Difference between revisions of "LIMS feature"
Shawndouglas (talk | contribs) m (→Audit trail: Flowlist) |
Shawndouglas (talk | contribs) m (Red links) |
||
| (23 intermediate revisions by 2 users not shown) | |||
| Line 2: | Line 2: | ||
| type = notice | | type = notice | ||
| style = width: 960px; | | style = width: 960px; | ||
| text = '''You can find a listing''' of all LIMS vendors—and by extension, the features their products offer—on the [[:LIMS vendor|LIMS vendor page]]. | | text = '''You can find a listing''' of all LIMS vendors—and by extension, the features their products offer—on the [[:Vendor:LIMS vendor|LIMS vendor page]]. | ||
}} | }} | ||
A '''LIMS feature''' is one or more pieces of functionality that appear within a [[laboratory information management system]] (LIMS). | A '''LIMS feature''' is one or more pieces of functionality that appear within a [[laboratory information management system]] (LIMS). | ||
The LIMS is an evolving concept, with new features and abilities being introduced every year. As laboratory demands change and technological progress continues, the functions of a LIMS will also change. Yet like the automobile, the LIMS tends to have a base set of functionality that defines it. That functionality can roughly be divided into five laboratory processing phases, with numerous software functions falling under each<ref name=MT5310>{{cite journal|journal=Measurement Techniques |year=2011 |volume=53 |issue=10 |pages=1182–1189 |title=Laboratory information management systems in the work of the analytic laboratory |author=Skobelev, D.O.; T.M. Zaytseva; A.D. Kozlov; V.L. Perepelitsa; A.S. Makarova |doi=10.1007/s11018-011-9638-7}}</ref>: | The LIMS is an evolving concept, with new features and abilities being introduced every year. As laboratory demands change and technological progress continues, the functions of a LIMS will also change. Yet like the automobile, the LIMS tends to have a base set of functionality that defines it. That functionality can roughly be divided into five laboratory processing phases, with numerous software functions falling under each<ref name="MT5310">{{cite journal|journal=Measurement Techniques |year=2011 |volume=53 |issue=10 |pages=1182–1189 |title=Laboratory information management systems in the work of the analytic laboratory |author=Skobelev, D.O.; T.M. Zaytseva; A.D. Kozlov; V.L. Perepelitsa; A.S. Makarova |doi=10.1007/s11018-011-9638-7}}</ref>: | ||
* the reception and log in of a sample and its associated customer data | * the reception and log in of a sample and its associated customer data; | ||
* the assignment, scheduling, and tracking of the sample and the associated analytical workload | * the assignment, scheduling, and tracking of the sample and the associated analytical workload; | ||
* the processing and quality control associated with the sample and the utilized equipment and inventory | * the processing and quality control associated with the sample and the utilized equipment and inventory; | ||
* the storage of data associated with the sample analysis | * the storage of data associated with the sample analysis; and | ||
* the inspection, approval, and compilation of the sample data for reporting and/or further analysis | * the inspection, approval, and compilation of the sample data for reporting and/or further analysis. | ||
Of course, there are LIMS features that are difficult to categorize under any of these phases. Such features often contribute to the entire LIMS and how it's utilized. For example, multilingual support appears in LIMS like [[Assaynet Inc.|Assaynet's]] Assaynet LIMS and [[SLCLAB Informática SL|SLCLAB Informática's]] ALFA21, allowing users to interact with the LIMS in more than one language. Some functionality may also overlap several laboratory phases, making it difficult to firmly classify. | Of course, there are LIMS features that are difficult to categorize under any of these phases. Such features often contribute to the entire LIMS and how it's utilized. For example, multilingual support appears in LIMS like [[Vendor:Assaynet Inc.|Assaynet's]] Assaynet LIMS and [[Vendor:SLCLAB Informática SL|SLCLAB Informática's]] ALFA21, allowing users to interact with the LIMS in more than one language. Some functionality may also overlap several laboratory phases, making it difficult to firmly classify. | ||
The features described below come from an analysis of freely available LIMS product information on vendor websites. An attempt was made to discover the features most utilized in vendors' LIMS products and collect information on those features for each LIMS. Not every possible feature is referenced here; some LIMS products fill specific niches, utilizing unique functionality to solve a specific problem. | The features described below come from an analysis of freely available LIMS product information on vendor websites. An attempt was made to discover the features most utilized in vendors' LIMS products and collect information on those features for each LIMS. Not every possible feature is referenced here; some LIMS products fill specific niches, utilizing unique functionality to solve a specific problem. | ||
| Line 58: | Line 58: | ||
===Task and event scheduling=== | ===Task and event scheduling=== | ||
Within the context of a LIMS, the ability to schedule a task or event is a natural extension of how work was done in a laboratory before the advent of data management systems. Workloads are assigned to technicians, maintenance schedules are created and followed, and research deadlines must be observed. While these tasks have in the past been performed without LIMS, a modern data information management system can now optimize those tasks and provide additional scheduling functionality to streamline the operation of a lab. [[Autoscribe Ltd.]], for example, offers a scheduling module for its LIMS that allows users to automatically schedule multiple jobs, data backups, alarms, and reports.<ref name="ASSched">{{cite web |url=https://www.autoscribeinformatics.com/resources/press-releases/easy-scheduling-of-lims-activities |title=Easy scheduling of LIMS activities |publisher=Autoscribe Ltd |accessdate=13 March 2020}}</ref> Some LIMS like [[LabWare, Inc.|LabWare, Inc.'s]] LabWare LIMS offer multiple types of schedulers that match to the particular functions of a research laboratory.<ref name="LWSched">{{cite web |url=https://www.labware.com/en/p/Products/Summary-of-Core-Product-Features |archiveurl=https://web.archive.org/web/20200512174255/https://www.labware.com/en/p/Products/Summary-of-Core-Product-Features |title=Summary of Core Product Features |publisher=LabWare, Inc |archivedate=12 May 2020 |accessdate=06 January 2022}}</ref> Additional functionality within this feature includes the ability to configure automated assignments of analysis requests, establish recurring events, and in most cases, create printable schedules. | Within the context of a LIMS, the ability to schedule a task or event is a natural extension of how work was done in a laboratory before the advent of data management systems. Workloads are assigned to technicians, maintenance schedules are created and followed, and research deadlines must be observed. While these tasks have in the past been performed without LIMS, a modern data information management system can now optimize those tasks and provide additional scheduling functionality to streamline the operation of a lab. [[Vendor:Autoscribe Ltd.|Autoscribe Ltd.]], for example, offers a scheduling module for its LIMS that allows users to automatically schedule multiple jobs, data backups, alarms, and reports.<ref name="ASSched">{{cite web |url=https://www.autoscribeinformatics.com/resources/press-releases/easy-scheduling-of-lims-activities |title=Easy scheduling of LIMS activities |publisher=Autoscribe Ltd |accessdate=13 March 2020}}</ref> Some LIMS like [[Vendor:LabWare, Inc.|LabWare, Inc.'s]] LabWare LIMS offer multiple types of schedulers that match to the particular functions of a research laboratory.<ref name="LWSched">{{cite web |url=https://www.labware.com/en/p/Products/Summary-of-Core-Product-Features |archiveurl=https://web.archive.org/web/20200512174255/https://www.labware.com/en/p/Products/Summary-of-Core-Product-Features |title=Summary of Core Product Features |publisher=LabWare, Inc |archivedate=12 May 2020 |accessdate=06 January 2022}}</ref> Additional functionality within this feature includes the ability to configure automated assignments of analysis requests, establish recurring events, and in most cases, create printable schedules. | ||
Examples of tasks and events that can feasibly be scheduled in a LIMS include: | Examples of tasks and events that can feasibly be scheduled in a LIMS include: | ||
| Line 76: | Line 76: | ||
Laboratories produce data, and LIMS exist to help manage that data. Additionally, even before the existence of LIMS, scientists have had a corresponding need for visually representing data. Today, a LIMS can not only collect and analyze data from samples, but it also can represent that data in reports, graphs, gradients, and spreadsheets. Depending on the LIMS, more than one way to visually represent the data may exist. | Laboratories produce data, and LIMS exist to help manage that data. Additionally, even before the existence of LIMS, scientists have had a corresponding need for visually representing data. Today, a LIMS can not only collect and analyze data from samples, but it also can represent that data in reports, graphs, gradients, and spreadsheets. Depending on the LIMS, more than one way to visually represent the data may exist. | ||
Some laboratory information management systems take a specialized approach to data views. For example, [[Biomatters Ltd.|Biomatters Ltd.'s]] Geneious offers multiple methods of viewing complicated sequence analysis data, including 3-D structuring and representations of plasmid vectors, as well as other [[DNA sequencing#High-throughput methods|next-generation sequencing]] (NGS) activities.<ref name="BioView">{{cite web |url=https://www.geneious.com/features/ngs-visualization-downstream-analysis/ |title=NGS Visualization and Downstream Analysis |publisher=Biomatters Ltd |accessdate=13 March 2020}}</ref> | Some laboratory information management systems take a specialized approach to data views. For example, [[Vendor:Biomatters Ltd.|Biomatters Ltd.'s]] Geneious offers multiple methods of viewing complicated sequence analysis data, including 3-D structuring and representations of plasmid vectors, as well as other [[DNA sequencing#High-throughput methods|next-generation sequencing]] (NGS) activities.<ref name="BioView">{{cite web |url=https://www.geneious.com/features/ngs-visualization-downstream-analysis/ |title=NGS Visualization and Downstream Analysis |publisher=Biomatters Ltd |accessdate=13 March 2020}}</ref> | ||
===Data and trend analysis=== | ===Data and trend analysis=== | ||
| Line 108: | Line 108: | ||
===Query capability=== | ===Query capability=== | ||
