype and Growing Year on the Nutritional, Phytochemical, and Antioxidant Properties of Industrial Hemp (Cannabis sativa L.) Seeds" (in en). Antioxidants 8 (10): 491. doi:10.3390/antiox8100491. ISSN 2076-3921. PMC PMC6826498. PMID 31627349. https://www.mdpi.com/2076-3921/8/10/491.
- ↑ 85.0 85.1 Leizer, Cary; Ribnicky, David; Poulev, Alexander; Dushenkov, Slavik; Raskin, Ilya (1 December 2000). "The Composition of Hemp Seed Oil and Its Potential as an Important Source of Nutrition" (in en). Journal of Nutraceuticals, Functional & Medical Foods 2 (4): 35–53. doi:10.1300/J133v02n04_04. ISSN 1089-4179. http://www.tandfonline.com/doi/full/10.1300/J133v02n04_04.
- ↑ 86.0 86.1 Dunford, N.T. (2015), "Hemp and flaxseed oil" (in en), Specialty Oils and Fats in Food and Nutrition (Elsevier): 39–63, doi:10.1016/b978-1-78242-376-8.00002-8, ISBN 978-1-78242-376-8, https://linkinghub.elsevier.com/retrieve/pii/B9781782423768000028
- ↑ Kriese, U.; Schumann, E.; Weber, W.E.; Beyer, M.; Brühl, L.; Matthäus, B. (1 June 2004). "Oil content, tocopherol composition and fatty acid patterns of the seeds of 51 Cannabis sativa L. genotypes" (in en). Euphytica 137 (3): 339–351. doi:10.1023/B:EUPH.0000040473.23941.76. ISSN 0014-2336. http://link.springer.com/10.1023/B:EUPH.0000040473.23941.76.
- ↑ Andre, Christelle M.; Hausman, Jean-Francois; Guerriero, Gea (4 February 2016). "Cannabis sativa: The Plant of the Thousand and One Molecules". Frontiers in Plant Science 7. doi:10.3389/fpls.2016.00019. ISSN 1664-462X. PMC PMC4740396. PMID 26870049. http://journal.frontiersin.org/Article/10.3389/fpls.2016.00019/abstract.
- ↑ Klingberg, Sofia; Ellegård, Lars; Johansson, Ingegerd; Jansson, Jan-Håkan; Hallmans, Göran; Winkvist, Anna (1 October 2013). "Dietary Intake of Naturally Occurring Plant Sterols Is Related to a Lower Risk of a First Myocardial Infarction in Men but Not in Women in Northern Sweden" (in en). The Journal of Nutrition 143 (10): 1630–1635. doi:10.3945/jn.113.178707. ISSN 0022-3166. https://academic.oup.com/jn/article/143/10/1630/4571618.
- ↑ Odani, Sumiko; Odani, Shoji (1 January 1998). "Isolation and Primary Structure of a Methionine- and Cystine-rich Seed Protein of Cannabis sativa" (in en). Bioscience, Biotechnology, and Biochemistry 62 (4): 650–654. doi:10.1271/bbb.62.650. ISSN 0916-8451. https://academic.oup.com/bbb/article/62/4/650-654/5947038.
- ↑ Callaway, J. C. (1 January 2004). "Hempseed as a nutritional resource: An overview" (in en). Euphytica 140 (1-2): 65–72. doi:10.1007/s10681-004-4811-6. ISSN 0014-2336. https://link.springer.com/10.1007/s10681-004-4811-6.
- ↑ Klein, R.F.X. (2017). "Analysis of “Marijuana Edibles” – Food Products Containing Marijuana or Marijuana Extracts – An Overview, Review, and Literature Survey". Microgram Journal 14 (1–4): 9–32. https://www.dea.gov/documents/2017/2017-01/2017-01-01/analysis-marijuana-edibles-food-products-containing-marijuana-or.
- ↑ European Commission. "Novel Food Catalogue". European Commission. https://food.ec.europa.eu/safety/novel-food/novel-food-catalogue_en.
- ↑ Cohen, Pieter A.; Sharfstein, Joshua (25 July 2019). "The Opportunity of CBD — Reforming the Law" (in en). New England Journal of Medicine 381 (4): 297–299. doi:10.1056/NEJMp1906409. ISSN 0028-4793. http://www.nejm.org/doi/10.1056/NEJMp1906409.
