Search results
From LIMSWiki
Jump to navigationJump to search
- |title_full = Guideline for software life cycle in health informatics |affiliations = Philipps University of Marburg, University Medical Center Göttingen61 KB (8,330 words) - 16:39, 29 April 2024
- ...ging systems. The DICOM standard has been instrumental in transforming the medical imaging world over the last three decades. Its adoption has been a signific ...ty]] management has not received enough attention in [[Clinical laboratory|medical laboratory]] education and tests in clinical [[biochemistry]] courses. Clin78 KB (10,739 words) - 15:33, 2 January 2024
- |title_full = An evaluation of regulatory regimes of medical cannabis: What lessons can be learned for the UK? |journal = ''Medical Cannabis and Cannabinoids''37 KB (5,382 words) - 23:56, 26 December 2023
- ...make decisions, in both diagnostics and therapeutics, built from existing medical evidence (i.e., knowledge-based [[Clinical decision support system|decision ...015 |pmid=25688383 |pmc=PMC4327872}}</ref> This fact reflects the need for medical practitioners, supported by their healthcare organizations, to make a shift32 KB (4,604 words) - 20:29, 13 March 2018
- |journal = ''BMC Medical Informatics & Decision Making'' ...oved significantly more than peers who used paper resources. When clinical guideline recommendations were presented on handheld computers, clinicians made signi51 KB (6,890 words) - 21:05, 28 December 2015
- ...ce]] (AI) has shown tremendous potential when applied in various fields of medical research and has opened a new era for precision medicine. The development o Here, we present a guideline for [[quality management system]]s (QMS) for academic organizations incorpo52 KB (7,400 words) - 18:25, 6 October 2021
- Appropriate [[laboratory]] [[Medical test|testing]] is critical in today's healthcare environment that aims to i ...ebatable, there is no question that laboratory diagnostics are central for medical practice today. The [[Coronavirus disease 2019|coronavirus disease 2019]] (93 KB (12,910 words) - 17:30, 10 August 2021
- ...e=Integrating the health-care enterprise pathology and laboratory medicine guideline for digital pathology interoperability |url=http://www.jpathinformatics.org ...given a set or requirements for the scanning phase (Table 4). As in other medical specialties, which have been dramatically changed by the introduction of a102 KB (14,629 words) - 22:54, 5 December 2021
- ...common, as many equate [[informatics]] with the management of [[electronic medical record]]s and [[Forensic science|forensics]] with the "criminal" investigat ...ucation in Clinical Informatics |author=Accreditation Council for Graduate Medical Education |date=03 February 2014 |accessdate=30 December 2014}}</ref> CI is35 KB (5,038 words) - 16:02, 28 March 2016
- ...ch as investigator, site coordinator, study monitor, data entry personnel, medical coder and statistician. Data should be organized in a format that facilitat ...dard CRF templates are inclusion criteria, exclusion criteria, demography, medical history, PE, AE, concomitant medication and study outcome modules, whereas,28 KB (4,313 words) - 20:51, 28 December 2015
- |journal = ''JMIR Medical Informatics'' ...care: a systematic review of the evidence |journal=Journal of the American Medical Informatics Association |author=Holroyd-Leduc, J.M.; Lorenzetti, D.; Straus75 KB (9,822 words) - 20:59, 28 December 2015