[[File:QueryBuilder.png|thumb|240px|Advanced query tools allow researchers to better complete project objectives.]]As was the case before the advent of databases and electronic data management solutions, today researchers must search through sample results, experiment notes, and other types of data to better draw conclusions about their research. Whereas this used to mean browsing through laboratory notebooks, Excel spreadsheets, or Access databases, now powerful query tools exists within data management tools like a LIMS. Not only can data be searched for based on name, number, or vendor, LIMSs like [[Abbott Informatics Corporation|Abbott's]] STARLIMS and [[Mountain States Consulting, LLC|Mountain States Consulting's]] MSC-LIMS allow for queries of attached [[metadata]] like user ID, project number, task number, sample type, location, and collection date.<ref name="AbbottStarlimsPDF">{{cite web |url=https://www.informatics.abbott/shared/biorepository-lims-spec-document.pdf |archiveurl=https://web.archive.org/web/20170810191058/https://www.informatics.abbott/shared/biorepository-lims-spec-document.pdf |format=PDF |title=STARLIMS Biorepository Industry LIMS Specification Document |publisher=Abbott Informatics Corporation |date=18 September 2017 |archivedate=10 August 2017 |accessdate=06 January 2022}}</ref><ref name="MSCFeats">{{cite web |url=http://www.msc-lims.com/lims/msc-lims.html |title=MSC-LIMS Product Summary |publisher=Mountain States Consulting, LLC |accessdate=17 March 2020}}</ref> Finally, as LIMS continue to include both sample management and experimental data management functionality, queries become more powerful in general as now sample and experiment can be matched together in one database.<ref name="AE0302" /> | [[File:QueryBuilder.png|thumb|240px|Advanced query tools allow researchers to better complete project objectives.]]As was the case before the advent of databases and electronic data management solutions, today researchers must search through sample results, experiment notes, and other types of data to better draw conclusions about their research. Whereas this used to mean browsing through laboratory notebooks, Excel spreadsheets, or Access databases, now powerful query tools exists within data management tools like a LIMS. Not only can data be searched for based on name, number, or vendor, LIMSs like [[Vendor:Abbott Informatics Corporation|Abbott's]] STARLIMS and [[Vendor:Mountain States Consulting, LLC|Mountain States Consulting's]] MSC-LIMS allow for queries of attached [[metadata]] like user ID, project number, task number, sample type, location, and collection date.<ref name="AbbottStarlimsPDF">{{cite web |url=https://www.informatics.abbott/shared/biorepository-lims-spec-document.pdf |archiveurl=https://web.archive.org/web/20170810191058/https://www.informatics.abbott/shared/biorepository-lims-spec-document.pdf |format=PDF |title=STARLIMS Biorepository Industry LIMS Specification Document |publisher=Abbott Informatics Corporation |date=18 September 2017 |archivedate=10 August 2017 |accessdate=06 January 2022}}</ref><ref name="MSCFeats">{{cite web |url=http://www.msc-lims.com/lims/msc-lims.html |title=MSC-LIMS Product Summary |publisher=Mountain States Consulting, LLC |accessdate=17 March 2020}}</ref> Finally, as LIMS continue to include both sample management and experimental data management functionality, queries become more powerful in general as now sample and experiment can be matched together in one database.<ref name="AE0302" /> | ||
Query functionality often includes the ability to: | Query functionality often includes the ability to: | ||
| Line 121: | Line 121: | ||
Data can originate from numerous places in the laboratory. The ability to import that data into a LIMS can be beneficial, especially when an instrument can't be connected or an external client provides a data feed independent of the LIMS. Some LIMS may even allow users to cross-reference laboratory nomenclature from received data sources with the recipient's. And of course [[LIMS feature#Instrument interfacing and management|instrument interfacing]] allows for even more importation options. Additional [[LIMS_feature#Data validation|data validation]] procedures may be applied to the imported data to guarantee information homogeneity. Some LIMS may also allow for the import of non-normalized legacy data tables and integration with LIMS data tables to form a single unified database. | Data can originate from numerous places in the laboratory. The ability to import that data into a LIMS can be beneficial, especially when an instrument can't be connected or an external client provides a data feed independent of the LIMS. Some LIMS may even allow users to cross-reference laboratory nomenclature from received data sources with the recipient's. And of course [[LIMS feature#Instrument interfacing and management|instrument interfacing]] allows for even more importation options. Additional [[LIMS_feature#Data validation|data validation]] procedures may be applied to the imported data to guarantee information homogeneity. Some LIMS may also allow for the import of non-normalized legacy data tables and integration with LIMS data tables to form a single unified database. | ||
However, potential buyers should ensure the LIMS vendor is clear in how their system manages data imports. For example, products such as [[AgileBio]]'s LabCollector may limit imports to a particular format such as CSV files.<ref name="ABLabcollectorImport">{{cite web |url=https://www.labcollector.com/labcollector-lims/features/reporting/ |title=LabCollector: Importing, Exporting, & Reporting |publisher=AgileBio |accessdate=17 March 2020}}</ref> In particular, data migrations require extra vigilance from the potential buyer, who should confirm the LIMS vendor's ability to cleanly, completely, and reliably import your data into a new system.<ref name="RooksbyNotAll20">{{cite web |url=https://www.interactivesoftware.co.uk/2020/01/28/lims-data-migration/ |title=Not All LIMS Data Migration Services Are Created Equal |author=Rooksby, G. |work=Interactive Software Blog |date=28 January 2020 |accessdate=17 March 2020}}</ref> | However, potential buyers should ensure the LIMS vendor is clear in how their system manages data imports. For example, products such as [[Vendor:AgileBio|AgileBio]]'s LabCollector may limit imports to a particular format such as CSV files.<ref name="ABLabcollectorImport">{{cite web |url=https://www.labcollector.com/labcollector-lims/features/reporting/ |title=LabCollector: Importing, Exporting, & Reporting |publisher=AgileBio |accessdate=17 March 2020}}</ref> In particular, data migrations require extra vigilance from the potential buyer, who should confirm the LIMS vendor's ability to cleanly, completely, and reliably import your data into a new system.<ref name="RooksbyNotAll20">{{cite web |url=https://www.interactivesoftware.co.uk/2020/01/28/lims-data-migration/ |title=Not All LIMS Data Migration Services Are Created Equal |author=Rooksby, G. |work=Interactive Software Blog |date=28 January 2020 |accessdate=17 March 2020}}</ref> | ||
===Internal file or data linking=== | ===Internal file or data linking=== | ||
| Line 145: | Line 145: | ||
===ELN support or integration=== | ===ELN support or integration=== | ||
As a software replacement for more traditional paper [[laboratory | As a software replacement for more traditional paper [[laboratory notebook]]s, the [[electronic laboratory notebook]] (ELN) has been important to laboratory functions. Yet the lines between ELN and LIMS began to blur in the 2000s, with both types of software incorporating features from the other.<ref name="SciCompWorld1">{{cite web |url=https://www.scientific-computing.com/feature/state-eln-market?feature_id=50 |author=Elliot, M.H. |title=The state of the ELN Market |work=Scientific Computing World |date=December 2006–January 2007 |accessdate=17 March 2020}}</ref><ref name="SciCompELN">{{cite web |url=http://www.scientificcomputing.com/articles-IN-Informatics-Convergence-Presents-Opportunities-and-Challenges-111111.aspx |archiveurl=https://web.archive.org/web/20160417085757/https://www.scientificcomputing.com/articles/2011/11/informatics-convergence-presents-opportunities-and-challenges |author=Elliot, Michael H. |title=Informatics Convergence Presents Opportunities and Challenges |publisher=Scientific Computing |date=October 2011 |archivedate=17 April 2016 |accessdate=17 March 2020}}</ref> More than a decade later, the ELN has become more distinct in purpose and functionality and is typically sold as a stand-alone solution. Despite this, some vendors such as [[Vendor:Agilent Technologies, Inc.|Agilent]] have integrated ELN functionality into their LIMS in order to provide greater flexibility to researchers.<ref name="GenohmHome">{{cite web |url=https://explore.agilent.com/AgilentSlims |title=Agilent SLIMS: Lab Workflow Management Software |publisher=Agilent Technologies, Inc |accessdate=06 January 2022}}</ref> Other vendors such as [[Vendor:Core Informatics, LLC|Core Informatics]] (part of [[Vendor:Thermo Scientific|Thermo Fisher Scientific]]) choose to keep the ELN as a separately managed solution but one that can easily integrate with a LIMS solution.<ref name="CICoreELNInt">{{cite web |url=https://www.coreinformatics.com/products/core-eln/core-eln-features/integration/ |archiveurl=https://web.archive.org/web/20160814211539/http://www.coreinformatics.com/products/core-eln/core-eln-features/integration/ |title=Core ELN Integration |publisher=Core Informatics, LLC |archivedate=14 August 2016 |accessdate=06 January 2022}}</ref> | ||
===Export to MS Excel=== | ===Export to MS Excel=== | ||
| Line 167: | Line 167: | ||
===Production control=== | ===Production control=== | ||
There are many types of businesses that produce goods, and in most cases there is a research and development (R&D) laboratory involved at some point in the process. This is especially true in the pharmaceutical and chemical industries, where production measurements such as yield, volume, activity, and impurity are vital. As LIMSs have already recorded such information during tests and analysis, the addition of production control functionality seems natural. Some LIMS take a very active approach to this. For example, the former [[2nd Sight Solutions|2nd Sight Solutions']] OhNo! featured production control as major functionality for the synthesis of [[Radiopharmacology|radiopharmaceuticals]].<ref name="2ndUseOhNo">{{cite web |url=http://www.2sslab.com/Production.html |title=Use OhNo! for complete control |publisher=2nd Sight Solutions |accessdate=17 March 2020}}</ref> Other LIMS may have less pronounced production functionality, while still offering the ability to track the production process in and out of the lab. And yet other LIMSs like [[dialog EDV Systementwicklung GmbH|dialog's]] diaLIMS offer robust production-based functionality but as a module or add-on to the base LIMS software.<ref name="DialogStandard">{{cite web |url=https://www.dialog-edv.de/produkte/standardloesungen/produktionsunterstuetzung/ |title=Standardlösung für die Produktion |publisher=dialog EDV Systementwicklung GmbH |accessdate=17 March 2020}}</ref> | There are many types of businesses that produce goods, and in most cases there is a research and development (R&D) laboratory involved at some point in the process. This is especially true in the pharmaceutical and chemical industries, where production measurements such as yield, volume, activity, and impurity are vital. As LIMSs have already recorded such information during tests and analysis, the addition of production control functionality seems natural. Some LIMS take a very active approach to this. For example, the former [[Vendor:2nd Sight Solutions|2nd Sight Solutions']] OhNo! featured production control as major functionality for the synthesis of [[Radiopharmacology|radiopharmaceuticals]].<ref name="2ndUseOhNo">{{cite web |url=http://www.2sslab.com/Production.html |title=Use OhNo! for complete control |publisher=2nd Sight Solutions |accessdate=17 March 2020}}</ref> Other LIMS may have less pronounced production functionality, while still offering the ability to track the production process in and out of the lab. And yet other LIMSs like [[Vendor:dialog EDV Systementwicklung GmbH|dialog's]] diaLIMS offer robust production-based functionality but as a module or add-on to the base LIMS software.<ref name="DialogStandard">{{cite web |url=https://www.dialog-edv.de/produkte/standardloesungen/produktionsunterstuetzung/ |title=Standardlösung für die Produktion |publisher=dialog EDV Systementwicklung GmbH |accessdate=17 March 2020}}</ref> | ||