- ↑ 95.0 95.1 95.2 Namdar, Dvora; Anis, Omer; Poulin, Patrick; Koltai, Hinanit (20 October 2020). "Chronological Review and Rational and Future Prospects of Cannabis-Based Drug Development" (in en). Molecules 25 (20): 4821. doi:10.3390/molecules25204821. ISSN 1420-3049. PMC PMC7587964. PMID 33092255. https://www.mdpi.com/1420-3049/25/20/4821.
- ↑ 96.0 96.1 Pearlson, Godfrey (2020), "Medical marijuana and clinical trials" (in en), Weed Science (Elsevier): 243–260, doi:10.1016/b978-0-12-818174-4.00010-0, ISBN 978-0-12-818174-4, https://linkinghub.elsevier.com/retrieve/pii/B9780128181744000100
- ↑ Urits, Ivan; Borchart, Matthew; Hasegawa, Morgan; Kochanski, Justin; Orhurhu, Vwaire; Viswanath, Omar (1 June 2019). "An Update of Current Cannabis-Based Pharmaceuticals in Pain Medicine" (in en). Pain and Therapy 8 (1): 41–51. doi:10.1007/s40122-019-0114-4. ISSN 2193-8237. PMC PMC6514017. PMID 30721403. http://link.springer.com/10.1007/s40122-019-0114-4.
- ↑ 98.0 98.1 98.2 98.3 Institute of Medicine (US) (1999). Joy, Janet E.; Watson, Stanley J.; Benson, John A.. eds. Marijuana and Medicine: Assessing the Science Base. Washington (DC): National Academies Press (US). ISBN 978-0-309-07155-0. PMID 25101425. http://www.ncbi.nlm.nih.gov/books/NBK230716/.
- ↑ 99.0 99.1 99.2 99.3 Verma, Rishabh; Hoda, Farazul; Arshad, Mawrah; Iqubal, Asif; Siddiqui, Ali Nasir; Khan, Mohammad Ahmed; Haque, Syed Ehtaishamul; Akhtar, Mohd et al. (21 May 2021). "Cannabis, a Miracle Drug with Polyvalent Therapeutic Utility: Preclinical and Clinical-Based Evidence" (in en). Medical Cannabis and Cannabinoids 4 (1): 43–60. doi:10.1159/000515042. ISSN 2504-3889. PMC PMC8525216. PMID 34676349. https://www.karger.com/Article/FullText/515042.
- ↑ 100.0 100.1 Ebbert, Jon O.; Scharf, Eugene L.; Hurt, Ryan T. (1 December 2018). "Medical Cannabis" (in en). Mayo Clinic Proceedings 93 (12): 1842–1847. doi:10.1016/j.mayocp.2018.09.005. https://linkinghub.elsevier.com/retrieve/pii/S0025619618307092.
- ↑ 101.0 101.1 Fisher, Emma; Eccleston, Christopher; Degenhardt, Louisa; Finn, David P.; Finnerup, Nanna B.; Gilron, Ian; Haroutounian, Simon; Krane, Elliot et al. (1 May 2019). "Cannabinoids, cannabis, and cannabis-based medicine for pain management: a protocol for an overview of systematic reviews and a systematic review of randomised controlled trials" (in en). PAIN Reports 4 (3): e741. doi:10.1097/PR9.0000000000000741. ISSN 2471-2531. PMC PMC6749927. PMID 31583356. https://journals.lww.com/01938936-201906000-00001.
- ↑ 102.0 102.1 Badowski, Melissa E. (1 September 2017). "A review of oral cannabinoids and medical marijuana for the treatment of chemotherapy-induced nausea and vomiting: a focus on pharmacokinetic variability and pharmacodynamics" (in en). Cancer Chemotherapy and Pharmacology 80 (3): 441–449. doi:10.1007/s00280-017-3387-5. ISSN 0344-5704. PMC PMC5573753. PMID 28780725. http://link.springer.com/10.1007/s00280-017-3387-5.
- ↑ 103.0 103.1 Musty, Richard E.; Rossi, Rita (1 January 2001). "Effects of Smoked Cannabis and Oral δ 9 -Tetrahydrocannabinol on Nausea and Emesis After Cancer Chemotherapy: A Review of State Clinical Trials" (in en). Journal of Cannabis Therapeutics 1 (1): 29–56. doi:10.1300/J175v01n01_03. ISSN 1529-9775. http://www.tandfonline.com/doi/abs/10.1300/J175v01n01_03.
