Difference between revisions of "Journal:Process variation detection using missing data in a multihospital community practice anatomic pathology laboratory"
Shawndouglas (talk | contribs) (Saving and adding more.) |
Shawndouglas (talk | contribs) (Saving and adding more.) |
||
| (One intermediate revision by the same user not shown) | |||
| Line 38: | Line 38: | ||
==Background== | ==Background== | ||
[[Sample (material)|Specimen]] identification errors in the [[laboratory]], from collection to sign off, are estimated to occur in 0.6% to 6% of cases, depending on the definition of error and the test phase studied.<ref name="NakhlehSurgical96">{{cite journal |title=Surgical pathology specimen identification and accessioning: A College of American Pathologists Q-Probes Study of 1,004,115 cases from 417 institutions |journal=Archives of Pathology and Laboratory Medicine |author=Nakhleh, R.E.; Zarbo, R.J. |volume=120 |issue=3 |pages=227–33 |year=1996 |pmid=8629896}}</ref><ref name="BanksAProp17">{{cite journal |title=A Proposed Set of Metrics to Reduce Patient Safety Risk From Within the Anatomic Pathology Laboratory |journal=Laboratory Medicine |author=Banks, P.; Brown, R.; Laslowski, A. et al. |volume=48 |issue=2 |pages=195–201 |year=2017 |doi=10.1093/labmed/lmw068 |pmid=28340232 |pmc=PMC5424539}}</ref><ref name="LayfieldSpec10">{{cite journal |title=Specimen labeling errors in surgical pathology: an 18-month experience |journal=American Journal of Clinical Pathology |author=Layfield, L.J.; Anderson, G.M. |volume=134 |issue=3 |pages=466-70 |year=2010 |doi=10.1309/AJCPHLQHJ0S3DFJK |pmid=20716804}}</ref><ref name="ZarboTHeHen09">{{cite journal |title=The Henry Ford Production System: reduction of surgical pathology in-process misidentification defects by bar code-specified work process standardization |journal=American Journal of Clinical Pathology |author=Zarbo, R.J.; Tuthill, J.M.; D'Angelo, R. et al. |volume=131 |issue=4 |pages=468-77 |year=2009 |doi=10.1309/AJCPPTJ3XJY6ZXDB |pmid=19289582}}</ref> Layfield and Anderson found that incorrect linkage of a specimen to a patient can comprise up to 73% of in-process errors.<ref name="LayfieldSpec10" /> One way to combat this is through [[barcode]]-driven histology processes, which been shown to be safer and reduce specimen identification errors.<ref name="ZarboTHeHen09" /> In fact, Zarbo ''et al.'' have shown that a barcode-enabled process can reduce laboratory misidentification errors by 62%.<ref name="ZarboTHeHen09" /> There are several reasons for this, including a reduction in typing, a reduced need for interpretation of hand-written numbers, and improved maintenance of the [[chain of custody]] of all the assets of the case, from receipt to sign off, better ensuring the produced pathology materials correspond to the patient when procedures are followed fully. However, barcode-enabled processes are only as good as their integration into the [[workflow]]s of the laboratory and use by the staff.<ref name="NakhlehCore09">{{cite journal |title=Core components of a comprehensive quality assurance program in anatomic pathology |journal=Advances in Anatomic Pathology |author=Nakhleh, R.E. |volume=16 |issue=6 |pages=418–23 |year=2009 |doi=10.1097/PAP.0b013e3181bb6bf7 |pmid=19851132}}</ref><ref name="HannaBar15">{{cite journal |title=Bar Coding and Tracking in Pathology |journal=Surgical Pathology Clinics |author=Hanna, M.G.; Pantanowitz, L. |volume=8 |issue=2 |pages=123–35 |year=2015 |doi=10.1016/j.path.2015.02.017 |pmid=26065787}}</ref> | [[Sample (material)|Specimen]] identification errors in the [[laboratory]], from collection to sign off, are estimated to occur in 0.6% to 6% of cases, depending on the definition of error and the test phase studied.