The types of functionality that may fall under this feature include: | The types of functionality that may fall under this feature include: | ||
| Line 381: | Line 381: | ||
===Version control=== | ===Version control=== | ||
Version control is a form of safeguard which helps preserve data integrity. This is typically done by creating a modifiable new version of a piece of information rather than allowing the original to be modified. Such versioning may be applied to a wide variety of digital information housed in the LIMS, including test methods, training certifications, instrument logs, specifications, and process and procedure (P&P) documentation. In LIMS like [[LabWare,_Inc.#LabWare_LIMS|LabWare LIMS]], reference data can also be versioned while also retaining the original relationship between samples and test results, including the version of reference data current at the time lab testing is performed.<ref name="LWExploreTables">{{cite web |url=https://limshelp.labware.com/limsv5/V5_Manual_3_15_nbsp__Explore_Tables.htm |archiveurl=https://web.archive.org/web/20170320223135/https://limshelp.labware.com/limsv5/V5_Manual_3_15_nbsp__Explore_Tables.htm |title=3.15 Explore Tables |publisher=LabWare, Inc |archivedate=20 March 2017 |accessdate=06 January 2022}}</ref> Information tracked with such revisions includes attributes like user name, time the edit was made, and what exactly was edited. This also benefits those managing [[LIMS feature#Audit trail|audit trails]] and [[LIMS feature#Chain of custody|chains of custody]]. | Version control is a form of safeguard which helps preserve data integrity. This is typically done by creating a modifiable new version of a piece of information rather than allowing the original to be modified. Such versioning may be applied to a wide variety of digital information housed in the LIMS, including test methods, training certifications, instrument logs, specifications, and process and procedure (P&P) documentation. In LIMS like [[Vendor:LabWare,_Inc.#LabWare_LIMS|LabWare LIMS]], reference data can also be versioned while also retaining the original relationship between samples and test results, including the version of reference data current at the time lab testing is performed.<ref name="LWExploreTables">{{cite web |url=https://limshelp.labware.com/limsv5/V5_Manual_3_15_nbsp__Explore_Tables.htm |archiveurl=https://web.archive.org/web/20170320223135/https://limshelp.labware.com/limsv5/V5_Manual_3_15_nbsp__Explore_Tables.htm |title=3.15 Explore Tables |publisher=LabWare, Inc |archivedate=20 March 2017 |accessdate=06 January 2022}}</ref> Information tracked with such revisions includes attributes like user name, time the edit was made, and what exactly was edited. This also benefits those managing [[LIMS feature#Audit trail|audit trails]] and [[LIMS feature#Chain of custody|chains of custody]]. | ||
[[File:Freezers for cryopreservation at IMSC.JPG|thumb|The temperature of an open cryopreservation container may be monitored on a computer via a connection to a LIMS with environmental monitoring functionality.]] Some LIMS vendors may choose to employ a different form of version control called file locking, which simply puts the affected information into a read-only mode for users while someone else is busy editing it.<ref name="MonkeyTheVal17">{{cite web |url=https://corengen.wordpress.com/2017/01/23/the-value-of-a-control-file-when-writing-data-files/ |title=The value of a control file when writing data files |author=Monkey, S. |work=ShortStories: Tales from software development |date=23 January 2017 |accessdate=18 March 2020}}</ref> Another popular strategy is to, rather than locking the file, allow multiple people edit to a piece of information, later merging the various edits. Potential LIMS buyers may need to inquire with developers to determine what type of versioning scheme is used in the vendor's software. | [[File:Freezers for cryopreservation at IMSC.JPG|thumb|The temperature of an open cryopreservation container may be monitored on a computer via a connection to a LIMS with environmental monitoring functionality.]] Some LIMS vendors may choose to employ a different form of version control called file locking, which simply puts the affected information into a read-only mode for users while someone else is busy editing it.<ref name="MonkeyTheVal17">{{cite web |url=https://corengen.wordpress.com/2017/01/23/the-value-of-a-control-file-when-writing-data-files/ |title=The value of a control file when writing data files |author=Monkey, S. |work=ShortStories: Tales from software development |date=23 January 2017 |accessdate=18 March 2020}}</ref> Another popular strategy is to, rather than locking the file, allow multiple people edit to a piece of information, later merging the various edits. Potential LIMS buyers may need to inquire with developers to determine what type of versioning scheme is used in the vendor's software. | ||
| Line 391: | Line 391: | ||
===Environmental monitoring=== | ===Environmental monitoring=== | ||
Some LIMS like [[Core Informatics, LLC#Core LIMS|Core LIMS]] and [[Autoscribe Informatics|Matrix Gemini]] allow users to monitor the environmental conditions of not only sample storage containers but also the entire laboratory itself.<ref name="CoreEnviro">{{cite web |url=http://corelims.com/products/core-lims-product-summary/features/environmental-monitoring/ |archiveurl=https://web.archive.org/web/20170227121045/http://corelims.com/products/core-lims-product-summary/features/environmental-monitoring/ |title=Environmental Monitoring |publisher=Core Informatics, LLC |archivedate=27 February 2017 |accessdate=06 January 2022}}</ref><ref name="Orchard-WebbMatrix19">{{cite web |url=https://www.biobanking.com/matrix-gemini-biobank-management-lims-includes-new-monitoring-and-maintenance-solutions/ |title=Matrix Gemini Biobank Management LIMS Includes New Monitoring And Maintenance Solutions |author=Orchard-Webb, D. |work=Biobanking.com |date=28 August 2019 |accessdate=18 March 2020}}</ref> Attributes like humidity, air quality, and temperature may be monitored to ensure sample storage units and experiments maintain desired conditions. Alarms may be able to be configured to notify staff if a storage container's environmental attributes go beyond a certain threshold. Manufacturers utilizing a LIMS like [[Novatek International#NOVA-LIMS|NOVA-LIMS]] may also be able to employ more advanced environmental tracking features in the plant to guarantee a more consistent, higher quality product is created.<ref name="NINovaEnviro">{{cite web |url=https://ntint.com/product/novatek-environmental-monitoring/ |title=Novatek Environmental Monitoring |publisher=Novatek International |accessdate=18 March 2020}}</ref> | Some LIMS like [[Vendor:Core Informatics, LLC#Core LIMS|Core LIMS]] and [[Vendor:Autoscribe Informatics|Matrix Gemini]] allow users to monitor the environmental conditions of not only sample storage containers but also the entire laboratory itself.<ref name="CoreEnviro">{{cite web |url=http://corelims.com/products/core-lims-product-summary/features/environmental-monitoring/ |archiveurl=https://web.archive.org/web/20170227121045/http://corelims.com/products/core-lims-product-summary/features/environmental-monitoring/ |title=Environmental Monitoring |publisher=Core Informatics, LLC |archivedate=27 February 2017 |accessdate=06 January 2022}}</ref><ref name="Orchard-WebbMatrix19">{{cite web |url=https://www.biobanking.com/matrix-gemini-biobank-management-lims-includes-new-monitoring-and-maintenance-solutions/ |title=Matrix Gemini Biobank Management LIMS Includes New Monitoring And Maintenance Solutions |author=Orchard-Webb, D. |work=Biobanking.com |date=28 August 2019 |accessdate=18 March 2020}}</ref> Attributes like humidity, air quality, and temperature may be monitored to ensure sample storage units and experiments maintain desired conditions. Alarms may be able to be configured to notify staff if a storage container's environmental attributes go beyond a certain threshold. Manufacturers utilizing a LIMS like [[Vendor:Novatek International#NOVA-LIMS|NOVA-LIMS]] may also be able to employ more advanced environmental tracking features in the plant to guarantee a more consistent, higher quality product is created.<ref name="NINovaEnviro">{{cite web |url=https://ntint.com/product/novatek-environmental-monitoring/ |title=Novatek Environmental Monitoring |publisher=Novatek International |accessdate=18 March 2020}}</ref> | ||
</div> | </div> | ||
| Line 518: | Line 518: | ||
===Usage-based cost=== | ===Usage-based cost=== | ||
While rare, some LIMS vendors allow potential clients to license and utilize the vendor's software under a usage-based cost model. An example of this model in use is Bytewize AB's [[Bytewize AB#O3 Lims and O3 LimsXpress|O3 LimsXpress]], which has a cost directly related to the amount of samples processed each month.<ref name="BWO3X">{{cite web |url=https://www.bytewize.com/o3limsxpress |title=O3 LimsXpress |publisher=Bytewize AB |accessdate=18 March 2020}}</ref> You may also see this sort of usage-based cost on some cloud-based LIMS. | While rare, some LIMS vendors allow potential clients to license and utilize the vendor's software under a usage-based cost model. An example of this model in use is Bytewize AB's [[Vendor:Bytewize AB#O3 Lims and O3 LimsXpress|O3 LimsXpress]], which has a cost directly related to the amount of samples processed each month.<ref name="BWO3X">{{cite web |url=https://www.bytewize.com/o3limsxpress |title=O3 LimsXpress |publisher=Bytewize AB |accessdate=18 March 2020}}</ref> You may also see this sort of usage-based cost on some cloud-based LIMS. | ||
</div> | </div> | ||
Latest revision as of 17:56, 12 April 2024
|
|
You can find a listing of all LIMS vendors—and by extension, the features their products offer—on the LIMS vendor page. |
A LIMS feature is one or more pieces of functionality that appear within a laboratory information management system (LIMS).
The LIMS is an evolving concept, with new features and abilities being introduced every year. As laboratory demands change and technological progress continues, the functions of a LIMS will also change. Yet like the automobile, the LIMS tends to have a base set of functionality that defines it. That functionality can roughly be divided into five laboratory processing phases, with numerous software functions falling under each[1]:
- the reception and log in of a sample and its associated customer data;
- the assignment, scheduling, and tracking of the sample and the associated analytical workload;
- the processing and quality control associated with the sample and the utilized equipment and inventory;
- the storage of data associated with the sample analysis; and
- the inspection, approval, and compilation of the sample data for reporting and/or further analysis.
Of course, there are LIMS features that are difficult to categorize under any of these phases. Such features often contribute to the entire LIMS and how it's utilized. For example, multilingual support appears in LIMS like Assaynet's Assaynet LIMS and SLCLAB Informática's ALFA21, allowing users to interact with the LIMS in more than one language. Some functionality may also overlap several laboratory phases, making it difficult to firmly classify.