- ↑ Ben Amar, Mohamed (1 April 2006). "Cannabinoids in medicine: A review of their therapeutic potential" (in en). Journal of Ethnopharmacology 105 (1-2): 1–25. doi:10.1016/j.jep.2006.02.001. https://linkinghub.elsevier.com/retrieve/pii/S0378874106000821.
- ↑ Mücke, Martin; Weier, Megan; Carter, Christopher; Copeland, Jan; Degenhardt, Louisa; Cuhls, Henning; Radbruch, Lukas; Häuser, Winfried et al. (1 April 2018). "Systematic review and meta-analysis of cannabinoids in palliative medicine: Cannabinoids in palliative medicine" (in en). Journal of Cachexia, Sarcopenia and Muscle 9 (2): 220–234. doi:10.1002/jcsm.12273. PMC PMC5879974. PMID 29400010. https://onlinelibrary.wiley.com/doi/10.1002/jcsm.12273.
- ↑ Whiting, Penny F.; Wolff, Robert F.; Deshpande, Sohan; Di Nisio, Marcello; Duffy, Steven; Hernandez, Adrian V.; Keurentjes, J. Christiaan; Lang, Shona et al. (23 June 2015). "Cannabinoids for Medical Use: A Systematic Review and Meta-analysis" (in en). JAMA 313 (24): 2456. doi:10.1001/jama.2015.6358. ISSN 0098-7484. http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.2015.6358.
- ↑ Gieringer, Dale; St. Laurent, Joseph; Goodrich, Scott (26 February 2004). "Cannabis Vaporizer Combines Efficient Delivery of THC with Effective Suppression of Pyrolytic Compounds" (in en). Journal of Cannabis Therapeutics 4 (1): 7–27. doi:10.1300/J175v04n01_02. ISSN 1529-9775. http://www.tandfonline.com/doi/abs/10.1300/J175v04n01_02.
- ↑ 108.0 108.1 Turgeman, Ilit; Bar-Sela, Gil (4 March 2019). "Cannabis for cancer – illusion or the tip of an iceberg: a review of the evidence for the use of Cannabis and synthetic cannabinoids in oncology" (in en). Expert Opinion on Investigational Drugs 28 (3): 285–296. doi:10.1080/13543784.2019.1561859. ISSN 1354-3784. https://www.tandfonline.com/doi/full/10.1080/13543784.2019.1561859.
- ↑ Corroon, Jamie; Kight, Rod (1 September 2018). "Regulatory Status of Cannabidiol in the United States: A Perspective" (in en). Cannabis and Cannabinoid Research 3 (1): 190–194. doi:10.1089/can.2018.0030. ISSN 2378-8763. PMC PMC6154432. PMID 30283822. https://www.liebertpub.com/doi/10.1089/can.2018.0030.
- ↑ 110.0 110.1 110.2 110.3 Balter, R.E; Haney, M. (2017), "The Synthetic Analog of Δ 9 -Tetrahydrocannabinol (THC): Nabilone. Pharmacology and Clinical Application" (in en), Handbook of Cannabis and Related Pathologies (Elsevier): 821–827, doi:10.1016/b978-0-12-800756-3.00099-5, ISBN 978-0-12-800756-3, https://linkinghub.elsevier.com/retrieve/pii/B9780128007563000995
- ↑ Taylor, Bryce N.; Mueller, Matthew; Sauls, Ryan S. (2022), "Cannaboinoid Antiemetic Therapy", StatPearls (Treasure Island (FL): StatPearls Publishing), PMID 30571051, http://www.ncbi.nlm.nih.gov/books/NBK535430/
- ↑ Cameron, Colin; Watson, Diane; Robinson, Jeffrey (1 October 2014). "Use of a Synthetic Cannabinoid in a Correctional Population for Posttraumatic Stress Disorder–Related Insomnia and Nightmares, Chronic Pain, Harm Reduction, and Other Indications: A Retrospective Evaluation" (in en). Journal of Clinical Psychopharmacology 34 (5): 559–564. doi:10.1097/JCP.0000000000000180. ISSN 0271-0749. PMC PMC4165471. PMID 24987795. https://journals.lww.com/00004714-201410000-00005.
- ↑ Sekar, Krithiga; Pack, Alison (28 February 2019). "Epidiolex as adjunct therapy for treatment of refractory epilepsy: a comprehensive review with a focus on adverse effects" (in en). F1000Research 8: 234. doi:10.12688/f1000research.16515.1. ISSN 2046-1402. PMC PMC6396837. PMID 30854190. https://f1000research.com/articles/8-234/v1.