<ref name="NakhlehSurgical96">{{cite journal |title=Surgical pathology specimen identification and accessioning: A College of American Pathologists Q-Probes Study of 1,004,115 cases from 417 institutions |journal=Archives of Pathology and Laboratory Medicine |author=Nakhleh, R.E.; Zarbo, R.J. |volume=120 |issue=3 |pages=227–33 |year=1996 |pmid=8629896}}</ref><ref name="BanksAProp17">{{cite journal |title=A Proposed Set of Metrics to Reduce Patient Safety Risk From Within the Anatomic Pathology Laboratory |journal=Laboratory Medicine |author=Banks, P.; Brown, R.; Laslowski, A. et al. |volume=48 |issue=2 |pages=195–201 |year=2017 |doi=10.1093/labmed/lmw068 |pmid=28340232 |pmc=PMC5424539}}</ref><ref name="LayfieldSpec10">{{cite journal |title=Specimen labeling errors in surgical pathology: an 18-month experience |journal=American Journal of Clinical Pathology |author=Layfield, L.J.; Anderson, G.M. |volume=134 |issue=3 |pages=466-70 |year=2010 |doi=10.1309/AJCPHLQHJ0S3DFJK |pmid=20716804}}</ref><ref name="ZarboTHeHen09">{{cite journal |title=The Henry Ford Production System: reduction of surgical pathology in-process misidentification defects by bar code-specified work process standardization |journal=American Journal of Clinical Pathology |author=Zarbo, R.J.; Tuthill, J.M.; D'Angelo, R. et al. |volume=131 |issue=4 |pages=468-77 |year=2009 |doi=10.1309/AJCPPTJ3XJY6ZXDB |pmid=19289582}}</ref> Layfield and Anderson found that incorrect linkage of a specimen to a patient can comprise up to 73% of in-process errors.<ref name="LayfieldSpec10" /> One way to combat this is through [[barcode]]-driven histology processes, which been shown to be safer and reduce specimen identification errors.<ref name="ZarboTHeHen09" /> In fact, Zarbo ''et al.'' have shown that a barcode-enabled process can reduce laboratory misidentification errors by 62%.<ref name="ZarboTHeHen09" /> There are several reasons for this, including a reduction in typing, a reduced need for interpretation of hand-written numbers, and improved maintenance of the [[chain of custody]] of all the assets of the case, from receipt to sign off, better ensuring the produced pathology materials correspond to the patient when procedures are followed fully. However, barcode-enabled processes are only as good as their integration into the [[workflow]]s of the laboratory and use by the staff.<ref name="NakhlehCore09">{{cite journal |title=Core components of a comprehensive quality assurance program in anatomic pathology |journal=Advances in Anatomic Pathology |author=Nakhleh, R.E. |volume=16 |issue=6 |pages=418–23 |year=2009 |doi=10.1097/PAP.0b013e3181bb6bf7 |pmid=19851132}}</ref><ref name="HannaBar15">{{cite journal |title=Bar Coding and Tracking in Pathology |journal=Surgical Pathology Clinics |author=Hanna, M.G.; Pantanowitz, L. |volume=8 |issue=2 |pages=123–35 |year=2015 |doi=10.1016/j.path.2015.02.017 |pmid=26065787}}</ref> | ||