The features described below come from an analysis of freely available LIMS product information on vendor websites. An attempt was made to discover the features most utilized in vendors' LIMS products and collect information on those features for each LIMS. Not every possible feature is referenced here; some LIMS products fill specific niches, utilizing unique functionality to solve a specific problem.
That said, keep in mind the categorization of features below is very loose. It may be viable to argue a feature belongs under a different section or multiple sections. For the purposes of organizing this information in an uncomplicated manner, however, some liberty has been taken in the categorizing of features.
Sample, inventory, and data management
To hide the contents of this section for easier reading of other sections, click the "Collapse" link to the right.
Sample login and management
Sample accessioning and management is one of the core functions a modern LIMS is tasked with, whether it's in a manufacturing, water treatment, or pharmaceutical laboratory.[2] As such, researchers who work in these types of labs are unable to complete their experiment-based goals without an effective method of managing samples. The process of sample management for experiments includes, but is not limited to[2]:
- storing related sample information, including aliquot numbers, dates, and external links
- setting user alerts for sample status
- creating and documenting viewable sample container schemas with name and status
- assigning sample access rights
- assigning custom sample ID numbers based on a specification
Additional functionality that could potentially fall under this feature:
- utilizing a unique ID system
- barcoding of samples
- defining sample points and series
- creating data associations for samples, such as pedigree for sample/aliquot relationships or relationships based on experiment, etc.
- issuing sample receipts
Sample tracking
After sample reception and its initial handling procedures, many LIMS can then track sample location as well as chain of custody. Location tracking usually involves assigning the sample to a particular freezer location, often down to the granular level of shelf, rack, box, row, and column. The process of tracking a sample has become more streamlined with increasing support of barcode and radio frequency ID (RFID) technology.[4] While handwritten labels were the norm, barcode and RFID support in a LIMS can "tie together a vast amount of information, clearly relating each sample to a specific case."[3] Other event tracking such as freeze and thaw cycles that a sample undergoes in the laboratory may also be required. As each laboratory's needs for tracking additional data points can vary widely, many modern LIMS have implemented extensive configurability to compensate for varying environments.[5]
The functionality of sample tracking strongly ties into the audit trail and chain of custody features of a LIMS.
Sample and result batching
What is batching? The United States Environmental Protection Agency (EPA) defines a batch as "a group of samples which behave similarly with respect to the sampling or testing procedures being employed and which are processed as a unit."[6] This definition can be applied to many laboratories which handle large quantities of samples for some form of analysis or processing. A LIMS that has the ability to check in, link, and track groups of samples across one or multiple facilities is valuable to such laboratories. Additionally, batching the analysis results of multiple samples or groups of samples gives laboratories more flexibility with how they manage their data. Batching also offers the benefit of mimicking the production groups of samples while also capturing quality control data for the entire group.
Task and event scheduling
Within the context of a LIMS, the ability to schedule a task or event is a natural extension of how work was done in a laboratory before the advent of data management systems. Workloads are assigned to technicians, maintenance schedules are created and followed, and research deadlines must be observed. While these tasks have in the past been performed without LIMS, a modern data information management system can now optimize those tasks and provide additional scheduling functionality to streamline the operation of a lab. Autoscribe Ltd., for example, offers a scheduling module for its LIMS that allows users to automatically schedule multiple jobs, data backups, alarms, and reports.[7] Some LIMS like LabWare, Inc.'s LabWare LIMS offer multiple types of schedulers that match to the particular functions of a research laboratory.[8] Additional functionality within this feature includes the ability to configure automated assignments of analysis requests, establish recurring events, and in most cases, create printable schedules.
Examples of tasks and events that can feasibly be scheduled in a LIMS include:
- registration of received samples into the system
- production of reports
- creation and sending of e-mails and alerts
- maintenance of equipment
- assignment of workloads to personnel
Option for manual result entry
While many LIMS vendors tout the ability of their product to automate the entry of results into the LIMS database, the need for manual data entry of analysis results still exists. This feature is important to laboratories obtaining analysis results from multiple sources, including non-digital paper-based results and instruments that can't be connected to the LIMS. Additional functionality associated with this feature includes a customizable spell check dictionary and the ability to add comments, notes, and narratives to most anything in the LIMS.
Multiple data viewing methods
Laboratories produce data, and LIMS exist to help manage that data. Additionally, even before the existence of LIMS, scientists have had a corresponding need for visually representing data. Today, a LIMS can not only collect and analyze data from samples, but it also can represent that data in reports, graphs, gradients, and spreadsheets. Depending on the LIMS, more than one way to visually represent the data may exist.
Some laboratory information management systems take a specialized approach to data views. For example, Biomatters Ltd.'s Geneious offers multiple methods of viewing complicated sequence analysis data, including 3-D structuring and representations of plasmid vectors, as well as other next-generation sequencing (NGS) activities.[9]
Data and trend analysis
Sample experimentation and analysis play an important part of laboratory informatics, helping laboratories make better sense of their experiments and reach valuable conclusions about them. While this important phase of laboratory work has often been done externally from the LIMS, it's now common to see data analysis tools being included. Such tools allow raw instrument data as well as other imported and internal LIMS data to be analyzed, which then can be stored, processed, and reported on. Additionally, built-in calculations and functions used in the analysis are usually definable and editable for further flexibility, and custom calculations and functions are often able to be made by the end user.As with the feature "multiple data viewing methods," data and trend analysis is also increasingly important in laboratories that have specialized data management needs. When even in 2009 genetic scientists in large- and medium-sized sequencing and core centers were voicing concerns about "a lack of adequate vendor-supported software and laboratory information management systems (LIMS)" that had the necessary analytical features[10], today's data management options, like the previously mentioned Geneious[9], readily offer data analysis tools for a diverse array of laboratory types.
Data and equipment sharing
Aside from data storage and sample registration, a modern LIMS' major contribution to the laboratory is aiding in the sharing of experiment results, reports, and other data types with those who need it most. Rather than pieces of information becoming misplaced or forgotten in laboratory notebooks, the LIMS makes it easier to share sample test results and increase the efficiency of collaboration inside and outside the laboratory. Yet data is more than just sample test results; it also comes in the form of charts, reports, policy and procedure documents, and other document types. Additionally, the need for controlling who has access to these and other types of data is also an important consideration. As such, this feature is at least partially tied to other features like document management and configurable security.
Customizable fields and/or interface
As thorough as some user interface (UI) developers may be in adding relevant fields and interface options for LIMS end users, there are at times options that are either omitted or unanticipated. This has traditionally required the end user to contact the vendor and ask if the needed option(s) can be added in the next release. However, modern LIMS vendors have responded instead by adding functionality that gives end users and/or LIMS administrators more control over the user interface, including the addition and customization of user fields and inputs.
Aspects of the LIMS' user interface that are becoming more customizable by the end user include:
- system nomenclature
- equations used in calculations
- data and universal fields
- data tables
- appearance of the interface and/or menus
- primary system language
- the LIMS source code, especially if in a non-proprietary format
Increasingly, today's vendors tend to refer to their systems as being "configurable" rather than "customizable."[11][12] Vendors such as Autoscribe Informatics will include screen editing and other tools that administrators can user to configure the various screens, data fields, nomenclature, etc. of the LIMS.[11]
Query capability
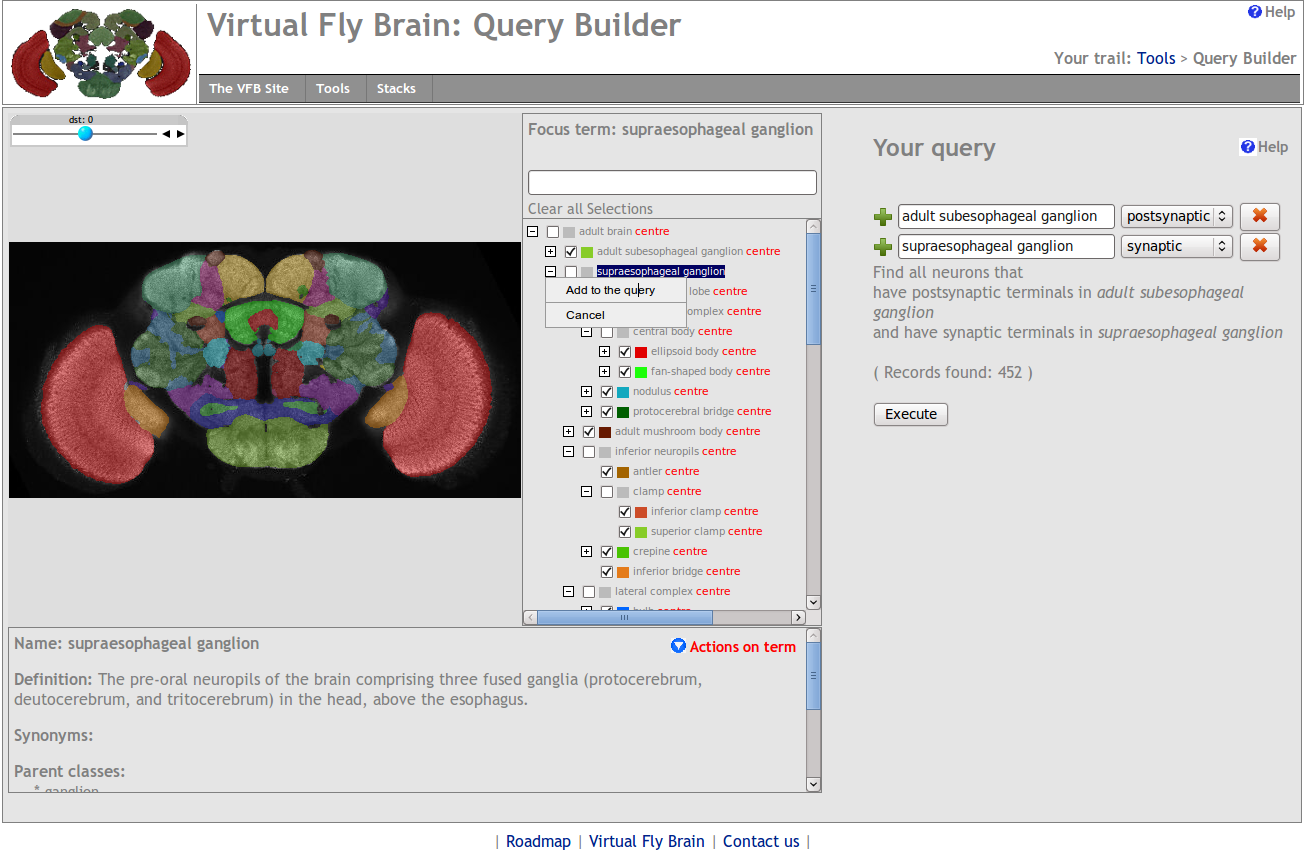
As was the case before the advent of databases and electronic data management solutions, today researchers must search through sample results, experiment notes, and other types of data to better draw conclusions about their research. Whereas this used to mean browsing through laboratory notebooks, Excel spreadsheets, or Access databases, now powerful query tools exists within data management tools like a LIMS. Not only can data be searched for based on name, number, or vendor, LIMSs like Abbott's STARLIMS and Mountain States Consulting's MSC-LIMS allow for queries of attached metadata like user ID, project number, task number, sample type, location, and collection date.[13][14] Finally, as LIMS continue to include both sample management and experimental data management functionality, queries become more powerful in general as now sample and experiment can be matched together in one database.[2]Query functionality often includes the ability to:
- search both transactional data and archived data tables
- search multiple databases via an application programming interface (API) or open database connectivity (ODBC) connection
- filter and sort data
- create ad hoc queries
Import data
Data can originate from numerous places in the laboratory. The ability to import that data into a LIMS can be beneficial, especially when an instrument can't be connected or an external client provides a data feed independent of the LIMS. Some LIMS may even allow users to cross-reference laboratory nomenclature from received data sources with the recipient's. And of course instrument interfacing allows for even more importation options. Additional data validation procedures may be applied to the imported data to guarantee information homogeneity. Some LIMS may also allow for the import of non-normalized legacy data tables and integration with LIMS data tables to form a single unified database.