- ↑ 114.0 114.1 114.2 114.3 Abu-Sawwa, Renad; Scutt, Brielle; Park, Yong (1 August 2020). "Emerging Use of Epidiolex (Cannabidiol) in Epilepsy" (in en). The Journal of Pediatric Pharmacology and Therapeutics 25 (6): 485–499. doi:10.5863/1551-6776-25.6.485. ISSN 1551-6776. PMC PMC7439947. PMID 32839652. https://meridian.allenpress.com/jppt/article/25/6/485/443971/Emerging-Use-of-Epidiolex-Cannabidiol-in-Epilepsy.
- ↑ Rosenkrantz, Harris; Fleischman, Robert W.; Grant, R.Jeffrey (1 March 1981). "Toxicity of short-term administration of cannabinoids to rhesus monkeys" (in en). Toxicology and Applied Pharmacology 58 (1): 118–131. doi:10.1016/0041-008X(81)90122-8. https://linkinghub.elsevier.com/retrieve/pii/0041008X81901228.
- ↑ Ewing, Laura E.; Skinner, Charles M.; Quick, Charles M.; Kennon-McGill, Stefanie; McGill, Mitchell R.; Walker, Larry A.; ElSohly, Mahmoud A.; Gurley, Bill J. et al. (30 April 2019). "Hepatotoxicity of a Cannabidiol-Rich Cannabis Extract in the Mouse Model" (in en). Molecules 24 (9): 1694. doi:10.3390/molecules24091694. ISSN 1420-3049. PMC PMC6539990. PMID 31052254. https://www.mdpi.com/1420-3049/24/9/1694.
- ↑ Huestis, Marilyn A.; Solimini, Renata; Pichini, Simona; Pacifici, Roberta; Carlier, Jeremy; Busardò, Francesco Paolo (13 September 2019). "Cannabidiol Adverse Effects and Toxicity" (in en). Current Neuropharmacology 17 (10): 974–989. doi:10.2174/1570159X17666190603171901. PMC PMC7052834. PMID 31161980. http://www.eurekaselect.com/172430/article.
- ↑ 118.0 118.1 Walker, Larry A.; Koturbash, Igor; Kingston, Rick; ElSohly, Mahmoud A.; Yates, Charles Ryan; Gurley, Bill J.; Khan, Ikhlas (2 September 2020). "Cannabidiol (CBD) in Dietary Supplements: Perspectives on Science, Safety, and Potential Regulatory Approaches" (in en). Journal of Dietary Supplements 17 (5): 493–502. doi:10.1080/19390211.2020.1777244. ISSN 1939-0211. https://www.tandfonline.com/doi/full/10.1080/19390211.2020.1777244.
- ↑ 119.0 119.1 119.2 Russo, Ethan B.; Guy, Geoffrey W.; Robson, Philip J. (1 August 2007). "Cannabis, Pain, and Sleep: Lessons from Therapeutic Clinical Trials ofSativex®, a Cannabis-Based Medicine" (in en). Chemistry & Biodiversity 4 (8): 1729–1743. doi:10.1002/cbdv.200790150. https://onlinelibrary.wiley.com/doi/10.1002/cbdv.200790150.
- ↑ 120.0 120.1 Keating, Gillian M. (1 April 2017). "Delta-9-Tetrahydrocannabinol/Cannabidiol Oromucosal Spray (Sativex®): A Review in Multiple Sclerosis-Related Spasticity" (in en). Drugs 77 (5): 563–574. doi:10.1007/s40265-017-0720-6. ISSN 0012-6667. http://link.springer.com/10.1007/s40265-017-0720-6.
- ↑ Rice, Jessica; Cameron, Michelle (1 August 2018). "Cannabinoids for Treatment of MS Symptoms: State of the Evidence" (in en). Current Neurology and Neuroscience Reports 18 (8): 50. doi:10.1007/s11910-018-0859-x. ISSN 1528-4042. http://link.springer.com/10.1007/s11910-018-0859-x.
- ↑ Fragoso, Yara Dadalti; Carra, Adriana; Macias, Miguel Angel (2 August 2020). "Cannabis and multiple sclerosis" (in en). Expert Review of Neurotherapeutics 20 (8): 849–854. doi:10.1080/14737175.2020.1776610. ISSN 1473-7175. https://www.tandfonline.com/doi/full/10.1080/14737175.2020.1776610.