Evaluation of workflows and processes in our anatomic pathology (AP) laboratory tend to be reactive or retrospective. The data points within an accessioned case or group of cases can be evaluated during root cause analyses (RCA) and may consume a significant amount of time, but that time is typically limited to the RCA and not necessarily routine or ongoing. Prospective evaluation and auditing of process variation and [[laboratory information system]] (LIS) integration into workflows may help identify performance improvement projects to enhance safety and identify areas that need focus. | |||
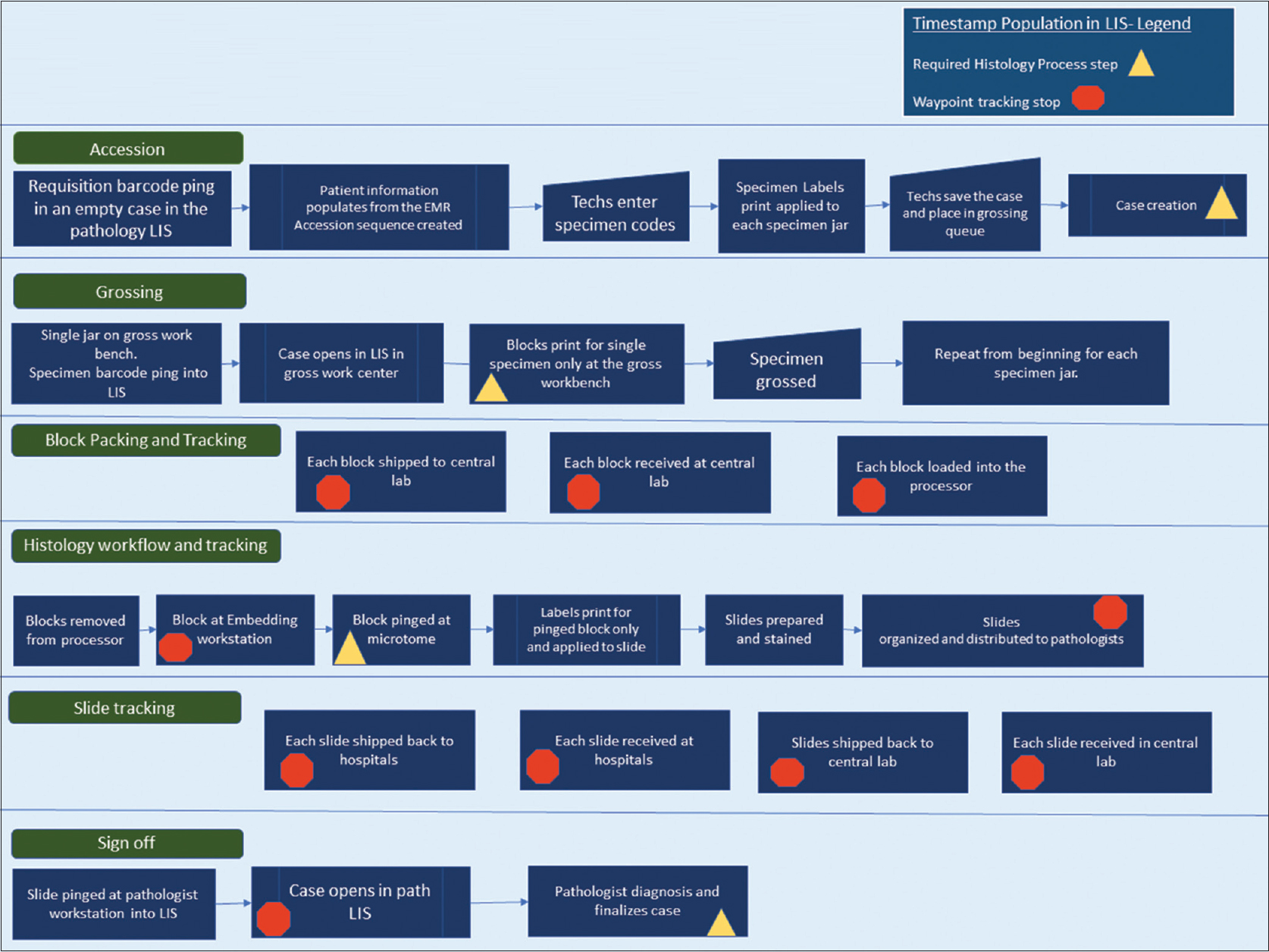
The individual pathology cases in our AP LIS contain two main types of timestamp data, each of which include date and time to the second. The first main type of timestamp data populates when a predefined workflow step or action is completed within the system. The case is then advanced to the next phase in the process in a pending state. As seen in Figure 1, the predefined process steps of the histology workflow include case creation (accession), grossing (gross worklist), histology preparation (slide creation after embedding), and case sign off (yellow triangles in Figure 1). | |||
[[File:Fig1 Galliano JofPathInfo2019 10.jpg|1200px]] | |||
{{clear}} | |||
{| | |||
| STYLE="vertical-align:top;"| | |||
{| border="0" cellpadding="5" cellspacing="0" width="1200px" | |||
|- | |||
| style="background-color:white; padding-left:10px; padding-right:10px;"| <blockquote>'''Figure 1.''' Process map of our pathology workflow</blockquote> | |||
|- | |||
|} | |||
|} | |||
The second main type of timestamp data is tracking data (internally defined as "waypoints"). Waypoint data are recorded when a barcoding action is performed for grossing, processor loads, embedding, histology, and sign out. Additional timestamps are populated when blocks and slides are shipped at the point of origin and at the point of receipt (orange polygons in Figure 1). All specimen labels and pathology materials contain a two-dimensional (2D) barcode, and the types of materials that are barcoded include specimen jar labels after accessioning, blocks, and slides. Expected timestamps are included in the process map shown in Figure 1. Each timestamp can be viewed within each case or called in bulk through various management reports. | |||
This project was developed to determine if evaluating cases with missing predefined process timestamps improved the ability to detect other data variations and procedure noncompliance in the AP workflow in a prospective fashion. This would potentially help identify workflows or facets of the laboratory information system that need focus for quality projects. | |||
==Materials and methods== | |||
Pathology information system raw timestamp data were extracted from the AP LIS (PathView Systems, Ltd. 2002–2019) for all surgical pathology cases in the Ochsner Health System from January 1, 2018 to December 15, 2018. The data table includes the following process, steps, and data points: accession number, collection date and time (inbound message from the [[electronic medical record]]), case creation date and time, grossing action timestamp, histology preparation timestamp (histology slide creation after embedding), and sign off. (See the Supplemental Material for an example of data table and case-level data). An additional waypoint timestamp is included in this raw data report, which marks the timestamp when histology has completed the case and is giving the case to the pathologist (slide distribution). The raw comma-separated value file data were analyzed for missing timestamp data ([[R (programming language)|statistical programming language R]] in R studio [R version 3.5.1]) primarily using homebrew coding scripts to process and filter the data. The <tt>naniar</tt> package was used, which was developed to visualize missing data.