- ...guided this scoping review. A combination of keywords, Boolean terms, and medical subject headings was used to search [[PubMed]], EBSCOhost (CINAHL with full ...se, Infant Injury Database, Ibadan pilot fatal injury surveillance system, Medical Examiners and Coroners Alert System, National Violent Deaths Reporting Syst85 KB (10,856 words) - 23:13, 24 February 2023
- ...re evidence-based and user-friendly for individuals of varying digital and medical literacy. Indeed, studies suggest electronic healthcare tool development pr ...sults, increase accuracy in test result interpretation, and aid in complex medical diagnostic management. These resources can be useful to clinicians, [[labor35 KB (4,683 words) - 18:20, 17 July 2022
- Journal:Practical considerations for laboratories: Implementing a holistic quality management system...there are many QMS frameworks (e.g., a model such as a consensus standard, guideline, or regulation) that may apply to laboratories, ensuring that the appropria ...Errors within the total laboratory testing process, from test selection to medical decision-making - A review of causes, consequences, surveillance and soluti36 KB (5,066 words) - 18:35, 31 January 2023
- |journal = ''BMC Medical Informatics and Decision Making'' ...inical laboratory |author=Laposata, M. |edition=3rd |publisher=McGraw-Hill Medical |year=2019 |isbn=9781260116793}}</ref>104 KB (13,305 words) - 23:06, 4 August 2020
- |affiliations = Isfahan University of Medical Sciences, Southwest College of Naturopathic Medicine ...ons. Several countries and states have facilitated the use of THC-dominant medical cannabis for certain conditions, while other countries continue to ban all142 KB (19,594 words) - 22:55, 27 December 2023
- ...10.1002/cbdv.200790144}}</ref> Although, there is a lack of evidence-based medical information that can prove the potential benefit of the therapy with [[Cann ...(cannabis floss), published by the German Federal Institute for Drugs and Medical Devices (BfArM) in the ''German Pharmacopoeia''<ref>{{Cite web |last=Bundes45 KB (6,145 words) - 23:50, 26 December 2023
- |journal = ''Journal of Medical Biochemistry'' ...15189 as the most appropriate and useful standard for the accreditation of medical laboratories. In fact, only this international standard recognizes the impo31 KB (4,315 words) - 20:50, 26 November 2018
- ...C6697544 |pmid=31419828}}</ref> Subareas of health informatics may include medical informatics, nursing informatics, [[Consumer health informatics|consumer in .../www.thieme-connect.de/DOI/DOI?10.15265/IYS-2016-s005 |journal=Yearbook of Medical Informatics |language=en |volume=25 |issue=S 01 |pages=S42–S47 |doi=10.15111 KB (15,972 words) - 18:30, 6 October 2021
- ...instances of standards, regulations, and guidance documents affecting how medical diagnostic and research laboratories should operate. This collection doesn' ! style="color:brown; background-color:#ffffee;"|Relevant medical or research field(s)25 KB (3,239 words) - 00:08, 20 January 2022
- ...c.oup.com/jamia/article/28/9/1874/6321440 |journal=Journal of the American Medical Informatics Association |language=en |volume=28 |issue=9 |pages=1874–1884 ...transformation will have an impact on the care of patients treated by all medical and surgical specialists.94 KB (13,139 words) - 16:21, 12 April 2022