- ↑ Committee on the Health Effects of Marijuana: An Evidence Review and Research Agenda; Board on Population Health and Public Health Practice; Health and Medicine Division; National Academies of Sciences, Engineering, and Medicine (31 March 2017). The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington, D.C.: National Academies Press. doi:10.17226/24625. ISBN 978-0-309-45304-2. https://www.nap.edu/catalog/24625.
- ↑ European Monitoring Centre for Drugs and Drug Addiction. (2018). Cannabis legislation in Europe: an overview.. LU: Publications Office. doi:10.2810/566650. https://data.europa.eu/doi/10.2810/566650.
- ↑ 125.0 125.1 125.2 Johnson, R. (22 June 2018). "Hemp as an Agricultural Commodity" (PDF). Congressional Research Service. https://sgp.fas.org/crs/misc/RL32725.pdf.
- ↑ 126.0 126.1 126.2 Glivar, Taja; Eržen, Jan; Kreft, Samo; Zagožen, Marjeta; Čerenak, Andreja; Čeh, Barbara; Tavčar Benković, Eva (1 March 2020). "Cannabinoid content in industrial hemp (Cannabis sativa L.) varieties grown in Slovenia" (in en). Industrial Crops and Products 145: 112082. doi:10.1016/j.indcrop.2019.112082. https://linkinghub.elsevier.com/retrieve/pii/S0926669019310921.
- ↑ 127.0 127.1 127.2 MacKay, R.' Phillips, K. (6 September 2016). "The Legal Regulation of Marijuana in Canada and Selected Other Countries" (PDF). Library of Parliament. https://lop.parl.ca/staticfiles/PublicWebsite/Home/ResearchPublications/BackgroundPapers/PDF/2016-94-e.pdf.
- ↑ Ghiabi, Maziyar; Maarefvand, Masoomeh; Bahari, Hamed; Alavi, Zohreh (1 June 2018). "Islam and cannabis: Legalisation and religious debate in Iran" (in en). International Journal of Drug Policy 56: 121–127. doi:10.1016/j.drugpo.2018.03.009. PMC PMC6153265. PMID 29635140. https://linkinghub.elsevier.com/retrieve/pii/S0955395918300793.
- ↑ International Narcotics Control Board (2017). Narcotic Drugs: Estimated World Requirements for 2017, Statistics for 2015. United Nations. pp. 350. ISBN 978-92-1-060062-0. https://www.incb.org/documents/Narcotic-Drugs/Technical-Publications/2016/Narcotic_Drugs_Publication_2016.pdf.
- ↑ 130.0 130.1 130.2 130.3 Stefkov, Gjoshe; Cvetkovikj Karanfilova, Ivana; Stoilkovska Gjorgievska, Veronika; Trajkovska, Ana; Geskovski, Nikola; Karapandzova, Marija; Kulevanova, Svetlana (1 February 2022). "Analytical Techniques for Phytocannabinoid Profiling of Cannabis and Cannabis-Based Products—A Comprehensive Review" (in en). Molecules 27 (3): 975. doi:10.3390/molecules27030975. ISSN 1420-3049. PMC PMC8838193. PMID 35164240. https://www.mdpi.com/1420-3049/27/3/975.
- ↑ 131.0 131.1 131.2 131.3 131.4 Citti, Cinzia; Braghiroli, Daniela; Vandelli, Maria Angela; Cannazza, Giuseppe (1 January 2018). "Pharmaceutical and biomedical analysis of cannabinoids: A critical review" (in en). Journal of Pharmaceutical and Biomedical Analysis 147: 565–579. doi:10.1016/j.jpba.2017.06.003. https://linkinghub.elsevier.com/retrieve/pii/S0731708517311895.
- ↑ Fischedick, Justin T.; Glas, Ronald; Hazekamp, Arno; Verpoorte, Rob (1 September 2009). "A qualitative and quantitative HPTLC densitometry method for the analysis of cannabinoids in Cannabis sativa L." (in en). Phytochemical Analysis 20 (5): 421–426. doi:10.1002/pca.1143. https://onlinelibrary.wiley.com/doi/10.1002/pca.1143.