<ref name="RProj">{{cite web |url=https://www.r-project.org/ |title=The R Project for Statistical Computing |publisher=The R Foundation |accessdate=30 March 2019}}</ref><ref name="Tierney_naniar">{{cite web |url=http://naniar.njtierney.com/ |title=naniar |author=Tierney, N.; Cook, D.; McBain, M. et al. |work=Credibly Curious |date=2019}}</ref> (See Additional File 1 in the Supplemental Material). Subsets were created by type of data missing, focusing on the following: (1) cases with missing gross plus histology plus slide distribution timestamps (“all three”), (2) cases with grossing action missing alone, (3) cases with histology action missing alone, and (4) cases with slide distribution waypoint missing alone. Random samples of cases with 100% populated timestamps for all process steps were selected as a comparison group from the database using the base R random sample function. Subsets with >400 cases with missing data were also sampled using the random sample function. | |||
| Line 44: | Line 67: | ||
==Notes== | ==Notes== | ||
This presentation is faithful to the original, with only a few minor changes to presentation. Grammar was cleaned up for smoother reading. In some cases important information was missing from the references, and that information was added. | This presentation is faithful to the original, with only a few minor changes to presentation. Grammar was cleaned up for smoother reading. In some cases important information was missing from the references, and that information was added. At the time of loading of this article, the links to the Additional File 1 and 2 were broken on the original site; a request to fix the errors has been sent to the journal. | ||
<!--Place all category tags here--> | <!--Place all category tags here--> | ||
Revision as of 21:53, 4 November 2019
| Full article title | Process variation detection using missing data in a multihospital community practice anatomic pathology laboratory |
|---|---|
| Journal | Journal of Pathology Informatics |
| Author(s) | Galliano, Gretchen E. |
| Author affiliation(s) | Ochsner Health System |
| Primary contact | Email: login at original journal required |
| Year published | 2019 |
| Volume and issue | 10 |
| Page(s) | 25 |
| DOI | 10.4103/jpi.jpi_18_19 |
| ISSN | 2153-3539 |
| Distribution license | Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International |
| Website | http://www.jpathinformatics.org/ |
| Download | http://www.jpathinformatics.org/temp/JPatholInform10125-733533_202233.pdf (PDF) |
|
|
This article should be considered a work in progress and incomplete. Consider this article incomplete until this notice is removed. |
Abstract
Objectives: Barcode-driven workflows reduce patient identification errors. Missing process timestamp data frequently confound our health system's pending lists and appear as actions left undone. Anecdotally, it was noted that missing data could be found when there is procedure noncompliance. This project was developed to determine if missing timestamp data in the histology barcode-driven workflow correlated with other process variations, procedure noncompliance, or is an indicator of workflows needing focus for improvement projects.