However, potential buyers should ensure the LIMS vendor is clear in how their system manages data imports. For example, products such as AgileBio's LabCollector may limit imports to a particular format such as CSV files.[15] In particular, data migrations require extra vigilance from the potential buyer, who should confirm the LIMS vendor's ability to cleanly, completely, and reliably import your data into a new system.[16]
Internal file or data linking
This feature allows LIMS users to link together reports, protocols, sample results, and more, providing greater contextual clarity to projects. Examples include:
- linking a sample batch to a test or sample preparation methodology
- linking a test process to a particular customer
- linking a report to a sample batch
- linking a group of test results to a raw data file
- linking an image to a work order
- linking all lab results with the correct reporting test method
- linking data forms created offline (remote data collection) to the submitted data in the system
External file or data linking
This feature allows LIMS users to link together data and files in the LIMS with data, files, and customers outside the domain of the LIMS. Examples include:
- linking external file attachments—such as images and documents—to a LIMS record[17]
- linking data files from chromatography equipment to synthesis data[18]
- linking external standard operating procedure documents with an internal test specification[8]
ELN support or integration
As a software replacement for more traditional paper laboratory notebooks, the electronic laboratory notebook (ELN) has been important to laboratory functions. Yet the lines between ELN and LIMS began to blur in the 2000s, with both types of software incorporating features from the other.[19][20] More than a decade later, the ELN has become more distinct in purpose and functionality and is typically sold as a stand-alone solution. Despite this, some vendors such as Agilent have integrated ELN functionality into their LIMS in order to provide greater flexibility to researchers.[21] Other vendors such as Core Informatics (part of Thermo Fisher Scientific) choose to keep the ELN as a separately managed solution but one that can easily integrate with a LIMS solution.[22]
Export to MS Excel
While Microsoft Excel had long been used within the laboratory setting, a slow shift towards relational databases and LIMS occurred in the late 1990s and early 2000s.[23] Additional concerns with the difficulties of Excel's validation and compliance with FDA 21 CFR Part 11 and other regulations ultimately led many labs to turn to data management solutions that are easier to validate.[24] Several decades later, some laboratories continue to use Excel in some fashion, and thus having a LIMS capable of handling Excel data remains useful. In particular, selecting and exporting the LIMS' data (from test results to quality control charts) into a format such as Excel may further data visualization, insights, and dissemination for some users.[25][26]
Raw data management
While not described as a feature on most LIMS vendor websites, a few indicate that their LIMS is capable of managing (import, export, editing, etc.) data in its raw format for future analysis. This raw data is typically implied to be originating from instruments that interface with the LIMS, which in itself is not unusual. That raw data may remain housed in instruments, getting transformed into reportable data in the LIMS, but presumably most LIMS are also capable of housing the raw instrument data as well.[27] Labs may also turn to scientific data management systems to better manage their raw data sources.[28]
Data warehouse
A LIMS' data warehouse serves the important function of storing, extracting, and managing the data that laboratories produce for the purposes or analysis, reporting, and process management, typically separate from the primary storage database. Data warehouses also offer the benefit of speeding up queries, making queries and data mining more user-friendly, and smoothing out data gaps.[29]
Deadline control
Deadline control is functionality within a LIMS that allows users to manage and be notified of events that occur within the laboratory. With this functionality users can also be notified of upcoming deadlines on anything from sample analysis to license renewal.
Note that this functionality may also feasibly fall under the task and event scheduling or alarms features. As deadline control seems to be advertised as a notable feature by only a few vendors, it seems even more likely that this functionality is practically considered part of scheduling or alarms.
Production control
There are many types of businesses that produce goods, and in most cases there is a research and development (R&D) laboratory involved at some point in the process. This is especially true in the pharmaceutical and chemical industries, where production measurements such as yield, volume, activity, and impurity are vital. As LIMSs have already recorded such information during tests and analysis, the addition of production control functionality seems natural. Some LIMS take a very active approach to this. For example, the former 2nd Sight Solutions' OhNo! featured production control as major functionality for the synthesis of radiopharmaceuticals.[30] Other LIMS may have less pronounced production functionality, while still offering the ability to track the production process in and out of the lab. And yet other LIMSs like dialog's diaLIMS offer robust production-based functionality but as a module or add-on to the base LIMS software.[31]
The types of functionality that may fall under this feature include:
- recipe management
- consumable tracking
- batch traceability
- production planning
- enterprise resource planning
Finally, in production environments the LIMS is often referenced in conjunction with a manufacturing execution system (MES). The MES is used for tracking and documenting how raw materials are transformed into finished products, as well as optimizing the various workflows in the production processes. The MES may either interface with a LIMS for quality testing, management, and statistical analysis, or the MES may actually contain a LIMS module.[32][33]
Project and/or task management
Project and task management within a LIMS typically involves the scheduling of tasks to workers and organizing associated tasks into a more cohesive unit for better tracking and management. While the functionality of task and event scheduling can also be found in project and task management, many LIMS include functionality beyond scheduling that warrants the addition of the project and/or task management feature. This functionality includes:
- job allocation and rescheduling
- instrument workload tracking
- time tracking
- pending workload verification
- priority setting
- project-based workflow management
- sample, batch, and document linking
- work list sharing
- recurring event management
Inventory management
- register origin, demographics of incoming materials
- track used and in-use items via barcodes or RFID
- track inventory reduction based on usage and shipping out of the lab
- create alerts for when items reach a certain stock level
- calculate inventory cost and fluctuation
- manage transportation and routing
- manual incrementing/decrementing of item counts
- track location and usage of laboratory equipment
- assign storage locations
- track forensic evidence
Document creation and management
Standard operating procedures (SOPs), specifications, safety manuals and protocols, regulatory files, certificates, reports, graphs, images, and receipts are all collected and used in the average laboratory. With a LIMS already designed to manage and store sample and experiment data, it makes sense to include functionality to create, import, export, and manage other sorts of data files. As sample and experimental data can be indexed, queried, and linked, so too can documents. Functionality of a typical document management system includes the ability to:
- upload and index documents of most any file type and size
- enforce version control
- provide full text search
- convert and/or export to PDF or other relevant formats
- add documents as attachments to other analyses, files, projects, etc. in the system
Case management
The laboratory information system (LIS) has played an important role in the clinical case management tasks of patient-centric and clinical laboratories. However, LIMS begun taking on clinical functionality such as case management around the late 2000s, which began to effectively blur the lines between an LIS and a LIMS.[34]. Since then, many more self-proclaimed LIMS products have emerged in the clinical, public health, and veterinary industries, areas that have historically been served by LIS software. This has resulted in numerous LIMS options that offer clinical functionality such as case management. When also taking into consideration the fields of law enforcement and forensic science, case management plays an important role in some LIMS. Functionality seen in the case management feature includes:
- case accessioning and assignment
- disease tracking
- trend analysis
- clinical history follow-up
- outreach services
- out-of-range result alerts
- document and result association
- evidence control
- study management
Workflow management
Workflow management is common in the laboratory, acting as a graphical representation of planned sequential steps to either fully or partially automate a process within the lab. Separate standards-based workflow management systems (in the form of a software component) have traditionally performed this task.[35] However, in the late 2000s LIMS vendors began incorporating workflow management functionality into their LIMS software, reducing the headaches that customization of a LIMS often brought.[36]Today's commercial and open-source LIMS solutions frequently feature workflow management functionality, including[37][38][36]:
- attribute definition of activities
- definition of inputs and outputs of activities
- assignment of documentation to activities
- setting of quality control limits
- dynamically modify workflow in case of future changes
- receive notification of changes to the workflow
- sending user-defined messages during the process
Specification management
Specification (spec) management is vital to not only the manufacturing and research industries but also to a host of other laboratories requiring precise measurements and infallible test methods. Just as ASTM offers standards and specs for LIMS[39], so too do LIMS users have standards and specs for their laboratory. With spec management in place within the LIMS, laboratories can then:
- enforce standard operating procedures and business rules
- create specs down to a project or sample level
- validate recipes and procedures
- accept or reject sample batches
- document internal and external spec history
Note that some of the functionality of spec management may cross over into the realm of quality control and data validation.
Customer and supplier management
Unless a laboratory is conducting internalized independent research, in most cases it will do business with external entities such as contract labs, sample providers, equipment providers, and reagent suppliers. In some cases, even internal employees may be considered a customer, necessitating documentation of who is using the system and in what ways. For a veterinary lab, the customer may be the animal and handler. For a forensic lab the customer may be more complex: internal staff, university staff, police departments, and maintainers of nationwide crime databases may all at some point act as customers. In these cases, documenting these various points of contact and linking them to samples, equipment, and tests becomes vital. Managing demographics, complaints, correspondence, and history are all feasible with customer management functionality. This process is often made simpler through the use of a more context-neutral entity creation system, which allows for more flexible management of contacts.
This feature may also be referred to as contact management, an address book module, or a customer service module.