- ...cience for active surveillance: rationale and design for the Observational Medical Outcomes Partnership |journal=Annals of Internal Medicine |author=Stang, P. ...ements within the data while restricting access to private sections of the medical data. These mid-level approaches facilitate collaboration both within the i42 KB (6,110 words) - 17:06, 14 March 2017
- ...keholders for future discussions related to regulation of [[laboratory]] [[Medical test|tests]]. The current state of [[laboratory]] [[Medical test|test]] regulation in the United States (U.S.) is complex, and the pros52 KB (7,250 words) - 18:46, 16 June 2020
- |journal = ''JMIR Medical Informatics'' ...ation]] professionals play a key role in closing the knowledge gap between medical research and clinical practice. Their work involves meticulous searching of49 KB (6,622 words) - 19:45, 19 September 2021
- ...e development of an integrated platform at a midlevel Appalachian academic medical center, along with its initial uses ... ('''[[Journal:Fueling clinical and [[Data sharing|Information sharing]] across medical institutions is restricted to [[information]] exchange between specific par79 KB (10,982 words) - 23:51, 11 April 2024
- ...ew York is used, as New York regulations are strict, and using a stringent guideline for the rest of this guide is preferred: * '''Pediatricians''': Pediatricians are trained to treat medical conditions found in infants, children, and adolescents. Generally, they do57 KB (8,487 words) - 18:48, 9 February 2022
- :[[Medical diagnosis]] :[[Medical research]]6 KB (645 words) - 14:40, 7 December 2018
- |affiliations = Paracelsus Medical University ...ps://pmj.bmj.com/lookup/doi/10.1136/pgmj.2006.049551 |journal=Postgraduate Medical Journal |language=en |volume=82 |issue=974 |pages=823–829 |doi=10.1136/pg74 KB (10,573 words) - 20:34, 9 December 2021
- Diagnostic investigations (i.e., pathology [[laboratory]] analysis and [[medical imaging]]) aim to increase the certainty of the presence of or absence of d Diagnostic investigations and tests (e.g., pathology and [[medical imaging]]) involve the observation of personal characteristics, symptoms, s31 KB (4,246 words) - 16:14, 29 June 2021
- |journal = ''Journal of Medical Biochemistry'' ...study evaluation in two private [[hospital]]s and a [[Clinical laboratory|medical laboratory]]. We collected data using interviews, observations, and documen48 KB (6,610 words) - 23:29, 27 October 2021
- ...al data to be released without manual verification. With new advances in [[medical informatics]], the [[laboratory information system]] (LIS) has growing pote ...perating protocols for the optimization of Cannabis-based formulations for medical purposes]]"'''83 KB (11,691 words) - 15:36, 2 January 2024
- ...ih.gov/iccvam/suppdocs/feddocs/oecd/oecd_gl420.pdf |format=PDF |title=OECD Guideline for Testing of Chemicals |author=Organisation for Economic Co-operation and ...me. Practical laboratory examples include testing packaging for sterilized medical devices<ref name="NelsonAccel">{{cite web |url=https://www.nelsonlabs.com/T102 KB (13,327 words) - 20:58, 19 September 2021
- ...al of the information needed by physicians to make patient care decisions. Medical and technical advances in genomics that require processing of an increased The creation of medical notes in software applications poses an intrinsic problem in workflow as th88 KB (12,401 words) - 19:57, 3 April 2024