Materials and methods: Data extracts of timestamp data from January 1, 2018 to December 15, 2018 for the major histology process steps were analyzed for missing data. Case-level analysis to determine the presence or absence of expected barcoding events was performed on 1031 surgical pathology cases to determine the cause of the missing data and determine if additional data variations or procedure noncompliance events were present. The data variations were classified according to a scheme defined in the study.
Results: Of 70,085 cases, there were 7218 (10.3%) with missing process timestamp data. Missing histology process step data was associated with other additional data variations in case-level deep dives (P < 0.0001). Of the cases missing timestamp data in the initial review, 18.4% of the cases had no identifiable cause for the missing data (all expected events took place in the case-level deep dive).
Conclusions: Operationally, valuable information can be obtained by reviewing the types and causes of missing data in the anatomic pathology laboratory information system, but only in conjunction with user input and feedback.
Keywords: anatomic pathology laboratory information system, anatomic pathology, laboratory information systems, laboratory management
Background
Specimen identification errors in the laboratory, from collection to sign off, are estimated to occur in 0.6% to 6% of cases, depending on the definition of error and the test phase studied.[1][2][3][4] Layfield and Anderson found that incorrect linkage of a specimen to a patient can comprise up to 73% of in-process errors.[3] One way to combat this is through barcode-driven histology processes, which been shown to be safer and reduce specimen identification errors.[4] In fact, Zarbo et al. have shown that a barcode-enabled process can reduce laboratory misidentification errors by 62%.[4] There are several reasons for this, including a reduction in typing, a reduced need for interpretation of hand-written numbers, and improved maintenance of the chain of custody of all the assets of the case, from receipt to sign off, better ensuring the produced pathology materials correspond to the patient when procedures are followed fully. However, barcode-enabled processes are only as good as their integration into the workflows of the laboratory and use by the staff.[5][6]
Evaluation of workflows and processes in our anatomic pathology (AP) laboratory tend to be reactive or retrospective. The data points within an accessioned case or group of cases can be evaluated during root cause analyses (RCA) and may consume a significant amount of time, but that time is typically limited to the RCA and not necessarily routine or ongoing. Prospective evaluation and auditing of process variation and laboratory information system (LIS) integration into workflows may help identify performance improvement projects to enhance safety and identify areas that need focus.
The individual pathology cases in our AP LIS contain two main types of timestamp data, each of which include date and time to the second. The first main type of timestamp data populates when a predefined workflow step or action is completed within the system. The case is then advanced to the next phase in the process in a pending state. As seen in Figure 1, the predefined process steps of the histology workflow include case creation (accession), grossing (gross worklist), histology preparation (slide creation after embedding), and case sign off (yellow triangles in Figure 1).
|
The second main type of timestamp data is tracking data (internally defined as "waypoints"). Waypoint data are recorded when a barcoding action is performed for grossing, processor loads, embedding, histology, and sign out. Additional timestamps are populated when blocks and slides are shipped at the point of origin and at the point of receipt (orange polygons in Figure 1). All specimen labels and pathology materials contain a two-dimensional (2D) barcode, and the types of materials that are barcoded include specimen jar labels after accessioning, blocks, and slides. Expected timestamps are included in the process map shown in Figure 1. Each timestamp can be viewed within each case or called in bulk through various management reports.