- ↑ Dussy, Franz E.; Hamberg, Cornelia; Luginbühl, Marco; Schwerzmann, Thomas; Briellmann, Thomas A. (1 April 2005). "Isolation of Δ9-THCA-A from hemp and analytical aspects concerning the determination of Δ9-THC in cannabis products" (in en). Forensic Science International 149 (1): 3–10. doi:10.1016/j.forsciint.2004.05.015. https://linkinghub.elsevier.com/retrieve/pii/S0379073804003408.
- ↑ Kowalska, Teresa; Sajewicz, Mieczyslaw; Sherma, Joseph, eds. (18 November 2015) (in en). Planar Chromatography - Mass Spectrometry (0 ed.). CRC Press. doi:10.1201/b19090. ISBN 978-0-429-17193-2. https://www.taylorfrancis.com/books/9781498705899.
- ↑ Knezevic, Filip; Nikolai, Andreas; Marchart, Rudolf; Sosa, Silvio; Tubaro, Aurelia; Novak, Johannes (1 September 2021). "Residues of herbal hemp leaf teas – How much of the cannabinoids remain?" (in en). Food Control 127: 108146. doi:10.1016/j.foodcont.2021.108146. https://linkinghub.elsevier.com/retrieve/pii/S095671352100284X.
- ↑ 136.0 136.1 136.2 136.3 136.4 Pourseyed Lazarjani, Masoumeh; Torres, Stephanie; Hooker, Thom; Fowlie, Chris; Young, Owen; Seyfoddin, Ali (1 December 2020). "Methods for quantification of cannabinoids: a narrative review" (in en). Journal of Cannabis Research 2 (1): 35. doi:10.1186/s42238-020-00040-2. ISSN 2522-5782. PMC PMC7819317. PMID 33526084. https://jcannabisresearch.biomedcentral.com/articles/10.1186/s42238-020-00040-2.
- ↑ Hillig, Karl W.; Mahlberg, Paul G. (1 June 2004). "A chemotaxonomic analysis of cannabinoid variation in Cannabis (Cannabaceae)" (in en). American Journal of Botany 91 (6): 966–975. doi:10.3732/ajb.91.6.966. http://doi.wiley.com/10.3732/ajb.91.6.966.
- ↑ Leghissa, Allegra; Hildenbrand, Zacariah L.; Foss, Frank W.; Schug, Kevin A. (1 January 2018). "Determination of cannabinoids from a surrogate hops matrix using multiple reaction monitoring gas chromatography with triple quadrupole mass spectrometry" (in en). Journal of Separation Science 41 (2): 459–468. doi:10.1002/jssc.201700946. https://onlinelibrary.wiley.com/doi/10.1002/jssc.201700946.
- ↑ Leghissa, Allegra; Smuts, Jonathan; Qiu, Changling; Hildenbrand, Zacariah L.; Schug, Kevin A. (1 January 2018). "Detection of cannabinoids and cannabinoid metabolites using gas chromatography with vacuum ultraviolet spectroscopy" (in en). Separation Science Plus 1 (1): 37–42. doi:10.1002/sscp.201700005. https://onlinelibrary.wiley.com/doi/10.1002/sscp.201700005.
- ↑ Omar, Jone; Olivares, Maitane; Amigo, José Manuel; Etxebarria, Nestor (1 April 2014). "Resolution of co-eluting compounds of Cannabis Sativa in comprehensive two-dimensional gas chromatography/mass spectrometry detection with Multivariate Curve Resolution-Alternating Least Squares" (in en). Talanta 121: 273–280. doi:10.1016/j.talanta.2013.12.044. https://linkinghub.elsevier.com/retrieve/pii/S0039914013010370.
- ↑ Hazekamp, Arno; Peltenburg, Anja; Verpoorte, Rob; Giroud, Christian (1 September 2005). "Chromatographic and Spectroscopic Data of Cannabinoids from Cannabis sativa L." (in en). Journal of Liquid Chromatography & Related Technologies 28 (15): 2361–2382. doi:10.1080/10826070500187558. ISSN 1082-6076. https://www.tandfonline.com/doi/full/10.1080/10826070500187558.
- ↑ Citti, Cinzia; Ciccarella, Giuseppe; Braghiroli, Daniela; Parenti, Carlo; Vandelli, Maria Angela; Cannazza, Giuseppe (1 September 2016). "Medicinal cannabis: Principal cannabinoids concentration and their stability evaluated by a high performance liquid chromatography coupled to diode array and quadrupole time of flight mass spectrometry method" (in en). Journal of Pharmaceutical and Biomedical Analysis 128: 201–209. doi:10.1016/j.jpba.2016.05.033. https://linkinghub.elsevier.com/retrieve/pii/S0731708516302722.