- ...he fastest growing disciplines in [[clinical laboratory]] medicine. POCT [[Medical device|devices]] are widely used in both acute and chronic patient manageme ...''': point-of-care testing, data management, quality assurance, electronic medical record45 KB (6,030 words) - 19:45, 10 November 2020
- <blockquote>'''Figure 1.''' Common pipeline of processing free text medical report</blockquote> ...mens in support of translational research |journal=Journal of the American Medical Informatics Association |author=Crowley, R.S.; Castine, M.; Mitchell, K.; C30 KB (4,294 words) - 21:01, 28 December 2015
- ...rchive.org/web/20190612144526/https://www.leafly.com/news/politics/arizona-medical-marijuana-fees-lowered-testing-added |title=Arizona Just Made Cannabis Safe ...ite web |url=https://www.bplans.com/laboratory-business-plan/ |title=Fargo Medical Laboratories Laboratory Business Plan |work=Bplans.com |publisher=Palo Alto110 KB (14,785 words) - 19:00, 11 April 2024
- ...ref name="WeiskopfAData17">{{cite journal |title=A Data Quality Assessment Guideline for Electronic Health Record Data Reuse |journal=EGEMS |author=Weiskopf, N. ...819-153-9-201011020-00010 |pmid=21041580}}</ref>, with an initial focus on medical product safety surveillance. The OHDSI portfolio also includes work on comp32 KB (4,578 words) - 15:35, 11 August 2020
- ...safety-white-paper-2019.pdf |format=PDF |title=Interim Clinical Laboratory Guideline for Biological Safety |author=Buchan, B.W.; Mahlen, S.D.; Relich, R.F. |pub ...ite web |url=https://www.iso.org/standard/56115.html |title=ISO 15189:2012 Medical laboratories — Requirements for quality and competence |publisher=Interna19 KB (2,564 words) - 23:44, 18 September 2021
- |journal = ''BMC Medical Informatics and Decision Making'' |affiliations = Jinan Kingmed Center for Clinical Laboratory, Guangzhou Medical University48 KB (6,399 words) - 18:33, 10 June 2021
- ...}</ref> This, though, is the case for many other areas of research such as medical or social sciences, and the scripts need not be different in cybersecurity. .../ |title=Researchers: MedSec, Muddy Waters Set Bad Precedent With St. Jude Medical Short |author=Spring, T. |work=Threat Post |date=31 August 2016 |accessdate67 KB (9,997 words) - 21:00, 19 September 2021
- ...forms of hardware than electronic hardware, like mechanical, construction, medical, optical, agricultural, or textile hardware. From a design point-of-view, t ...satisfies this criterion when all published content is editable or when a guideline for participation is available.105 KB (14,517 words) - 18:47, 6 October 2021
- *During the same 36 years, at Mount Sinai Medical Center's chemistry department, the productivity (indicated as the number of ...we showed a bulleted list of considerations for the development of such a guideline-based framework. What follows is a more organized version of those points,121 KB (18,783 words) - 01:40, 28 March 2024
- |affiliations = University of Rostock, University Medical Center Rostock, University of Amsterdam ...esearch findings. This does not restrict researchers but rather provides a guideline based on earlier experiments. Specifically, they include parameters, timest124 KB (16,896 words) - 16:29, 29 April 2024
- ...clinical variables and hundreds of gene expression signatures |journal=BMC Medical Genomics |author=Fan, C.; Prat, A.; Parker, J.S. et al. |volume=4 |pages=3 ...ta submitter during the submission process. Predicting metadata could be a guideline for template authors during the process of metadata definition. This facili51 KB (7,302 words) - 16:16, 23 August 2017