This project was developed to determine if evaluating cases with missing predefined process timestamps improved the ability to detect other data variations and procedure noncompliance in the AP workflow in a prospective fashion. This would potentially help identify workflows or facets of the laboratory information system that need focus for quality projects.
Materials and methods
Pathology information system raw timestamp data were extracted from the AP LIS (PathView Systems, Ltd. 2002–2019) for all surgical pathology cases in the Ochsner Health System from January 1, 2018 to December 15, 2018. The data table includes the following process, steps, and data points: accession number, collection date and time (inbound message from the electronic medical record), case creation date and time, grossing action timestamp, histology preparation timestamp (histology slide creation after embedding), and sign off. (See the Supplemental Material for an example of data table and case-level data). An additional waypoint timestamp is included in this raw data report, which marks the timestamp when histology has completed the case and is giving the case to the pathologist (slide distribution). The raw comma-separated value file data were analyzed for missing timestamp data (statistical programming language R in R studio [R version 3.5.1]) primarily using homebrew coding scripts to process and filter the data. The naniar package was used, which was developed to visualize missing data.[7][8] (See Additional File 1 in the Supplemental Material). Subsets were created by type of data missing, focusing on the following: (1) cases with missing gross plus histology plus slide distribution timestamps (“all three”), (2) cases with grossing action missing alone, (3) cases with histology action missing alone, and (4) cases with slide distribution waypoint missing alone. Random samples of cases with 100% populated timestamps for all process steps were selected as a comparison group from the database using the base R random sample function. Subsets with >400 cases with missing data were also sampled using the random sample function.
References
- ↑ Nakhleh, R.E.; Zarbo, R.J. (1996). "Surgical pathology specimen identification and accessioning: A College of American Pathologists Q-Probes Study of 1,004,115 cases from 417 institutions". Archives of Pathology and Laboratory Medicine 120 (3): 227–33. PMID 8629896.
- ↑ Banks, P.; Brown, R.; Laslowski, A. et al. (2017). "A Proposed Set of Metrics to Reduce Patient Safety Risk From Within the Anatomic Pathology Laboratory". Laboratory Medicine 48 (2): 195–201. doi:10.1093/labmed/lmw068. PMC PMC5424539. PMID 28340232. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5424539.
- ↑ 3.0 3.1 Layfield, L.J.; Anderson, G.M. (2010). "Specimen labeling errors in surgical pathology: an 18-month experience". American Journal of Clinical Pathology 134 (3): 466-70. doi:10.1309/AJCPHLQHJ0S3DFJK. PMID 20716804.
- ↑ 4.0 4.1 4.2 Zarbo, R.J.; Tuthill, J.M.; D'Angelo, R. et al. (2009). "The Henry Ford Production System: reduction of surgical pathology in-process misidentification defects by bar code-specified work process standardization". American Journal of Clinical Pathology 131 (4): 468-77. doi:10.1309/AJCPPTJ3XJY6ZXDB. PMID 19289582.
- ↑ Nakhleh, R.E. (2009). "Core components of a comprehensive quality assurance program in anatomic pathology". Advances in Anatomic Pathology 16 (6): 418–23. doi:10.1097/PAP.0b013e3181bb6bf7. PMID 19851132.
- ↑ Hanna, M.G.; Pantanowitz, L. (2015). "Bar Coding and Tracking in Pathology". Surgical Pathology Clinics 8 (2): 123–35. doi:10.1016/j.path.2015.02.017. PMID 26065787.
- ↑ "The R Project for Statistical Computing". The R Foundation. https://www.r-project.org/. Retrieved 30 March 2019.
- ↑ Tierney, N.; Cook, D.; McBain, M. et al. (2019). "naniar". Credibly Curious. http://naniar.njtierney.com/.
Notes
This presentation is faithful to the original, with only a few minor changes to presentation. Grammar was cleaned up for smoother reading. In some cases important information was missing from the references, and that information was added. At the time of loading of this article, the links to the Additional File 1 and 2 were broken on the original site; a request to fix the errors has been sent to the journal.