Billing management
While the finances of a laboratory are important, they've typically been handled separately as a business process. However, some LIMS include additional functionality to make handling financial transactions and documentation of all sorts possible within the LIMS. In theory, such functionality brings the possibility of keeping more of a laboratory's data centrally located and queryable. This feature may include:
- payment processing
- expense reporting
- price quotes
- revenue cycle management
- workload tracking of billable hours
- bill of materials
- grant management
Quality, security, and compliance
To hide the contents of this section for easier reading of other sections, click the "Collapse" link to the right.
Regulatory compliance
The topic of whether or not a LIMS meets regulatory compliance is often a complex one. While Title 21 CFR Part 11 has arguably had the largest influence on an electronic data management system's compliance, other influential standards have shaped the way LIMS and other systems handle and store data. Other compliance-based codes, standards, and regulations include:
- ASTM standards
- ASCLD/LAB crime lab accreditation
- Freedom of Information Act
- GALP and GAMP
- HIPAA
- Health Level 7
- ICD
- ISO/IEC 17025
- ISO 9000/9001
- ISO/TS 16949
- ODBC
- TNI and NELAP enviromnental lab accreditation
- Title 40 CFR Part 3
With so many codes, standards, and regulations, it can be challenging for potential LIMS buyers to fully vet whether a vendor's solution meets regulatory requirements, let alone helps labs meet the regulations and standards that affect them. In truth, most reputable vendors today have regulatory compliance well considered for their LIMS solutions, which are often purpose-built. However, it is ultimately up to the potential buyer to confirm that the vendor's solution complies to activities within their industry. Buyers are advised to contact vendors with their user requirements and ask how the vendor's software meets and/or exceeds those requirements.
QA/QC functions
The quality management functions of a LIMS allow users to maintain a level of necessary quality across many of the activites in a laboratory. From running quality assurance tests to ensuring employed researchers are proficient at certain tasks, the QA/QC functionality of a LIMS is largely responsible for the output of consistent data and manufactured products in and out of the lab.
Common functionality includes[40][41]:
- single or batch QA/QC tests
- quality control charts and reports
- proficiency testing
- document management
- instrument maintenance
- data acceptance/rejection
- certificates of analysis (COA)
- data types defined by QC analysis
Performance evaluation
As prevalent as document management has become in LIMS, it makes sense to that the LIMS should also provide functionality to collate and store all the documentation associated with employee training and certification. Changes to laboratory techniques, scientific understanding, and business practices force researchers to learn, reevaluate, and demonstrate competency in order to maintain quality levels in the laboratory. Evaluations can frequently extend beyond staff members, however. Clinics, visit types, vendors, or test species can also be tracked and evaluated based on custom criteria. The performance evaluation functionality of a LIMS makes this possible.
That functionality typically includes the ability to maintain training records and history, and also to link that training to a technique or piece of equipment. Afterwards, the staff member, vendor, etc. can be marked as competent or certified in the equipment, knowledge, or process. Periodical assessment of the training and its practical effectiveness can later be performed. Productivity of an entity or process can also be gauged over a certain date range based on tracked time, pre-determined milestones, or some other criteria.
Audit trail

Information recorded in the audit trail typically includes:
- operator code
- time stamp
- location
- case number
- transaction type
- amount and quantity prior to change
- user notes (e.g., reason for change)
- in some cases, data in temporary memory[44]
Chain of custody
The chain of custody (COC) of an item is of varying importance, depending on the type of laboratory. A highly regulated laboratory that works under Code of Federal Regulation or other guidelines makes tracking COC a vital part of its operations. This is especially true in forensic labs, which depend on continuous accountability of their evidence collection, retention, and disposal procedures.[45] At its core, the COC strives to answer at least these five questions[46]:
- Where is my sample or specimen now?
- Who possess my sample or specimen now?
- When did that individual take possession of my sample or specimen?
- Where has my sample or specimen been?
- Who has been in possession of my sample or specimen?
As with an audit trail, a laboratory depends on recorded information like user ID, time stamp, and location ID to maintain a robust and accurate COC. Barcodes, inventory management, and configurable security roles all play an important part in maintaining chain of custody.
Configurable roles and security
Many roles exist within the laboratory setting, each with its own set of responsibilities. And just as the role an individual plays within the laboratory may change, so may the responsibilities associated with each role. This sort of change necessitates a flexible and configurable security system, one that allows for the placement of individual LIMS users into defined security roles which provide role-specific access to certain LIMS functionality. Additionally, as responsibilities change within roles, that same flexible configuration is necessary for assigning or restricting access to certain LIMS functionality for each existing or newly created role.
Of course, roles aren't always assigned on an individual level. Often large groups of individuals may need to be assigned to roles, necessitating group assignments for security purposes. For example, a group of laboratory trainees may only be given read-only access to the sample login and sample tracking functionality of the system through a custom "Trainees" group role, while the head researcher of the lab may be given the "Administrator" role, which allows that individual to access most if not all of the LIMS' functionality.
Data normalization
For the purposes of describing LIMS functionality, "data normalization" specifically refers to the process of ensuring data that moves into the LIMS is formatted in a standardized way, matching the format of existing LIMS data.
Here's an example to better explain this issue. When a LIMS is initially configured, in most if not all cases a clear standard can be set for how logged samples and their associated measurements pre- and post-analysis are recorded in the system. Perhaps all temperatures will be recorded in Celsius to two decimal places. If temperature data imported from a spreadsheet or a lab instrument is not in this format, the LIMS can normalize the incoming data to match the standard already set for existing LIMS temperature data. This ensures consistency within the LIMS database and typically leads to better data validation efforts later on.
Note: Some LIMS developers may include data normalization functionality within what they may refer to as "data validation" functionality. The line between these two may be blurred or not exist at all.
Data validation
For the purposes of describing LIMS functionality, "data validation" specifically refers to the process of ensuring existing data in the LIMS—either pre-analysis or post-analysis—sufficiently meets any number of standards or thresholds set for sample login, sample analysis, or any other data management process in the LIMS. This validation process may be completely automatic and system-based, or it may also include additional steps on the part of the user base utilizing additional LIMS functionality, including verification of standard operating procedures (SOPs), QC samples, and QA approval.[47][48]
Note: This functionality shouldn't be confused with the process of validating the LIMS itself, which is an entirely different process partially falling under regulatory compliance and involves the process of ensuring "the software is performing in a manner for which it was designed."[49]
Data encryption
The existence of this functionality in LIMS software generally indicates the LIMS has the ability to protect the integrity and authenticity of its housed data through the use of a variety of technologies which makes data unreadable except to those users or systems possessing a key/right/etc. to unlock/read the data. This functionality has been important for the web-enabled LIMS, which transfers information over the internet in a client-server relationship, and it has become an increasingly vital part of cloud-based software offerings managing data in transit and at rest.[50] As a wide variety of encryption technologies exist, it's generally a good idea to consult with the developers of a LIMS to determine the strengths and weaknesses of their employed encryption methods.
Version control
Version control is a form of safeguard which helps preserve data integrity. This is typically done by creating a modifiable new version of a piece of information rather than allowing the original to be modified. Such versioning may be applied to a wide variety of digital information housed in the LIMS, including test methods, training certifications, instrument logs, specifications, and process and procedure (P&P) documentation. In LIMS like LabWare LIMS, reference data can also be versioned while also retaining the original relationship between samples and test results, including the version of reference data current at the time lab testing is performed.[51] Information tracked with such revisions includes attributes like user name, time the edit was made, and what exactly was edited. This also benefits those managing audit trails and chains of custody.
Automatic data backup
The functionality of automatic data backup in a LIMS usually means information contained in one or more associated databases or data warehouses can be automatically preserved in an additional backup file. The save location for that file as well as the scheduled backup time is configurable, typically through the administrative module of the LIMS. As cloud-based LIMS have taken hold, scheduled cloud-to-cloud data backups of software data are becoming more practical.[53]
Environmental monitoring
Some LIMS like Core LIMS and Matrix Gemini allow users to monitor the environmental conditions of not only sample storage containers but also the entire laboratory itself.[54][55] Attributes like humidity, air quality, and temperature may be monitored to ensure sample storage units and experiments maintain desired conditions. Alarms may be able to be configured to notify staff if a storage container's environmental attributes go beyond a certain threshold. Manufacturers utilizing a LIMS like NOVA-LIMS may also be able to employ more advanced environmental tracking features in the plant to guarantee a more consistent, higher quality product is created.[56]
Reporting, barcoding, and printing
To hide the contents of this section for easier reading of other sections, click the "Collapse" link to the right.
Custom reporting
Reporting is a vital part of a LIMS, as it allows users to gain a clearer picture of collected data and potential trends. At a minimum, a number of pre-configured report styles come standard with a LIMS. However, some LIMS are more flexible than others, offering the ability to customize reports in numerous ways. The most popular attributes of custom reporting include custom headers, custom information placement (such as branding), charts, pivot tables, and multiple output formats.
Note: Some LIMS vendors will offer custom reporting as an option as an added cost, depending on both the base reporting functionality of the LIMS and the level of customization required.
Report printing
Today's LIMS software almost universally offers the ability to print reports and other materials, so this feature may seem a bit redundant to list. Nonetheless, printer support is a feature worth confirming when considering a piece of LIMS software.
Label support
The label—typically affixed to a sample or evidence container—is a vital part of the sample tracking process.[57] Identifying information such as sample number, batch number, and barcodes are printed on such labels to ensure optimized sample management and more precise sample data. As such, some LIMS allow users to design and print labels directly from the software.
Barcode support
Barcodes offer many advantages to laboratory techs handling samples, including more accurate data input, tighter sample/instrument associations, tighter sample/study associations, and more room for human-readable information on a label.[57] As such, LIMS developers have integrated barcode support into their laboratory information management systems, including support for symbologies like Code 128, Code 39, and Interleaved 2 of 5. Aside from printing options, a LIMS may also offer support for a variety of barcode readers.
Radio-frequency identification (RFID) technology has also made inroads into the laboratory. Like barcode labels, RFID tags can be placed on samples and get read when exposed to active RFID scanning devices. This technology has some advantages over barcodes in that direct line of sight isn't required, the scanning and tracking process is more automated, and more data can be encoded.[58] With these apparent advantages, some LIMS vendors have also added support for RFID-fed sample information.
Barcode/RFID support and label support are typically found together in LIMS software, but not always, thus their separation into two features of a LIMS.
Export to PDF
A LIMS with this feature is able to collect and save information into a Portable Document Format (PDF).
Export to MS Word
A LIMS with this feature is able to collect and save information into a Microsoft Office Word format.
Export to HTML or XML
A LIMS with this feature is able to collect and save information into a HyperText Markup Language (HTML) and/or Extensible Markup Language (XML) format.