- ...become more prominent in the past decade, as they may provide a practical guideline to properly detect the abuse of cannabinoids in forensic cases. To better u ...eral, including noninvasive collection methods, no requirement for trained medical professionals, and the possibility of multiple sample collection allowing f85 KB (10,974 words) - 23:49, 26 December 2023
- ...title=Validating Whole Slide Imaging for Diagnostic Purposes in Pathology: Guideline from the College of American Pathologists Pathology and Laboratory Quality ...stic setting: navigating through the implementation journey in an academic medical centre |url=http://jcp.bmj.com/lookup/doi/10.1136/jclinpath-2015-203600 |jo83 KB (11,688 words) - 21:14, 11 April 2024
- ...tos Consultants, UNIFESP Foundation, Uppsala University, Leiden University Medical Center, Bayer CropScience, University of Basel, Cray, University of Groning ...efer to as the FAIR Data Principles. The intent is that these may act as a guideline for those wishing to enhance the reusability of their data holdings. Distin49 KB (7,038 words) - 16:24, 29 April 2024
- ...[informatics]] (e.g., public health, population health, nursing, clinical, medical, health, consumer, and biomedical) centers on the "application of informati ...disease outbreaks, health or food recalls, medical errors, deviations from guideline concordant care, risk behaviors linked to disease spread, budget shortfalls67 KB (9,519 words) - 18:29, 25 November 2019
- ...butes [[laboratory informatics]] software solutions for pharmaceutical and medical device companies in over 30 countries around the world.<ref name="HAHist">{ ...|accessdate=02 October 2021}}</ref> The CDMS is "FDA-regulations and GAMP guideline validated" and complies with [[21 CFR Part 11]] rules with audit trails and9 KB (1,043 words) - 18:59, 27 March 2024
- ...n |first3=Rizal |date=2020-03-02 |title=Architectural Design of Electronic Medical Record (EMR) using Enterprise Architecture Planning (EAP) Method |url=https ...eater purpose. Determining the future vision and mission is necessary as a guideline for determining various IT strategies needed to support the vision and miss45 KB (5,954 words) - 18:04, 30 May 2023
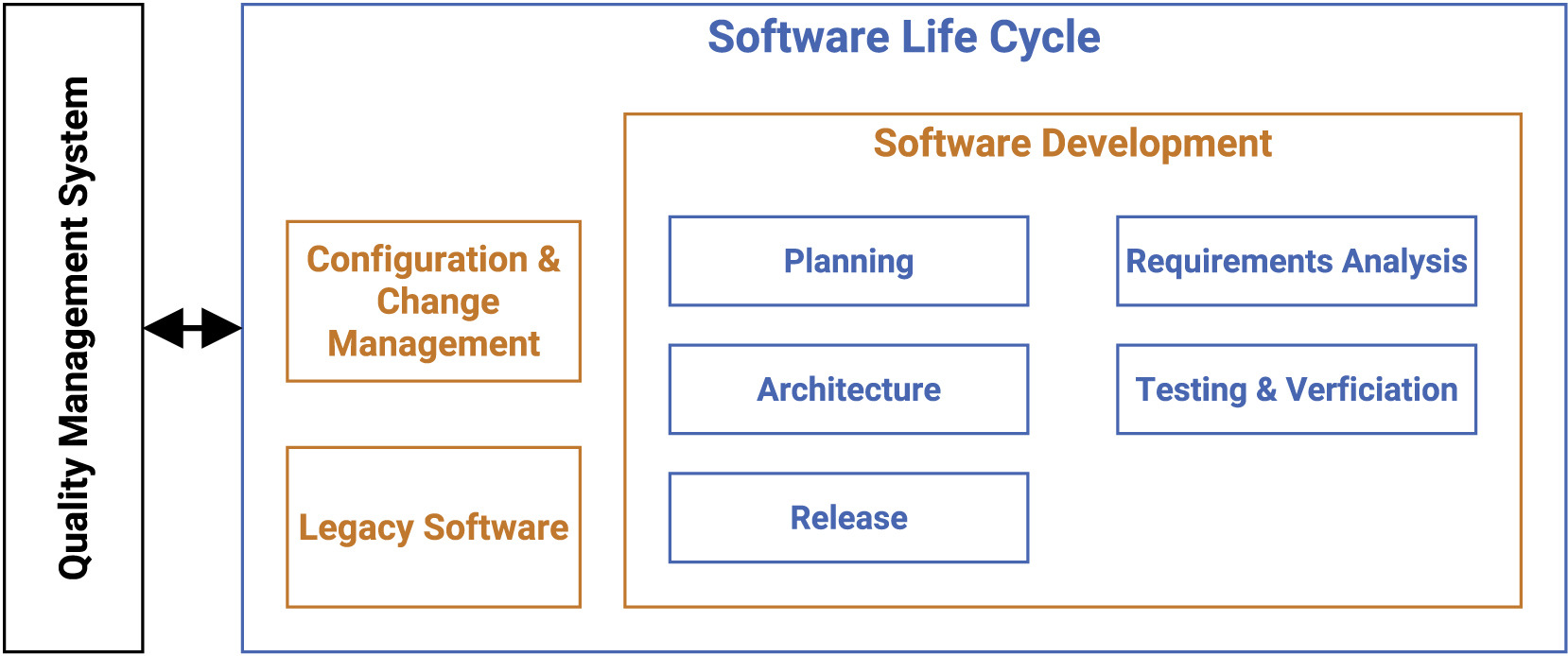
File:Fig1 Hauschild iScience2022 25-12.jpg ...1.''' Main components of the software life cycle (SLC) for research-based medical software |Source={{cite journal |title=Guideline for software life cycle in health informatics |journal=iScience |author=Hau(1,658 × 694 (174 KB)) - 16:49, 4 July 2023- ...owever, the success of such organization lies in the responsiveness of the medical teams, who should act upon the results soon after delivery by the laborator ...nosis and management of infections caused by multidrug-resistant bacteria: guideline endorsed by the Italian Society of Infection and Tropical Diseases (SIMIT),55 KB (7,348 words) - 21:39, 1 August 2023
- ...ption in the pharmaceutical and life sciences industry |journal=Journal of Medical Marketing: Device, Diagnostic and Pharmaceutical Marketing |author=Subraman It is important to note that the reference model and guideline recommended in the ANSI/ISA-88 standard are not to be strictly normative. T60 KB (8,463 words) - 16:32, 19 April 2024