- ↑ Peschel, Wieland; Politi, Matteo (1 August 2015). "1 H NMR and HPLC/DAD for Cannabis sativa L. chemotype distinction, extract profiling and specification" (in en). Talanta 140: 150–165. doi:10.1016/j.talanta.2015.02.040. https://linkinghub.elsevier.com/retrieve/pii/S0039914015001228.
- ↑ Giese, Matthew W; Lewis, Mark A; Giese, Laura; Smith, Kevin M (1 November 2015). "Method for the Analysis of Cannabinoids and Terpenes in Cannabis" (in en). Journal of AOAC INTERNATIONAL 98 (6): 1503–1522. doi:10.5740/jaoacint.15-116. ISSN 1060-3271. https://academic.oup.com/jaoac/article/98/6/1503-1522/5654599.
- ↑ Grauwiler, Sandra B.; Scholer, André; Drewe, Jürgen (1 May 2007). "Development of a LC/MS/MS method for the analysis of cannabinoids in human EDTA-plasma and urine after small doses of Cannabis sativa extracts" (in en). Journal of Chromatography B 850 (1-2): 515–522. doi:10.1016/j.jchromb.2006.12.045. https://linkinghub.elsevier.com/retrieve/pii/S1570023206010506.
- ↑ Aizpurua-Olaizola, Oier; Omar, Jone; Navarro, Patricia; Olivares, Maitane; Etxebarria, Nestor; Usobiaga, Aresatz (1 November 2014). "Identification and quantification of cannabinoids in Cannabis sativa L. plants by high performance liquid chromatography-mass spectrometry" (in en). Analytical and Bioanalytical Chemistry 406 (29): 7549–7560. doi:10.1007/s00216-014-8177-x. ISSN 1618-2642. http://link.springer.com/10.1007/s00216-014-8177-x.
- ↑ The United States Pharmacopeial Convention. "<561> Articles of Botanical Origin" (PDF). United States Pharmacopeia–National Formulary. The United States Pharmacopeial Convention. https://www.usp.org/sites/default/files/usp/document/our-work/DS/2015-dsc-chapters-561-616-1010-1092.pdf.
- ↑ 148.0 148.1 148.2 Chen, Xinlv; Wu, Xinyan; Luan, Tiangang; Jiang, Ruifen; Ouyang, Gangfeng (1 March 2021). "Sample preparation and instrumental methods for illicit drugs in environmental and biological samples: A review" (in en). Journal of Chromatography A 1640: 461961. doi:10.1016/j.chroma.2021.461961. https://linkinghub.elsevier.com/retrieve/pii/S0021967321000856.
- ↑ Valizadehderakhshan, Mehrab; Shahbazi, Abolghasem; Kazem-Rostami, Masoud; Todd, Matthew Scott; Bhowmik, Arnab; Wang, Lijun (23 April 2021). "Extraction of Cannabinoids from Cannabis sativa L. (Hemp)—Review" (in en). Agriculture 11 (5): 384. doi:10.3390/agriculture11050384. ISSN 2077-0472. https://www.mdpi.com/2077-0472/11/5/384.
- ↑ Karschner, Erin L; Swortwood-Gates, Madeleine J; Huestis, Marilyn A (1 July 2020). "Identifying and Quantifying Cannabinoids in Biological Matrices in the Medical and Legal Cannabis Era" (in en). Clinical Chemistry 66 (7): 888–914. doi:10.1093/clinchem/hvaa113. ISSN 0009-9147. https://academic.oup.com/clinchem/article/66/7/888/5867829.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. Some grammar and punctuation was cleaned up to improve readability. In some cases important information was missing from the references, and that information was added. The original lists citation alphabetically; this version, by design, lists them in order of appearance. The definition of hemp from the Agriculture Improvement Act of 2018 lacked a proper citation in the original; one was made for this version. The original is missing the citation for Fernandez et al. 2020; the correct citation was found and used for this version. The original is missing the citation for Stack 2021; the presumably correct citation was found and used for this version. The original is missing the citation for Selehi 2019; the correct citation was found and used for this version. The original is missing the citation for Joy et al. 2019; the correct citation was found and used for this version. The original is missing the citation for Keating 2017; the correct citation was found and used for this version. The original is missing the citation for USP <561> 2019; the correct citation was found and used for this version.