Fax integration
A LIMS with this feature is able to connect with a fax machine and send information to it via manual input, automatically, and/or at scheduled intervals. However, with the declining popularity of the fax machine[59], this feature is starting to disappear from modern LIMS.
Email integration
A LIMS with this feature is able to integrate with and use the electronic mail information exchange method to send reports, alerts, and more via manual input, automatically, and/or at scheduled intervals.
Base functionality
To hide the contents of this section for easier reading of other sections, click the "Collapse" link to the right.
Administrator management
The administrator management tools of a LIMS allow lab technicians to set up the LIMS most optimally for the laboratory. Through the administrator management interface of a LIMS, other features may be accessed like setting up user roles and scheduling automatic data backups.
Like report printing, administrator management is nearly ubiquitous in LIMS software, generally considered a mandatory feature. However, for the purposes of being thorough, it's important to point out its existence.
Modular
This feature indicates that a LIMS has an intentional modular design, which separates some functionality into manageable components of the overall system. Generally speaking, a modular design allows for 1. the structured addition of new functionality to a LIMS and 2. the limiting of overall effects on the system design as new functionality is added.
Instrument interfacing and management

The ability to calibrate and schedule maintenance for interfaced instruments may also be included in this category.
Mobile device integration
Some LIMS vendors tout support for mobile devices using their laboratory information management system. This feature has become increasingly common with the advent of first the web-based LIMS, followed by the cloud-based LIMS. This has given technicians the ability to collect data in the field, approve results remotely, and manage experiment workflow on the fly.[63]
Alarms and/or alerts
Alarms and alerts are an integral part of a LIMS. They can be automatic or scheduled, and they can come in the form of an e-mail, a pop-up message, or a mobile text message. When the results for a sample analysis go out out of range, an automatic warning message can appear on the screen of the technician responsible for the analysis. A scheduled alert can be e-mailed to a lab technician every month indicating a piece of laboratory equipment needs routine maintenance. If the LIMS is equipped with environmental monitoring, an alert can be sent in the form of an SMS text message to the head researcher if the temperature inside a freezer unexpectedly rises. All of these scenarios represent a tiny fraction of the possible implementation of alarms and alerts in a LIMS, highlighting how powerful (yet easy to take for granted) this feature is.
This feature specifically refers to a LIMS' ability to track the amount of time an employee spends at work in general (for payroll purposes) or on more specific projects and tasks (as part of an employee work evaluation program).
Voice recognition system
A LIMS with this feature allows some functions of the LIMS (for example, accessing sample analysis results) to be accessed via voice commands.
External monitoring
This feature allows clients outside the laboratory to monitor the status of sample batches, test results, and more via an online web portal or, less commonly, as activity alerts sent via e-mail, fax, or SMS.
Messaging
The messaging feature of a LIMS may refer to one (or both) of two things:- a built-in instant messaging system that allows users to converse with each other through text messages real-time, or
- SMS text messaging integration that allows users or the LIMS itself to send messages or alerts to a user's mobile or smart phone.
Multilingual
If a LIMS is listed as multilingual, its an indication the LIMS interface can be configured to display more than one language depending on the preference a user or administrator chooses. Some LIMS interfaces can only be displayed in one of two languages (English or German, for example), while others come configured with support for dozens of languages.
Network-capable
This feature is perhaps archaic and/or obvious, but it is mentioned nonetheless. It's generally applied to a non-web-based (on-premises) LIMS installed over a local or wide-area computer network, essentially indicating the LIMS is not an isolated application, but rather one that can interface with other instances of the LIMS or other networked instruments.
Web client or portal
A LIMS with a web client or portal is either a web-based LIMS (one that is not installed on every computer, but rather is hosted on a server and accessed via a web browser) or a non-web-based LIMS with an included portal to access it via the internet.
Online or integrated help
This indicates a LIMS has help infrastructure integrated into the software, support documentation via the LIMS vendor's website, or both.
Software as a service delivery model
This indicates the software can be licensed and utilized via the software as a service (SaaS) delivery model. This typically constitutes a LIMS vendor having a "cloud-based" offering.
Usage-based cost
While rare, some LIMS vendors allow potential clients to license and utilize the vendor's software under a usage-based cost model. An example of this model in use is Bytewize AB's O3 LimsXpress, which has a cost directly related to the amount of samples processed each month.[64] You may also see this sort of usage-based cost on some cloud-based LIMS.
References
- ↑ Skobelev, D.O.; T.M. Zaytseva; A.D. Kozlov; V.L. Perepelitsa; A.S. Makarova (2011). "Laboratory information management systems in the work of the analytic laboratory". Measurement Techniques 53 (10): 1182–1189. doi:10.1007/s11018-011-9638-7.
- ↑ 2.0 2.1 2.2 Macneil, R. (2011). "The benefits of integrated systems for managing both samples and experimental data: An opportunity for labs in universities and government research institutions to lead the way". Automated Experimentation 3 (2): 2. doi:10.1186/1759-4499-3-2. PMC PMC3146905. PMID 21707999. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3146905.
- ↑ 3.0 3.1 Murthy, T.; Hewson, B. (1 September 2010). "Effective Forensic Sample Tracking and Handling". American Laboratory. https://www.americanlaboratory.com/914-Application-Notes/506-Effective-Forensic-Sample-Tracking-and-Handling/. Retrieved 13 March 2020.
- ↑ Vaniotis, G. (19 July 2018). "How RFID Labels Can Smarten Up Your Laboratory". Labtag Blog. https://blog.labtag.com/rfid-labels-in-the-lab. Retrieved 13 March 2020.
- ↑ Muntean, E.; Munteanb, N.; Mihăiescua, T.; Mihăiescuc, R. (2008). "LIMS use in laboratory data management". ProEnvironment/Promediu 1 (2): 19–23. http://journals.usamvcluj.ro/index.php/promediu/article/view/2835.
- ↑ "Batch Sizes And QC Questions". U.S. EPA. 4 April 2016. https://archive.epa.gov/epawaste/hazard/web/html/faqs_qc.html. Retrieved 13 March 2020.
- ↑ "Easy scheduling of LIMS activities". Autoscribe Ltd. https://www.autoscribeinformatics.com/resources/press-releases/easy-scheduling-of-lims-activities. Retrieved 13 March 2020.
- ↑ 8.0 8.1 "Summary of Core Product Features". LabWare, Inc. Archived from the original on 12 May 2020. https://web.archive.org/web/20200512174255/https://www.labware.com/en/p/Products/Summary-of-Core-Product-Features. Retrieved 06 January 2022.
- ↑ 9.0 9.1 "NGS Visualization and Downstream Analysis". Biomatters Ltd. https://www.geneious.com/features/ngs-visualization-downstream-analysis/. Retrieved 13 March 2020.
- ↑ Richter, B.G.; Sexton, D.P. (2009). "Managing and Analyzing Next-Generation Sequence Data". PLoS Computational Biology 5 (6): e1000369. doi:10.1371/journal.pcbi.1000369. PMC PMC2667638. PMID 19557125. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2667638.
- ↑ 11.0 11.1 "LIMS Customization vs Configuration". Autoscribe Informatics, Inc. 8 March 2015. https://www.autoscribeinformatics.com/resources/blog/lims-customization-vs-configuration. Retrieved 13 March 2020.
- ↑ "The Benefits of a configurable COTS LIMS". Promium, LLC. https://www.promium.com/resources/news-articles/the-benefits-of-a-configurable-off-the-shelf-lims-cots/. Retrieved 13 March 2020.
- ↑ "STARLIMS Biorepository Industry LIMS Specification Document" (PDF). Abbott Informatics Corporation. 18 September 2017. Archived from the original on 10 August 2017. https://web.archive.org/web/20170810191058/https://www.informatics.abbott/shared/biorepository-lims-spec-document.pdf. Retrieved 06 January 2022.
- ↑ "MSC-LIMS Product Summary". Mountain States Consulting, LLC. http://www.msc-lims.com/lims/msc-lims.html. Retrieved 17 March 2020.
- ↑ "LabCollector: Importing, Exporting, & Reporting". AgileBio. https://www.labcollector.com/labcollector-lims/features/reporting/. Retrieved 17 March 2020.
- ↑ Rooksby, G. (28 January 2020). "Not All LIMS Data Migration Services Are Created Equal". Interactive Software Blog. https://www.interactivesoftware.co.uk/2020/01/28/lims-data-migration/. Retrieved 17 March 2020.
- ↑ "OasisLIMS 4.0 Enterprise". Oasis Infotech Pvt. Ltd. https://www.oasislims.com/lims.php. Retrieved 17 March 2020.
- ↑ "Get OhNo! for Radiopharmaceutical Control". 2nd Sight Solutions. Archived from the original on 23 January 2016. https://web.archive.org/web/20160123085133/http://www.2sslab.com/Features.html. Retrieved 06 January 2022.
- ↑ Elliot, M.H. (December 2006–January 2007). "The state of the ELN Market". Scientific Computing World. https://www.scientific-computing.com/feature/state-eln-market?feature_id=50. Retrieved 17 March 2020.
- ↑ Elliot, Michael H. (October 2011). "Informatics Convergence Presents Opportunities and Challenges". Scientific Computing. Archived from the original on 17 April 2016. https://web.archive.org/web/20160417085757/https://www.scientificcomputing.com/articles/2011/11/informatics-convergence-presents-opportunities-and-challenges. Retrieved 17 March 2020.
- ↑ "Agilent SLIMS: Lab Workflow Management Software". Agilent Technologies, Inc. https://explore.agilent.com/AgilentSlims. Retrieved 06 January 2022.
- ↑ "Core ELN Integration". Core Informatics, LLC. Archived from the original on 14 August 2016. https://web.archive.org/web/20160814211539/http://www.coreinformatics.com/products/core-eln/core-eln-features/integration/. Retrieved 06 January 2022.
- ↑ Williams, R.W. (2003). "Managing Your Lab Data Flux: Getting Beyond Excel" (PDF). The Bioinformatics of Brains: From Genes and Proteins to Behaviors. Society for Neuroscience. Archived from the original on 16 June 2010. https://web.archive.org/web/20100616100719/http://www.sfn.org/skins/main/pdf/ShortCourses/2003/sc1_9.pdf. Retrieved 17 March 2020.
- ↑ Howard, D.A.; Harrison, D. (2007). "A Pragmatic Approach to the Validation of Excel Spreadsheets – Overview" (PDF). Pharma IT 1 (2): 30–35. http://www.spreadsheetvalidation.com/pdf/Excel_Spreadsheet_Validation_Overview.pdf.
- ↑ "Excel Interface". Mountain States Consulting, LLC. https://www.msc-lims.com/demo/excelinterface.html. Retrieved 17 March 2020.
- ↑ "LIMS Quality Control QA/QC". Online LIMS Canada Limited. https://www.onlims.com/product/quality-control-qaqc/. Retrieved 17 March 2020.
- ↑ "LISA.lims - The Premier LIMS Solution". Systat Software, Inc. http://www.sigmaplot.co.uk/products/lisa/index.php. Retrieved 17 March 2020.
- ↑ CSols, Inc (25 July 2019). "The Value of a Scientific Data Management System (SDMS)". https://www.csolsinc.com/blog/the-value-of-a-scientific-data-management-system-sdms/. Retrieved 17 March 2020.
- ↑ Harris, J. (20 September 2018). "What Is A Data Warehouse? A Tutorial For Beginners". Panoply Blog. Panoply Technologies, Inc. https://blog.panoply.io/what-is-a-data-warehouse-a-tutorial-for-beginners. Retrieved 17 March 2020.
- ↑ "Use OhNo! for complete control". 2nd Sight Solutions. http://www.2sslab.com/Production.html. Retrieved 17 March 2020.
- ↑ "Standardlösung für die Produktion". dialog EDV Systementwicklung GmbH. https://www.dialog-edv.de/produkte/standardloesungen/produktionsunterstuetzung/. Retrieved 17 March 2020.
- ↑ "Manufacturing Execution Software - Lab Information Management Software". I-Track, Inc. https://www.i-trackcorp.com/what-is-mes.html. Retrieved 17 March 2020.
- ↑ "Manufacturing Execution System & LIMS for Strip Materials". NETRONIC Software GmbH. Archived from the original on 04 June 2018. https://web.archive.org/web/20180604124600/https://www.netronic.com/solutions/manufacturing-execution-system-and-lims. Retrieved 06 January 2022.
- ↑ Hice, R. (1 July 2009). "Swimming in the Clinical Pool: Why LIMS are supplanting old-school clinical LIS applications". STARLIMS' Laboratory Informatics Blog. Archived from the original on 19 January 2010. https://web.archive.org/web/20100119015222/http://blog.starlims.com/2009/07/01/swimming-in-the-clinical-pool-why-lims-are-supplanting-old-school-clinical-lis-applications/. Retrieved 17 March 2020.
- ↑ "Workflow Management Coalition Terminology & Glossary" (PDF). Workflow Management Coalition. February 1999. pp. 9. Archived from the original on 24 October 2020. https://web.archive.org/web/20201024165000/http://www.wfmc.org/standards/docs/TC-1011_term_glossary_v3.pdf. Retrieved 06 January 2022.
- ↑ 36.0 36.1 Maxwell, G. (1 November 2003). "Using Workflows in LIMS to Reduce Customization". Scientific Computing. Archived from the original on 07 August 2009. https://web.archive.org/web/20090807034051/http://www.scientificcomputing.com/using-workflows-in-lims-to-reduce.aspx. Retrieved 17 March 2020.
- ↑ Melo, A.; Faria-Campos, A.; DeLaat, D.M. et al. (2010). "SIGLa: an adaptable LIMS for multiple laboratories". BMC Genomics 11 (Suppl 5): S8. doi:10.1186/1471-2164-11-S5-S8. PMC PMC3045801. PMID 21210974. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3045801.
- ↑ "X-LIMS". EthoSoft, Inc. https://ethosoft.com/x-lims/. Retrieved 17 March 2020.
- ↑ "ASTM E1578 18 Standard Guide for Laboratory Informatics". ASTM International. https://www.astm.org/e1578-18.html. Retrieved 06 January 2022.
- ↑ Paszko, C.; Pugsley, C. (2000). "Considerations in selecting a laboratory information management system (LIMS)". American Laboratory 32 (18): 38–42. doi:10.1016/0169-7439(94)90006-X.
- ↑ Hull, C.; Wray, B.; Winslow, F.; Vilicich, M. (2011). "Tracking and Controlling Everything that Affects Quality is the Key to a Quality Management System". Combinatorial Chemistry & High Throughput Screening 14 (9): 772–780. doi:10.2174/138620711796957125. PMID 21631414.
- ↑ "Title 21: Food and Drugs - Chapter 1: Food and Drug Administration, Department of Health and Human Services, Subchapter A: General - Part 11: Electronic Records; Electronic Signatures". U.S. Food and Drug Administration. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=11. Retrieved 18 March 2020.
- ↑ McDowall, R.D. (June 2016). "How Can LIMS Help Ensure Data Integrity?" (PDF). LC-GC Europe. https://rx-360.org/wp-content/uploads/2018/08/How-Can-LIMS-Help-Ensure-Data-Integrity-by-R.D.-McDowall-2016.pdf. Retrieved 18 March 2020.
- ↑ LabVantage Solutions (14 September 2019). "Data Integrity is Here. What Does it Mean for CMOs?". LabVantage Blog. https://www.labvantage.com/data-integrity-is-here-what-does-it-mean-for-cmos/. Retrieved 18 March 2020.
- ↑ Jones, A.; Valli, C. (2009). "Chapter 1: An Introduction to Digital Forensics". Building a Digital Forensic Laboratory: Establishing and Managing a Successful Facility. Butterworth-Heinemann. p. 11. ISBN 9781856175104. https://books.google.com/books?id=F5IU7XXKwCQC.
- ↑ Tomlinson, J. J.; Elliott-Smith, W.; Radosta, T. (2006). "Laboratory Information Management System Chain of Custody: Reliability and Security" (in en). Journal of Automated Methods and Management in Chemistry 2006: 1–4. doi:10.1155/JAMMC/2006/74907. ISSN 1463-9246. PMC PMC1903459. PMID 17671623. http://www.hindawi.com/journals/jamc/2006/074907/abs/.
- ↑ "Quality Assurance - Data Management" (PDF). Stable Isotope Ratio Facility for Environmental Research (SIRFER) at the University of Utah. Archived from the original on 17 November 2011. https://web.archive.org/web/20111117174948/http://sirfer.utah.edu/qaqc.pdf. Retrieved 18 March 2020.
- ↑ Hitchcock, N. (2005). "Chapter 10: Efficient utilization of LIMS data and integration with mining process management systems". In Dessureault, S.D.; Ganguli, R.; Kecojevic, V.; Dwyer, J.G.. Application of Computers and Operations Research in the Mineral Industry. Taylor & Francis. pp. 85–88. ISBN 9780415374491.
- ↑ Turner, E.; Bolton, J. (2001). "Required steps for the validation of a Laboratory Information Management System". Quality Assurance 9 (3–4): 217–224. doi:10.1080/713844028. PMID 12553085.
- ↑ Al-Issa, Y.; Ottom, M.A.; Tamrawi, A. (2019). "eHealth Cloud Security Challenges: A Survey". Journal of Healthcare Engineering 2019: 7516035. doi:10.1155/2019/7516035. PMC PMC6745146. PMID 31565209. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6745146.
- ↑ "3.15 Explore Tables". LabWare, Inc. Archived from the original on 20 March 2017. https://web.archive.org/web/20170320223135/https://limshelp.labware.com/limsv5/V5_Manual_3_15_nbsp__Explore_Tables.htm. Retrieved 06 January 2022.
- ↑ Monkey, S. (23 January 2017). "The value of a control file when writing data files". ShortStories: Tales from software development. https://corengen.wordpress.com/2017/01/23/the-value-of-a-control-file-when-writing-data-files/. Retrieved 18 March 2020.
- ↑ Tsyktor, V. (30 December 2017). "How to Build a Data Backup Service for SaaS". Apriorit Dev Blog. https://www.apriorit.com/dev-blog/492-build-data-backup-service-saas. Retrieved 18 March 2020.
- ↑ "Environmental Monitoring". Core Informatics, LLC. Archived from the original on 27 February 2017. https://web.archive.org/web/20170227121045/http://corelims.com/products/core-lims-product-summary/features/environmental-monitoring/. Retrieved 06 January 2022.
- ↑ Orchard-Webb, D. (28 August 2019). "Matrix Gemini Biobank Management LIMS Includes New Monitoring And Maintenance Solutions". Biobanking.com. https://www.biobanking.com/matrix-gemini-biobank-management-lims-includes-new-monitoring-and-maintenance-solutions/. Retrieved 18 March 2020.
- ↑ "Novatek Environmental Monitoring". Novatek International. https://ntint.com/product/novatek-environmental-monitoring/. Retrieved 18 March 2020.
- ↑ 57.0 57.1 Gilles, C. (1 July 2008). "Bar Code and Sample Tracking: It All Starts with the Label". Scientific Computing. Archived from the original on 18 December 2010. https://web.archive.org/web/20101218190848/http://www.scientificcomputing.com/bar-code-and-sample-tracking.aspx. Retrieved 18 March 2020.
- ↑ "The Hidden Power of Barcodes". CSols, Inc. 8 February 2018. https://www.csolsinc.com/blog/hidden-power-barcodes/. Retrieved 18 March 2020.
- ↑ Haigney, S. (18 November 2018). "The Fax Is Not Yet Obsolete". The Atlantic. https://www.theatlantic.com/technology/archive/2018/11/why-people-still-use-fax-machines/576070/. Retrieved 18 March 2020.
- ↑ Pavlis, R. (May/June 2004). "Trends in instrument-to-LIMS interfacing". Scientific Computing World. Archived from the original on 10 September 2015. https://web.archive.org/web/20150910043807/http://www.scientific-computing.com/features/feature.php?feature_id=88. Retrieved 18 March 2020.
- ↑ DeHeer, L. (1 October 2009). "Instrument Interfacing - The Great Paradox of LIMS?". Blaze Systems Corporation. https://www.blazesystems.com/white-papers/instruments.html. Retrieved 18 March 2020.
- ↑ Curtis Jr., D. (12 December 2019). "Benefits and Best Practices for Integrating Laboratory Instruments with LIMS". Astrix Lab Informatics Blog. https://astrixinc.com/benefits-and-best-practices-for-integrating-laboratory-instruments-with-lims/. Retrieved 18 March 2020.
- ↑ Ryan, M. (28 August 2019). "Mobile LIMS Applications in the Laboratory". The Connected Lab. Thermo Fisher Scientific. https://www.thermofisher.com/blog/connectedlab/mobile-lims-applications-in-the-laboratory/. Retrieved 18 March 2020.
- ↑ "O3 LimsXpress". Bytewize AB. https://www.bytewize.com/o3limsxpress. Retrieved 18 March 2020.